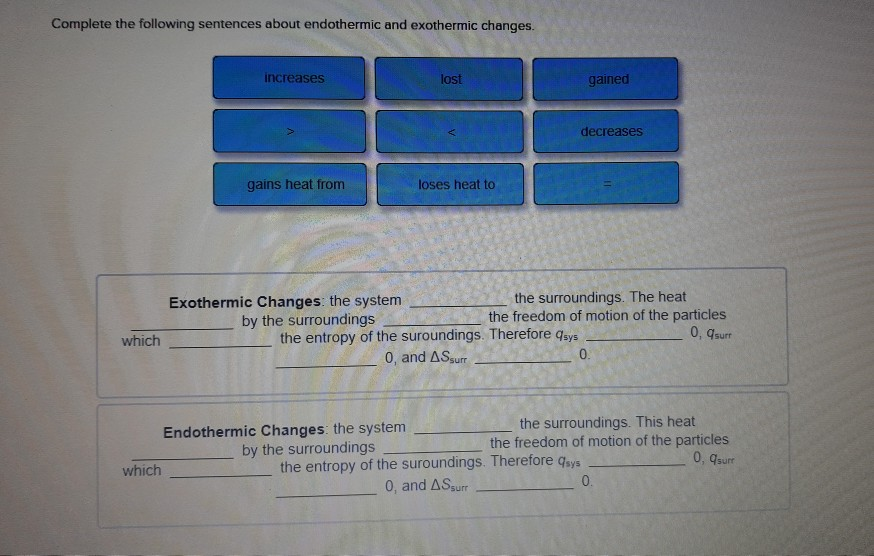
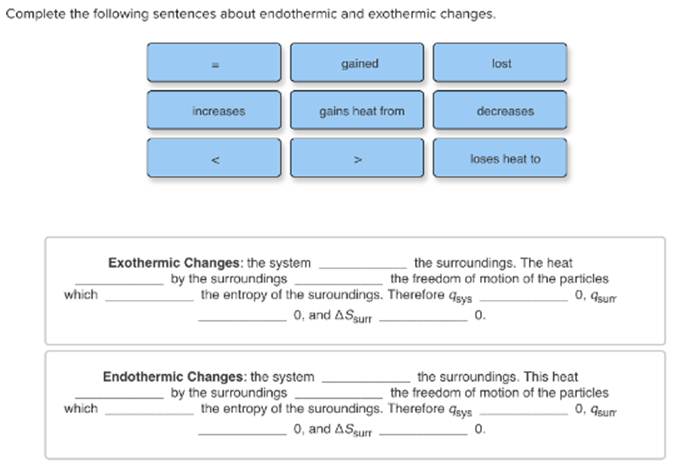
Complete The Following Sentences About Endothermic And Exothermic Changes

Let's dive into the wonderfully weird world of how things change, and how they feel when they do! Sometimes, when stuff happens, it gets all toasty. Other times, it feels like a tiny ice wizard just blasted the area. We're going to finish some sentences to explore these awesome energy shifts.
When a chemical reaction or a physical change happens and it feels warm to the touch, it's usually because of something called an endothermic process. Think of it like a thirsty sponge – it’s really good at soaking up heat from its surroundings. This is why when you’re making slime, and the mixture gets a little cool in your hands, you know it's busy being endothermic.
Complete this sentence: An endothermic change is like a party guest who always brings a cooler, because it ____.
...loves to soak up all the warmth from the room, leaving everyone else feeling a little chilly!
Imagine your hands on a cold winter day. You rub them together, right? That’s creating friction, and while it makes them warmer, the process of generating that movement could be considered endothermic if we were looking at the tiny molecules involved. But in the grand scheme of things, rubbing your hands is more about kinetic energy becoming thermal energy. For our fun sentences, let’s focus on the big picture feel!
When you dissolve an Alka-Seltzer tablet in water, have you ever noticed how it gets a bit cool? That’s a classic endothermic moment happening right in your glass! The tablet is grabbing that heat from the water to make its fizzy magic happen. It’s like the tablet is saying, "Give me all your heat, I need it for this spectacular show!"
Complete this sentence: For a plant to grow, it needs sunlight, and this process of photosynthesis is endothermic because it ____.
...absorbs energy from the sun to create food, like a tiny solar-powered chef!
using your knowledge of endothermic and exothermic reactions, complete
Think about baking a cake. When you put the batter into a hot oven, the batter absorbs heat. This is a crucial part of the transformation. The heat energy is what causes the chemical reactions to occur, making the cake rise and turn golden brown. So, the baking process itself, from the batter's perspective, is endothermic!
Have you ever used those instant cold packs for a bump or a bruise? You snap them, and then whoosh, they get really cold! That’s a super-fast endothermic reaction at work. The chemicals inside are desperate for energy, and they snatch it right from the air and your skin, making you feel that satisfying chill.
Complete this sentence: When ice melts into water, it's an endothermic process because the ice ____.
...needs to absorb heat from its surroundings to break those chilly bonds and become liquid, like waking up from a long, cold nap!
Now, let’s switch gears to the other side of the energy coin: exothermic changes! These are the ones that give us warmth. Think of a cozy campfire on a cool evening. That lovely heat you feel? That’s exothermic energy being released!

Complete this sentence: An exothermic change is like a hug that gives you extra warmth because it ____.
...releases a burst of heat energy, making everything around it feel wonderfully toasty and cheerful!
When wood burns in a fireplace, it’s a perfect example of an exothermic reaction. The chemical bonds in the wood are breaking and rearranging, and in the process, they release a ton of energy as heat and light. It’s the universe saying, "Here, have some free warmth and a little light show!"
Think about lighting a match. The initial strike gives it the spark, but the burning itself is exothermic. That little flame dances and produces heat, allowing you to light your candle or your campfire. It’s a small but mighty exothermic event!
Complete this sentence: When you mix certain chemicals together and they get hot, it’s because the reaction is exothermic and ____.
...is happily giving away energy in the form of heat, like a generous friend sharing their warmth!
You know when you’re making hot chocolate from scratch, maybe by dissolving a chocolate bar in milk and heating it? The act of dissolving the chocolate and the milk warming up is all part of a process that feels good and warm. While the primary heating is from an external source, the chemical interactions can also contribute to the overall exothermic feeling of enjoying a warm drink.
Have you ever seen those hand warmers you can buy in the winter? You activate them, and they get hot for hours! That’s a controlled exothermic reaction, usually involving iron powder oxidizing. It’s like a tiny, portable sun you can hold in your glove.
Complete this sentence: A combustion engine in a car works by releasing energy through exothermic reactions, which is why ____.
...all those explosions inside are what ultimately power your drive, turning fuel into movement and heat!
Solved Complete the following sentences about endothermic | Chegg.com
When you breathe, your body is constantly performing exothermic reactions to keep you alive and warm. Cellular respiration, where your cells break down glucose to get energy, releases heat as a byproduct. It’s your body’s own internal, ongoing, and essential exothermic party!
So, whether something is snatching heat like a thirsty sponge (endothermic) or giving it away like a warm hug (exothermic), these energy changes are happening all around us, all the time. They’re responsible for everything from ice cream melting on a hot day to the very warmth of our own bodies. It’s a surprisingly exciting world of energy at play!
Complete this sentence: Understanding whether a change is endothermic or exothermic helps us predict ____.
...whether we’ll need a sweater or a fan, and appreciate the fascinating ways energy shapes our world!