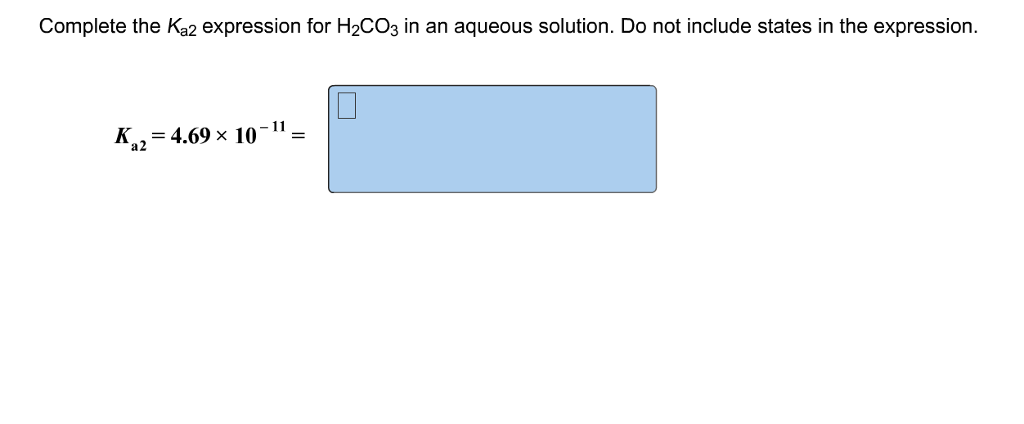
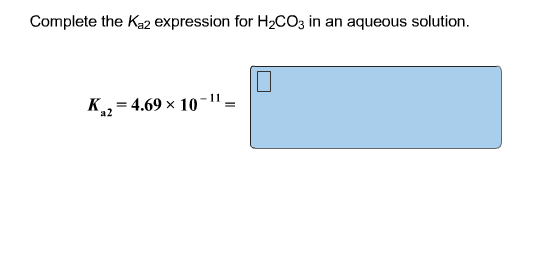
Complete The 𝐾a2 Expression For H2co3 In An Aqueous Solution.
Hey there, science explorers! Ever wondered what happens when you mix something a little fizzy with water? You know, like that delightful sparkle in your favorite soda, or the way a pinch of baking soda makes your lemonade dance? Well, today we’re diving headfirst into the bubbly, zesty world of carbonic acid – or as the cool kids call it, H₂CO₃! Prepare for a microscopic party because we're about to complete a super important chemistry expression that's as crucial as finding the perfect meme to sum up your day.
Think of H₂CO₃ as a slightly shy, two-part molecule hanging out in water. It’s like a polite guest at a very large party (the water molecules, obviously!). This little molecule has a couple of protons, like tiny, energetic guests, that it’s willing to share. We've already talked about the first time it decides to share a proton – that’s like it letting go of its first handshake. It’s a big deal, but there’s still more excitement to come!
Now, we’re focusing on the second act of generosity from our friend, H₂CO₃. This is when our slightly-less-shy carbonic acid, which has already shed one proton and is now called HCO₃⁻ (think of it as the "hydrogen carbonate ion" or "bicarbonate ion" – basically, it’s still got a proton-holding superpower, but it’s already used its first trick), decides it’s ready to share its second proton. This is where the magic really happens, and we need to write down the recipe for this incredible event!
Imagine you're at a potluck. The first dish (the first proton release) was a hit. Now, the host (our bicarbonate ion) has another delicious item to offer (the second proton). We need to capture the exact ingredients and the outcome of this second sharing spree. It’s all about balance, the dance between what’s there before and what’s there after.
It's like a chemistry soap opera, but way more predictable and, dare I say, even more satisfying to solve!
So, let's paint the picture for this second act. We start with our partially-de-protonated carbonic acid, the HCO₃⁻. It’s floating around in all that water, feeling generous. Then, poof! It decides to release its second proton. What’s left behind? Well, if it gives away another proton, it’s going to have one less positive charge. And what’s that little proton anyway? It's essentially a H⁺ ion, a tiny ball of positive energy. The molecule that remains after losing that second proton is the CO₃²⁻, which we call the carbonate ion. Think of it as the remaining, very stable part of the original carbonic acid, now with a double negative charge because it’s shed two protons in total.

This whole transformation, this second act of proton-giving, is what chemists love to express with a special equation. It’s our way of saying, "Here’s precisely what’s going on, down to the tiniest particle!" We represent this with an equilibrium constant. For the second ionization of carbonic acid, this is famously known as Kₐ₂. It’s like a judge’s score for how easily that second proton is released. A higher Kₐ₂ means it’s more eager to share, a lower one means it’s a bit more clingy.
So, how do we write this epic expression? It’s like writing a very important recipe where the order and ingredients are absolutely critical. We need to show what we start with and what we end up with, and crucially, how they relate to each other. The expression for Kₐ₂ is basically a fancy way of saying:
Kₐ₂ = ([Products]) / ([Reactants])
And in our case, the "products" are the things that are formed when HCO₃⁻ releases its proton. Those are our H⁺ ions (the free protons zipping around) and the CO₃²⁻ ions (the carbonate queens). The "reactant" is what we started with for this second act, which is our HCO₃⁻ ion.

So, drumroll, please! The complete Kₐ₂ expression for carbonic acid in an aqueous solution is:
Kₐ₂ = ([H⁺][CO₃²⁻]) / [HCO₃⁻]
There it is! Isn’t that just… chef’s kiss? This little equation tells us everything about how readily carbonic acid gives up its second proton in water. It’s like the chemical equivalent of saying, "Yep, this is how the fizzy water keeps its fizz going, just a little bit at a time!" Every time you hear about the acidity of things like carbonated drinks, or the chemistry happening in oceans and our own bodies, this little expression is probably playing a starring role, even if you can’t see it. It’s the unsung hero of many everyday phenomena. Pretty neat, right?
