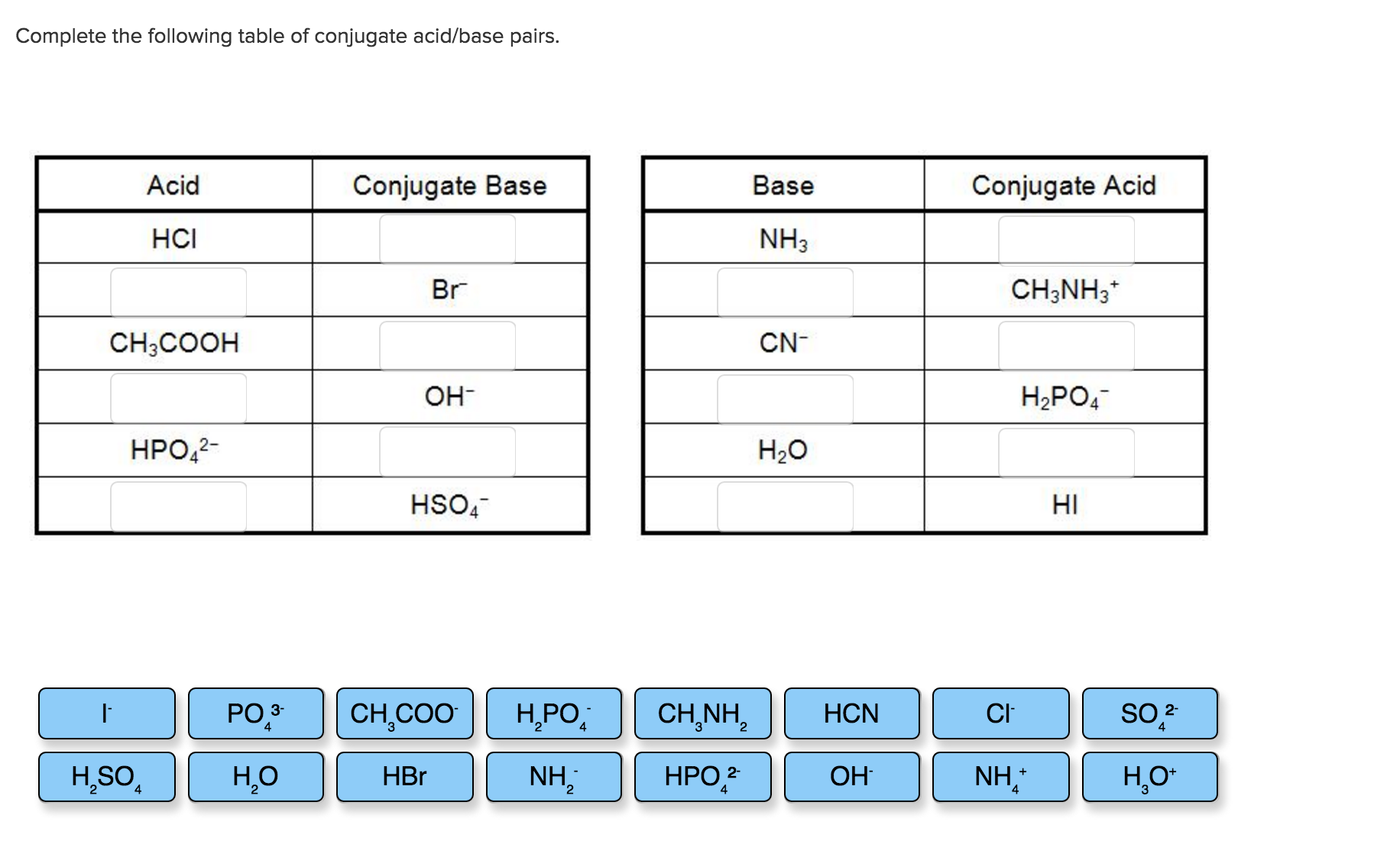
Complete The Following Table Of Conjugate Acid/base Pairs.
Ever found yourself staring at a chemistry textbook, perhaps even a little intimidated by all the talk of acids and bases? Don't worry, you're not alone! But what if I told you that understanding these seemingly complex concepts could actually be… fun? And not just fun, but also surprisingly relevant to the world around us? Well, get ready to peek behind the curtain of conjugate acid-base pairs, a neat little concept that unlocks a deeper understanding of how acids and bases behave.
So, what's the big deal about these pairs? Think of it like a chemical dance. When an acid loses a proton (that's a hydrogen ion, H+), it transforms into its conjugate base. And conversely, when a base gains a proton, it becomes its conjugate acid. They're essentially two sides of the same coin, forever linked by the simple act of proton exchange. Learning to identify these pairs helps us predict how substances will react in water and other solutions, which is a pretty big deal in chemistry!
The real purpose of this table of conjugate acid-base pairs is to serve as a handy reference. It's like having a cheat sheet that tells you, "Okay, if you see this acid, its partner in the dance will be that base." This allows us to understand the relative strengths of acids and bases. Strong acids have weak conjugate bases, and strong bases have weak conjugate acids. It's a beautiful balance! The benefits are manifold: improved problem-solving skills in chemistry, a better grasp of chemical reactions, and the ability to predict outcomes with more confidence.
Where do we see this in action? In education, it's a cornerstone of acid-base chemistry. Students learn to complete these tables to solidify their understanding of equilibrium and reaction mechanisms. But it’s not just for the classroom! Think about everyday life. The pH of our blood, for instance, is carefully regulated by buffer systems, which are essentially conjugate acid-base pairs. Without them, our bodies couldn't function! Antacids, those handy tablets for indigestion, work by neutralizing excess stomach acid, often involving conjugate pairs. Even the way your shampoo is formulated to be gentle on your hair involves understanding these chemical relationships.
Ready to explore this yourself? It’s simpler than you might think! Grab a periodic table and a list of common acids and bases. The next time you encounter an acid, like hydrochloric acid (HCl), ask yourself: "If HCl loses a proton, what's left?" The answer is chloride ion (Cl-), its conjugate base. Similarly, if you see ammonia (NH3), ask: "If NH3 gains a proton, what does it become?" You'll get the ammonium ion (NH4+), its conjugate acid. You can find many online resources and tables that list these pairs, making it easy to practice. Try identifying the pairs in common household substances, and you might be surprised by how often these chemical partners are at play!
