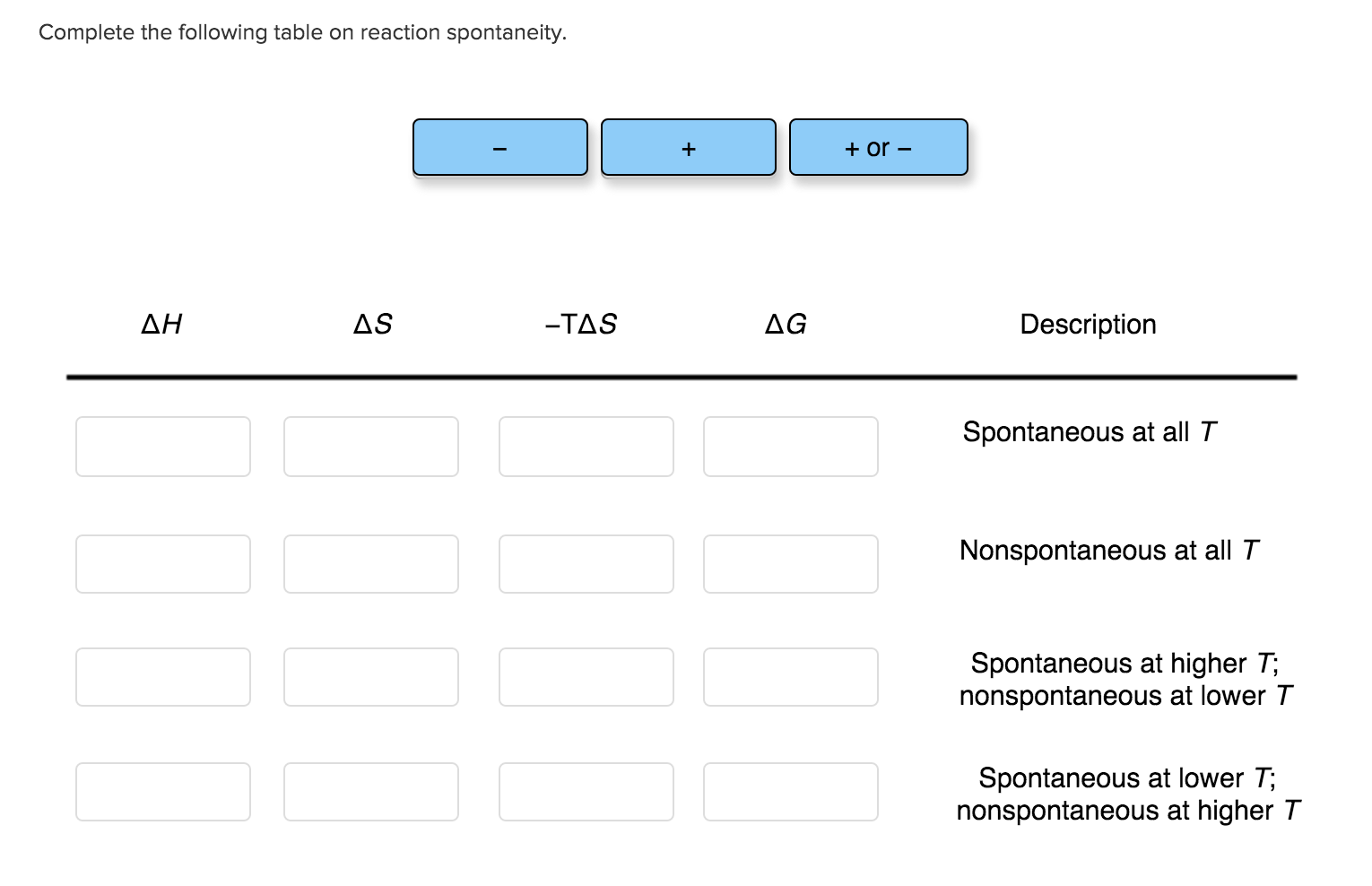
Complete The Following Table On Reaction Spontaneity
Ever wondered why some things just happen without you lifting a finger, while others require Herculean effort? Think about your laundry basket – it seems to spontaneously overflow, doesn't it? Or that perfectly ripe avocado that turns brown before you even get a chance to enjoy it? Well, my friends, science has a name for this delightful (and sometimes maddening) tendency of the universe: Reaction Spontaneity! It’s like the universe’s own "easy button" for certain events.
Now, don't let the fancy word "spontaneity" scare you. We're not talking about juggling flaming torches without practice here. We're talking about whether a reaction, a change, a transformation, is going to occur on its own, given the right conditions. Think of it as the universe’s internal "yes" or "no" signal. Some reactions are a resounding "YES! Let’s do this!", while others are a grumpy "Ugh, fine, but only if you really, really push me."
To help us sort out this cosmic tendency, we’ve got a super-duper handy table. It’s like a cheat sheet for predicting the universe's mood. It breaks down spontaneity based on two key players: Enthalpy Change (ΔH) and Entropy Change (ΔS).
Let’s break down these two characters. Enthalpy (ΔH) is all about the energy of a reaction. Is it releasing energy (like a perfectly toasted marshmallow, nice and warm)? That’s an exothermic reaction, and we often denote it with a negative ΔH. It’s like the universe giving you a warm hug. On the flip side, is it taking in energy (like trying to freeze a popsicle on a sweltering day)? That’s an endothermic reaction, with a positive ΔH. Think of it as the universe asking for a chilly boost.
Then we have Entropy (ΔS). This is the universe’s love for chaos and disorder! Imagine your bedroom after a particularly enthusiastic game of LEGOs. That's high entropy – everything is spread out, a glorious mess. A positive ΔS means things are getting more spread out, more disordered. Now, picture a perfectly organized deck of cards, all sorted by suit and number. That’s low entropy – neat and tidy. A negative ΔS means things are becoming more ordered, more structured. It’s like your mom tidying up your room – a definite decrease in entropy!

So, how do these two interact to decide if something is spontaneous? Our table is about to reveal all! We’re going to look at four glorious scenarios, each with its own personality.
First up, we have the dynamic duo: Exothermic (ΔH is negative) and Increasing Entropy (ΔS is positive). This is the universe's dream team, the ultimate "easy button" scenario! Think about a firework exploding. It releases heat (exothermic) and the smoke and sparks go everywhere in a beautiful display of chaos (increasing entropy). This reaction is practically shouting, "YES! Let's do this spontaneously!" It’s always, always spontaneous. No ifs, ands, or buts. This is like finding a forgotten ten-dollar bill in your pocket – pure, unadulterated, spontaneous joy!

Next, we have a slightly less enthusiastic pairing: Endothermic (ΔH is positive) and Decreasing Entropy (ΔS is negative). This is the universe saying, "Are you kidding me?" This is the opposite of spontaneous. It requires a massive amount of effort, like trying to un-melt an ice cream cone in the desert. This reaction is stubbornly non-spontaneous, no matter what. It’s the equivalent of your alarm clock going off on a Monday morning – you have to drag yourself out of bed, and the universe isn’t going to help.
Now, things get a little more interesting. Scenario three: Exothermic (ΔH is negative) but Decreasing Entropy (ΔS is negative). This is a bit of a mixed bag. Think of freezing water into ice. It releases heat (exothermic), but the water molecules become more ordered (decreasing entropy). Whether this happens spontaneously depends on the Temperature. At low temperatures, the urge to release heat wins, and it’s spontaneous. Imagine a cold, crisp winter day – ice forms naturally! But at higher temperatures, the drive for order is overwhelmed, and it’s non-spontaneous. That ice cube is not going to freeze itself on a hot summer day, no matter how much it wants to release energy. It needs a little help from your freezer!
Finally, we have the wild card: Endothermic (ΔH is positive) and Increasing Entropy (ΔS is positive). This is another temperature-dependent situation. Think about dissolving salt in water. It often takes a bit of heat (endothermic), but the salt crystals break apart and spread throughout the water, becoming more disordered (increasing entropy). At high temperatures, the drive for chaos wins, and it’s spontaneous. Imagine stirring sugar into your tea on a warm afternoon – it dissolves beautifully! But at low temperatures, the need for heat is too great, and it’s non-spontaneous. That sugar won’t dissolve itself in an ice-cold drink.
So, there you have it! The universe’s little decision-making process for spontaneity. It’s not magic, it’s just a dance between energy and disorder, influenced by the temperature of the party. Understanding this table is like having a secret decoder ring for why things happen the way they do. Now go forth and impress your friends with your newfound knowledge of cosmic tendencies!
