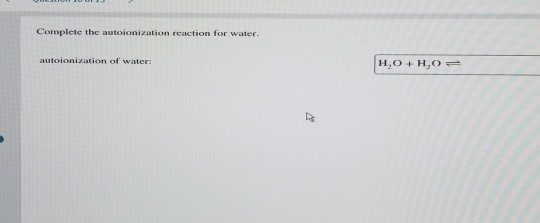
Complete The Autoionization Reaction For Water.
Hey there! So, you wanna chat about water, huh? Yeah, I know, sounds a little boring at first. Like, what’s there to say about something we basically see every single day? But guess what? Water’s got some secrets. It’s a little bit of a drama queen, and today we’re gonna dive into its most dramatic performance yet: autoionization. Don’t let the fancy word scare you; it’s actually super cool. Think of it like water doing a little solo act, all by itself. Wild, right?
Basically, what we’re talking about here is water being… well, a little bit extra. It can’t just sit there being plain old H₂O forever. Nope. It needs some excitement. So, sometimes, and I mean, like, really sometimes – not all the time, don’t worry – a water molecule decides to do something… unusual. It’s like it looks at its neighbor and says, "You know what? I’m gonna give you one of my hydrogens. Just for funsies." And poof! Magic happens.
So, let’s paint a picture, shall we? Imagine a bunch of water molecules, just chilling. They’re all holding hands, metaphorically speaking. Then, one of them, let’s call her H₂O-A, gets a little antsy. She leans over to her buddy, H₂O-B, and with a little nudge and a wink, she donates one of her hydrogen atoms. Just like that. Gone. Poof.
Now, what does that leave H₂O-A with? Well, she had two hydrogens, right? She gave one away. So now she’s just… OH⁻. Yep, that’s a hydroxide ion. It’s got a negative charge because the hydrogen she gave away took its electron with it. Sneaky! And that hydrogen, all by its lonesome, is now a positively charged little guy. We call that a proton, or more accurately, since it’s in water, an hydronium ion. Think of it as H₃O⁺. It’s like H₂O grabbed that stray proton and said, "Whoa, you look lost! Come here, let me help you out."
So, the whole reaction looks something like this, my friend: H₂O + H₂O → H₃O⁺ + OH⁻. See? Two water molecules doing their thing, and then suddenly you’ve got these two new, charged entities. It’s like a molecular breakup and a new couple forming, all in one go. Water splitting itself up! Can you believe it?
Now, here’s the kicker, the really mind-blowing part that makes you go, "Wait, that’s why my science teacher was so excited about water?" This happens all the time. Not like, a billion times a second in every single molecule, but in a huge amount of water, it’s happening constantly. Even in the purest water you can imagine, there are always these little H₃O⁺ and OH⁻ ions floating around. It’s like the water is perpetually creating and destroying tiny couples, a never-ending molecular dance party.
This is why water is such a fantastic solvent, by the way. Those charged ions? They’re like little magnets. They can attract other charged things, or even things that are a little bit polar, and pull them into the water. It’s like water is saying, "Come on in, the water’s fine! And hey, if you’re charged, I can totally hang out with you." Without this autoionization, water would be a much less interesting solvent. Imagine trying to dissolve salt in something that’s just plain, neutral H₂O all the time. It wouldn’t be nearly as effective. We’d be in a sticky situation, literally.
And this, my friend, is the fundamental reason why we talk about pH. You’ve heard of pH, right? Acidic, basic, neutral? It’s all about the balance of these little H₃O⁺ and OH⁻ ions. In pure water, the number of H₃O⁺ ions is exactly equal to the number of OH⁻ ions. It’s a perfect, neutral balance. Like a perfectly calibrated scale. And that, my friends, is why pure water is considered neutral. Not too acidic, not too basic. Just… watery.

But what happens when you add something to water? Ah, now that’s where the real fun begins! If you add an acid, like, say, a squeeze of lemon juice (which is citric acid, in case you were wondering), you’re basically chucking extra H₃O⁺ ions into the mix. It’s like you’re throwing a party and inviting a whole bunch of extra positive guests. What does that do to the balance? Well, it throws it off, doesn’t it? You suddenly have way more H₃O⁺ than OH⁻. And that, my friend, makes the solution acidic. More H₃O⁺ = acidic. Simple as that. It’s like the water is saying, "Whoa, so many positive vibes in here!"
On the other hand, if you add a base, like, oh, I don’t know, a bit of baking soda (sodium bicarbonate, for the geeks among us), you’re doing the opposite. You’re adding something that either has OH⁻ ions already, or it reacts with the H₃O⁺ ions to make more OH⁻. It’s like you’re inviting a bunch of negative ions to the party, or you’re bribing the positive ones to leave. Suddenly, you’ve got a whole lot more OH⁻ ions chilling around than H₃O⁺. And that, my dear friend, makes the solution basic, or alkaline, if you wanna use the fancier term. More OH⁻ = basic. See the pattern? It’s all about the H₃O⁺ versus OH⁻ showdown.
The amount of H₃O⁺ and OH⁻ ions in pure water is actually tiny. Like, mind-blowingly small. At room temperature (let’s say 25°C, because that’s the standard we usually use for these things), the concentration of H₃O⁺ is about 1.0 x 10⁻⁷ moles per liter. And guess what? The concentration of OH⁻ is exactly the same. That little number, 10⁻⁷, is what makes pure water neutral. It’s a delicate equilibrium, a whisper of ions in a sea of molecules. It's honestly amazing that so much of our world relies on such a tiny, almost imperceptible, process.

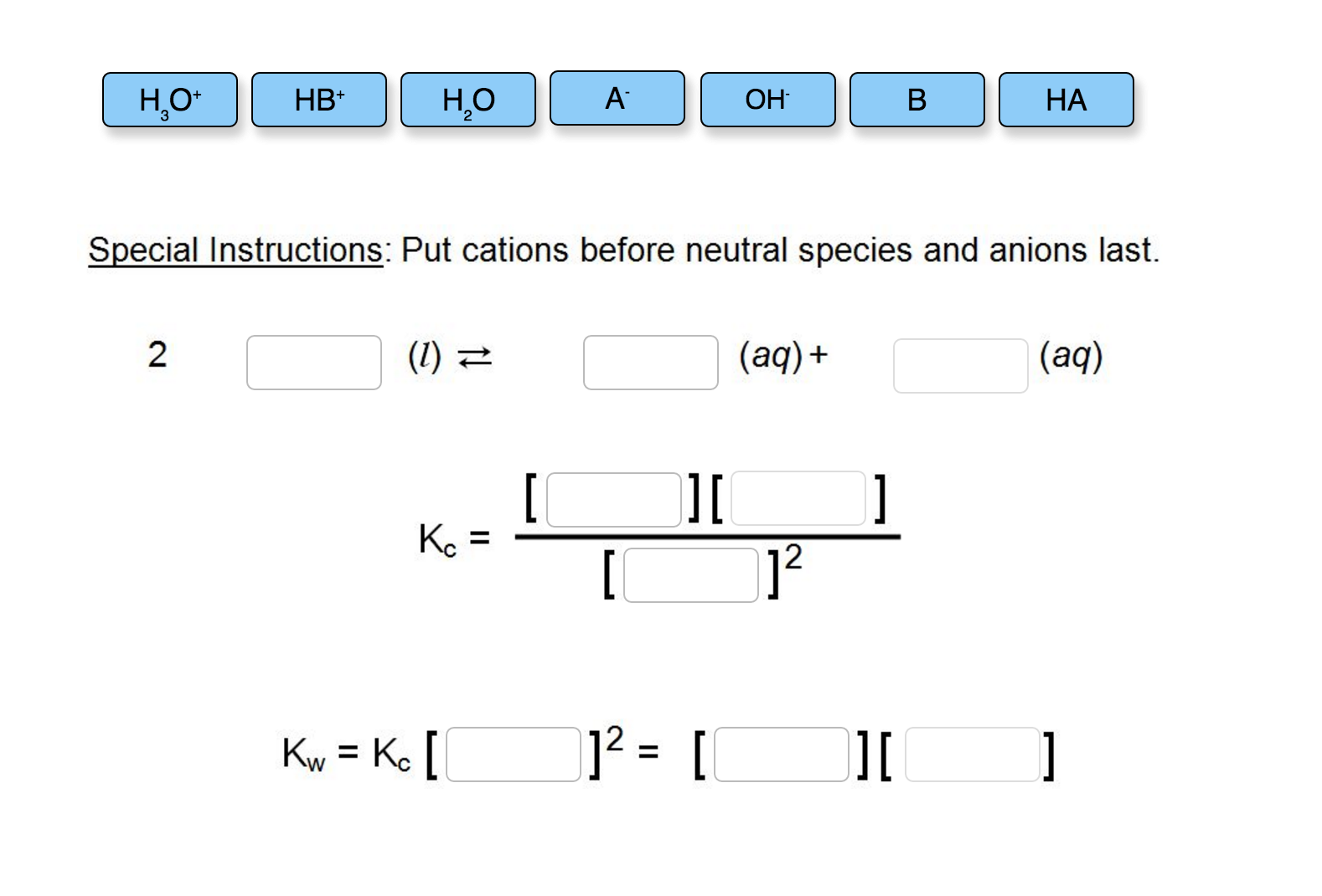
So, when we talk about completing the autoionization reaction for water, what we’re really doing is acknowledging this fundamental property. We write it as: 2H₂O(l) ⇌ H₃O⁺(aq) + OH⁻(aq). Let’s break that down, just so we’re all on the same page, because I know science-y symbols can look a little intimidating. The ‘2H₂O(l)’ means we’ve got two molecules of liquid water. The ‘⇌’ symbol is super important – it’s a double-headed arrow, which means the reaction goes both ways. It’s happening forward (water splitting) and backward (H₃O⁺ and OH⁻ coming back together to form water) simultaneously. It's a dynamic balance, always shifting, always moving. It’s never just one thing happening in isolation; it’s a constant molecular tango.
Then we have ‘H₃O⁺(aq)’ which is our hydronium ion, and ‘OH⁻(aq)’ which is our hydroxide ion. The ‘(aq)’ means they are dissolved in water, which makes sense, because they were born in water, right? They can’t really exist anywhere else without causing a whole heap of trouble, or just disappearing. They are water’s children, essentially, and they need their watery home.
This little equation, this seemingly simple representation of water splitting, is actually the key to understanding so much of chemistry and biology. It explains why our blood has a specific pH, why rain can be acidic, and why cleaning products work the way they do. It’s like the secret handshake of the molecular world, and once you know it, a whole new universe opens up. It’s not just about H₂O anymore; it’s about the dynamic, reactive, and utterly essential nature of water itself.

Think about it: if water didn't autoionize, if it just stayed as H₂O forever and ever, our bodies wouldn't be able to do a million things they do. Enzymes wouldn't function properly. Chemical reactions wouldn't happen at the right speeds. Life as we know it would be… well, it just wouldn't exist. So, next time you take a sip of water, or wash your hands, or even just feel the rain on your face, give a little nod to the H₂O molecules having their little autoionization party. They’re doing a lot more than you think!
And this autoionization is also why pure water has a specific conductivity. It's not a perfect conductor like a metal, but those ions, even in small amounts, allow a tiny bit of electricity to flow. It’s like the water is giving you a little jolt, a subtle reminder of its inner life. Add more ions, and conductivity goes way up. That's why salty water conducts electricity way better than fresh water, but that’s a story for another coffee chat. The point is, this basic act of water splitting is the foundation for a whole lot of measurable properties.
It’s also a reminder that nothing is ever truly static. Even something as seemingly simple and unchanging as water is in constant motion and transformation at the molecular level. It’s a beautiful dance of creation and destruction, a testament to the vibrant, ever-changing nature of the universe. So, yeah, water autoionization. It might sound like a mouthful, but it’s actually one of the coolest things going on in that glass of water you’re probably holding right now. Pretty neat, huh?
