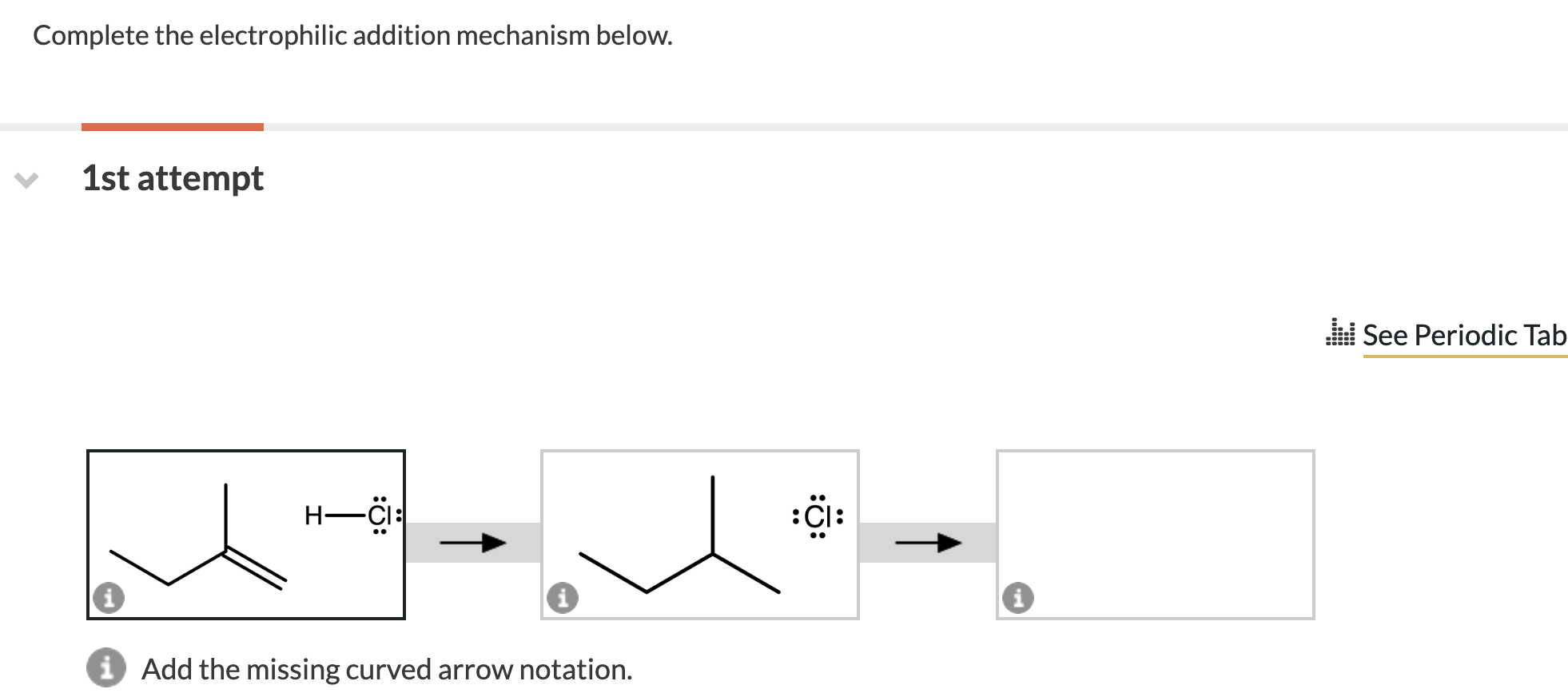
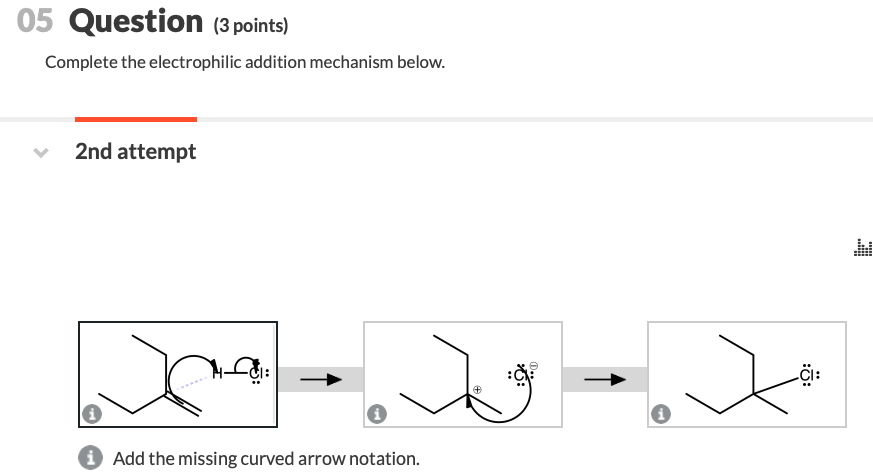
Complete The Electrophilic Addition Mechanism Below.
Ever felt like you were watching a tiny, molecular drama unfold? Well, get ready for one of the most captivating performances in the world of chemistry: the Electrophilic Addition Mechanism! It might sound super technical, but trust me, it's like a thrilling detective story happening at an unbelievably small scale. We're going to dive into a specific example, and I promise, by the end, you'll be looking at your everyday world with a whole new sense of wonder.
Imagine you've got this molecule. Let's call it Alkene X. Now, Alkene X is a bit like a shy teenager. It's got this double bond, which is basically two pairs of electrons hanging out together. These electrons are the life of the party, always eager to mingle. But they can also be a little vulnerable. That's where our star guest, the Electrophile, comes in.
Think of the Electrophile as a super popular kid at school. It's got a big appetite for electrons, a real "electron lover." It's positively charged, or it's missing electrons, making it desperately want to connect with that electron-rich double bond of Alkene X. It's like a magnetic pull, an irresistible attraction.
So, our shy Alkene X sees this flashy Electrophile approaching. The electrons in the double bond, feeling a bit bold, decide to make the first move. They reach out and grab onto the Electrophile. It's a swift, decisive action, like a quick handshake that turns into a hug. This is the very first step, the initial contact, and it's already got us on the edge of our seats!
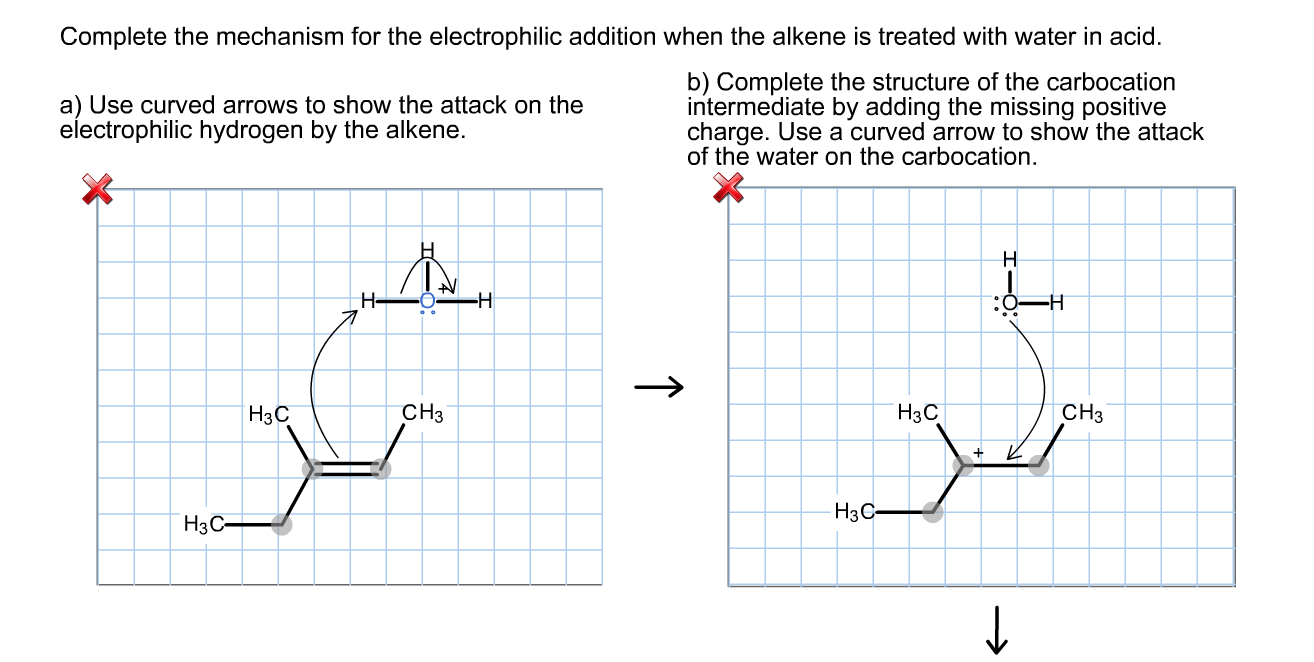
As the Electrophile bonds with one of the carbons in the original double bond, something pretty dramatic happens. The double bond breaks! One of the two electron pairs, the ones that formed the original double bond, is now completely dedicated to this new bond with the Electrophile. The other carbon that was part of the double bond is now a bit… lonely. It's lost its electron buddy, and as a result, it develops a positive charge. We call this intermediate a Carbocation.
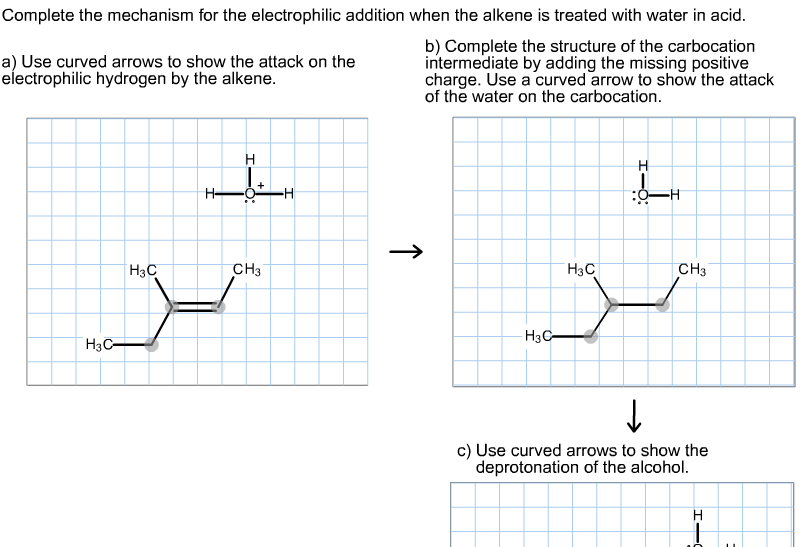
It's like a love triangle, but with atoms! The electrons, the Electrophile, and the carbons all playing their part.
Now, this Carbocation is unstable. It's like someone who just made a big decision and is feeling the pressure. It's got this positive charge, this "electron deficiency," and it needs to find stability. This is where the next character in our drama enters the scene: the Nucleophile.
If the Electrophile was the popular, electron-seeking kid, the Nucleophile is the generous, electron-donating friend. It's got extra electrons, a pair that it's just waiting to share. It's an "electron-rich" species, ready to swoop in and help out the struggling Carbocation. It's like a knight in shining armor, but with a pair of electrons instead of a sword.
The Nucleophile sees the positively charged carbon of the Carbocation. Boom! It attacks. Those extra electrons from the Nucleophile rush in and form a brand new bond with the positive carbon. It's a beautiful moment of connection and stabilization. The positive charge disappears, and both carbons involved in the original double bond are now happy, single-bonded to other atoms. The molecule has successfully undergone an addition reaction!
What makes this whole process so entertaining? It's the sheer dynamism. It's a step-by-step story of attraction, transformation, and resolution. You have the initial attraction between the electron-rich double bond and the electron-poor Electrophile. Then, the exciting formation of the unstable Carbocation, a fleeting moment of energetic tension. Finally, the triumphant arrival of the Nucleophile, bringing peace and stability to the system.
Each step is driven by fundamental principles of electron movement and stability. It's like watching a perfectly choreographed dance, where every move is dictated by the need to achieve a more stable electron configuration. The fact that these invisible forces are playing out constantly, all around us, in countless chemical reactions, is truly mind-boggling.
And the best part? This Electrophilic Addition Mechanism isn't just a theoretical concept. It's the backbone of so many important chemical processes. Think about how we make plastics, pharmaceuticals, and all sorts of everyday materials. This simple, yet elegant, mechanism is often at play. It's the unheralded hero behind a lot of modern life!
So, the next time you see a double bond, or hear about a chemical reaction, remember this little molecular soap opera. It's a story of desire, instability, and ultimate harmony. It's a reminder that even in the tiniest corners of existence, there are incredibly interesting and elegant dramas unfolding. It's easy to get lost in the details, but at its heart, the Electrophilic Addition Mechanism is a fantastic tale of atoms finding their perfect partners. It's chemistry at its most engaging, and it's definitely worth keeping an eye on! You might just find yourself hooked.
