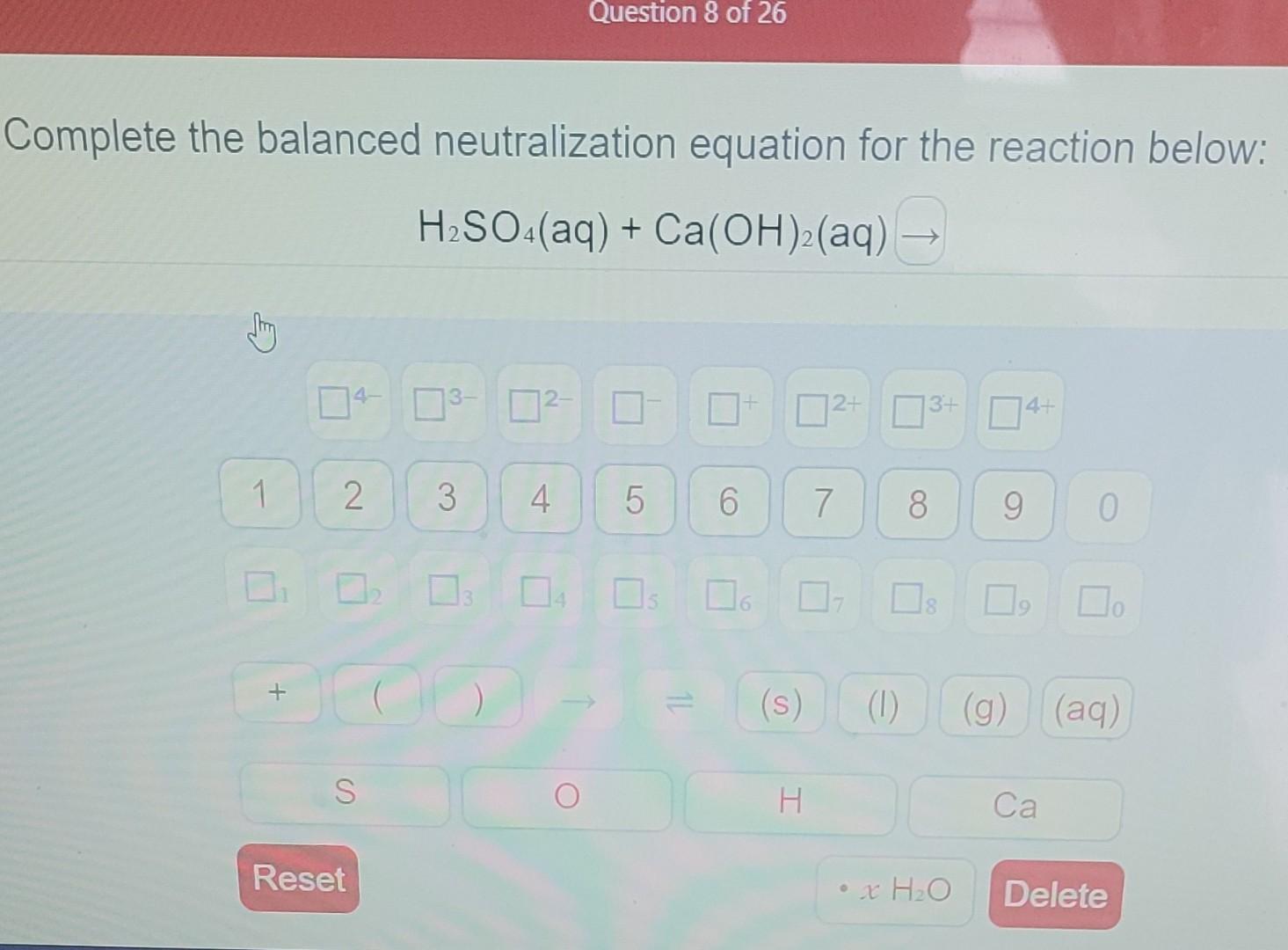
Complete The Balanced Neutralization Equation For The Reaction Below

Ever wondered what happens when you mix some of the most common household substances and get a perfectly balanced result? It's a bit like a culinary experiment, but instead of creating a delicious dish, you're aiming for a state of perfect equilibrium. This might sound like something straight out of a high-school chemistry class, but understanding how to complete a balanced neutralization equation is actually a surprisingly fun and incredibly useful skill. Think of it as unlocking a secret code to predict how different chemicals will interact, ensuring safety and efficiency in countless everyday situations and even in more complex scientific endeavors. It’s about bringing order to chaos, transforming potentially reactive substances into something stable and harmless.
So, what exactly are we talking about? At its heart, a neutralization reaction is the chemical meeting between an acid and a base. Acids, as you might recall, are often sour (like lemon juice) and can be quite corrosive. Bases, on the other hand, are often bitter and can feel slippery (like soap). When these two opposite forces collide, they don't just cancel each other out in a dramatic explosion. Instead, they engage in a very specific dance, swapping partners to form something new: usually water and a salt. This process is called neutralization because it effectively reduces the acidity and basicity of the mixture, bringing it closer to a neutral pH, like that of pure water.
The beauty of completing a balanced neutralization equation lies in its predictive power. Imagine you’re cleaning up a spill with an acidic cleaner and you need to neutralize it. Knowing the products of the reaction allows you to choose the right neutralizing agent, ensuring you don't create a worse mess or a hazardous situation. In industries, from food production to pharmaceuticals and manufacturing, precise control over chemical reactions is paramount. Balanced equations are the blueprints that allow scientists and engineers to achieve these precise outcomes, minimizing waste and maximizing safety. It's not just about avoiding unpleasant surprises; it's about enabling progress and innovation.
Let's dive into a specific example that might appear on your chemistry worksheet or in a lab manual: the reaction between hydrochloric acid and sodium hydroxide.
HCl + NaOH → ?
Solved Complete the balanced neutralization equation for the | Chegg.com
This might look like a cryptic puzzle at first glance. We have HCl, which is our familiar hydrochloric acid, and NaOH, our sodium hydroxide, a common base. Our mission, should we choose to accept it, is to figure out what these two reactants will produce when they neutralize each other, and importantly, to make sure the equation is balanced. This means ensuring that the number of atoms of each element on the reactant side (what you start with) is equal to the number of atoms of that same element on the product side (what you end up with). It’s like a cosmic accounting system for atoms!
In a neutralization reaction between an acid and a base, the general outcome we expect is water and a salt. So, we know that water (H₂O) will be one of the products. The other product will be a salt, formed from the cation of the base and the anion of the acid. In this case, the cation from NaOH is Na⁺ (sodium ion), and the anion from HCl is Cl⁻ (chloride ion). When these ions come together, they form sodium chloride (NaCl), which you might know as table salt! So, our reaction is starting to take shape:
HCl + NaOH → H₂O + NaCl
Now, the crucial step: balancing. Let's count the atoms on each side:
- Left side (Reactants):
- Hydrogen (H): 1 (from HCl) + 1 (from NaOH) = 2
- Chlorine (Cl): 1 (from HCl)
- Sodium (Na): 1 (from NaOH)
- Oxygen (O): 1 (from NaOH)
- Right side (Products):
- Hydrogen (H): 2 (from H₂O)
- Chlorine (Cl): 1 (from NaCl)
- Sodium (Na): 1 (from NaCl)
- Oxygen (O): 1 (from H₂O)
Take a look! On both the left and right sides, we have exactly 2 hydrogen atoms, 1 chlorine atom, 1 sodium atom, and 1 oxygen atom. This means our equation is already perfectly balanced!
The completed balanced neutralization equation is:

HCl(aq) + NaOH(aq) → H₂O(l) + NaCl(aq)
The little letters in parentheses (aq for aqueous, meaning dissolved in water, and l for liquid) are called state symbols and are often included in chemical equations to provide extra information about the physical state of the substances involved. While not always strictly necessary for basic balancing, they are good practice!
Why is this simple balancing act so important? Because even a slight imbalance can lead to incorrect calculations in real-world applications. If you were trying to determine the exact amount of an antacid tablet needed to neutralize stomach acid, or the precise concentration of a cleaning solution, an unbalanced equation would give you the wrong answer. It's the difference between getting it just right and potentially causing more problems than you solve. So, the next time you encounter a neutralization equation, remember that you're not just filling in blanks; you're mastering a fundamental principle of chemistry that keeps our world running smoothly, safely, and predictably. It's a small skill with a surprisingly big impact!

