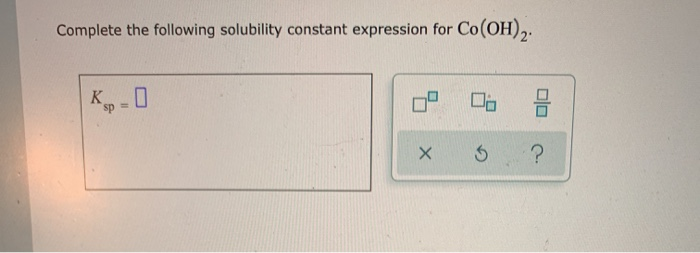
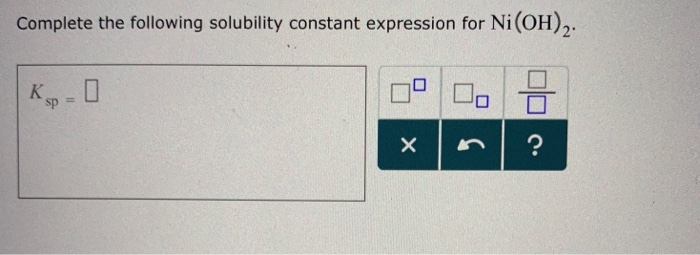
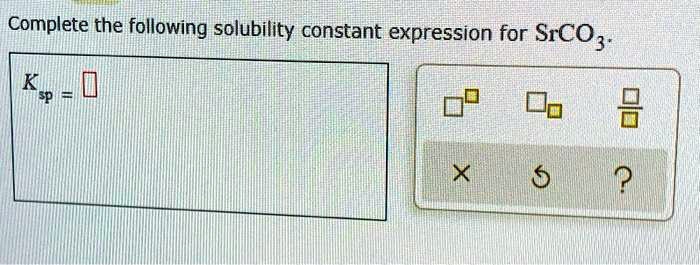
Complete The Following Solubility Constant Expression For
Ever found yourself staring at a chemistry problem and feeling a tiny bit lost? We've all been there! But what if I told you there's a way to make those tricky solubility questions not just understandable, but genuinely fun? Get ready to dive into the wonderfully weird world of Ksp!
Think of Ksp as a secret handshake for how much of a solid can actually dissolve in water. It’s like a tiny, invisible rulebook for saltiness. Some salts are super social and dissolve easily, while others are more like lone wolves, sticking together in their solid form.
Today, we're going to play a game of "Complete the Expression." It sounds fancy, but it's more like a puzzle. We're going to look at a chemical reaction where a solid is trying its best to break apart into its tiny ion buddies in water.
The Great Dissolving Race
Imagine you have a little pebble of, let’s say, Silver Chloride. We'll call it AgCl for short. This pebble is chilling in a glass of water. At first, it just sits there, all solid and unbothered.
But, if you wait long enough, or if the water is really patient, a few of the AgCl bits will start to get lonely. They’ll break off from the main pebble and float around as individual Silver ions (Ag+) and Chloride ions (Cl-). This is the "dissolving" part.
Now, there's a limit to how many of these ions can hang out in the water. Once the water is "full" of these particular ions, the AgCl pebble will just sit there, looking smug, while the few dissolved ions happily bounce around. It’s a dynamic balance, like a seesaw that’s perfectly still because it’s got equal weight on both sides.
Introducing the Star of the Show: Ksp!
This is where our hero, Ksp, makes its grand entrance. Ksp stands for the "Solubility Product Constant." Catchy, right? It’s basically a number that tells us how much dissolving is happening at that perfect balance point.
A big Ksp means the solid is pretty happy to dissolve. It's like saying, "Come on in, water, there's plenty of room for my ions!" A small Ksp means the solid is a bit shy and doesn't want to let go of its ions very easily. It's more like, "Uh, maybe just one or two of you can come out, but don't get too comfortable."

So, for our AgCl dissolving in water, the reaction looks something like this:
AgCl (s) ⇌ Ag+ (aq) + Cl- (aq)
See the little "(s)" next to AgCl? That means it's a solid. And the "(aq)" next to Ag+ and Cl- means they are dissolved in water, or "aqueous." The little arrows going in opposite directions mean it’s a reversible reaction – dissolving and re-forming are happening at the same time.
Completing the Ksp Expression: It's Easier Than You Think!
Now, let's get to the fun part: completing the solubility constant expression for this very reaction. Remember how Ksp is a number that describes the balance? We write it as an equation.
For any solid dissolving, the Ksp expression is generally the product of the concentrations of its dissolved ions. Think of it as multiplying how many of each type of ion you have floating around.
So, for our AgCl equation, we have two ions: Ag+ and Cl-. To write the Ksp expression, we just take the concentration of Ag+ and multiply it by the concentration of Cl-. We use square brackets, like [Ag+], to mean "the concentration of Ag+."
So, the Ksp expression for AgCl is:
Ksp = [Ag+] [Cl-]
And there you have it! You've just completed a solubility constant expression. How cool is that? You're basically reading the secret code of how easily a solid dissolves.
Let's Try Another One: Calcium Phosphate Fun!
Ready for a slightly more complex but equally entertaining challenge? Let's look at Calcium Phosphate. Its chemical formula is Ca3(PO4)2. This is a much bigger solid molecule.
When Ca3(PO4)2 dissolves in water, it breaks into a whole bunch of calcium ions and phosphate ions. For every one unit of Ca3(PO4)2 that dissolves, you get three calcium ions (Ca2+) and two phosphate ions (PO4^3-).
The dissolving reaction looks like this:

Ca3(PO4)2 (s) ⇌ 3Ca^2+ (aq) + 2PO4^3- (aq)
Notice the numbers "3" and "2" in front of the ions? Those are super important! They tell us how many of each ion are produced when the solid breaks apart.
Now, when we write the Ksp expression, we have to account for these numbers. We raise the concentration of each ion to the power of the number of those ions produced. It’s like giving each ion a "multiplier" based on how many there are.
So, for Calcium Phosphate, the Ksp expression will be:
Ksp = [Ca^2+]³ [PO4^3-]²
See how we used the '3' as an exponent for [Ca^2+] and the '2' as an exponent for [PO4^3-]? This is the magic of completing the Ksp expression! You're not just listing the ions; you're showing their proportions in the dissolved state.
Why Is This So Entertaining?
You might be thinking, "Okay, but why is this entertaining?" Well, it's all about the puzzle-solving aspect! It's like deciphering a hidden message. Each chemical formula gives you clues about how its ions will behave in water.
It's also incredibly satisfying when you finally get the expression right. It’s a small victory in the grand scheme of chemistry, but a victory nonetheless! You've tamed a little piece of the chemical world.
Plus, think about the real-world implications. Understanding Ksp helps scientists figure out how medicines dissolve in our bodies, how minerals form, and even how pollution might spread in water. It’s not just abstract numbers; it’s a key to understanding the world around us.
The Special Spark of Ksp
What makes Ksp special is its elegance. It takes a complex process – a solid breaking apart into ions – and boils it down to a simple, quantifiable relationship. It's a snapshot of equilibrium, a moment of perfect balance.
It's also incredibly versatile. Whether you're dealing with common table salt or some obscure mineral, the concept of Ksp and its expression remains the same. You just need to know the chemical formula and how it dissociates.
So, next time you encounter a solubility problem, don't shy away. Embrace the challenge! Think of yourself as a chemical detective, ready to complete the solubility constant expression and unlock the secrets of dissolving. It's a tiny bit of magic, and it’s surprisingly enjoyable. Give it a try, and you might just find yourself hooked!
