Complete The 𝐾a1 Expression For H2co3 In An Aqueous Solution.

Ever felt like some science concepts are hidden treasures, just waiting to be discovered? Well, today we're diving into one of those sparkling gems. It's all about something called carbonic acid, or H2CO3, and how it behaves when you mix it with water.
Think of it like a little chemical party. Carbonic acid, when it hits the water, doesn't just sit there. It starts to, shall we say, "unzip" itself a bit. It's a fascinating process to watch, in a nerdy, chemistry-loving kind of way!
And the star of our show today is the Ka1 expression. Don't let the fancy name scare you! It's basically a way to describe how well carbonic acid splits up in that water party. It tells us a story about its strength.
So, what exactly is this H2CO3? Imagine it as a molecule with a couple of hydrogen atoms ready for an adventure. When it meets water, it's like they get a little excited and decide to break away. It's a reversible reaction, meaning they can also decide to come back together!
This whole "splitting up" business in water is what chemists call dissociation. It’s like a molecule taking a deep breath and letting go of a part of itself. For carbonic acid, it's a two-step process, and we're focusing on the first step today.
The Ka1 expression is the special formula that captures this first step perfectly. It's like a recipe for understanding how much of that carbonic acid decides to go solo. This little expression is key to unlocking secrets about the solution.
Why is this so entertaining, you ask? Because it’s all about balance and change. In chemistry, things are rarely static. They're always in motion, reacting, and adjusting. It’s a constant dance.
And carbonic acid is a fantastic dancer! It’s involved in so many things around us, from the fizz in your soda to the very oceans that cover our planet. Understanding its behavior is like getting a backstage pass to some amazing natural phenomena.
Let's talk about what makes the Ka1 expression so special. It's not just a string of symbols. It's a powerful representation of a fundamental chemical principle. It’s elegant in its simplicity, yet profound in its implications.
Think of it as a detective’s clue. The Ka1 value tells us how readily carbonic acid will release its first hydrogen ion. A higher Ka1 means it’s a bit more eager to let go, while a lower Ka1 means it’s more attached.
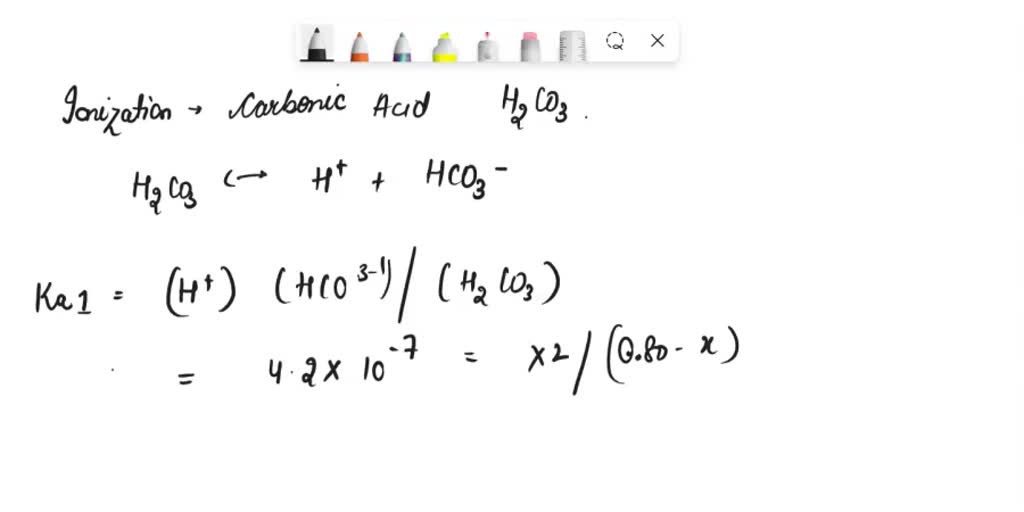
In an aqueous solution, meaning water is the solvent, this dissociation happens. Water molecules are busy little things, and they help facilitate this split. They surround the ions and keep them separate.
The Ka1 expression involves concentrations. Yes, those brackets you might see in chemistry? They mean concentration. We look at how much of the split-up pieces are present compared to what's left of the original carbonic acid.
It’s a ratio, really. A comparison of the "products" of the reaction to the "reactants." This ratio is incredibly informative. It's the core of what the Ka1 expression is all about.
So, imagine our carbonic acid molecule, H2CO3. When it hits the water, one of those hydrogen atoms might decide to take a dip on its own. It becomes a hydrogen ion, often written as H+.
What's left of the carbonic acid? It's now a bicarbonate ion, which is HCO3-. It’s like the remaining piece after a small section has detached.
The Ka1 expression is our way of quantifying this equilibrium. It’s not just a one-way street; the bicarbonate ion can grab a hydrogen ion back if it wants to. This is the fascinating world of chemical equilibrium.
The expression looks something like this: Ka1 = ([H+][HCO3-]) / [H2CO3]. See? It’s the concentrations of the hydrogen ion and the bicarbonate ion multiplied together, divided by the concentration of the original carbonic acid.
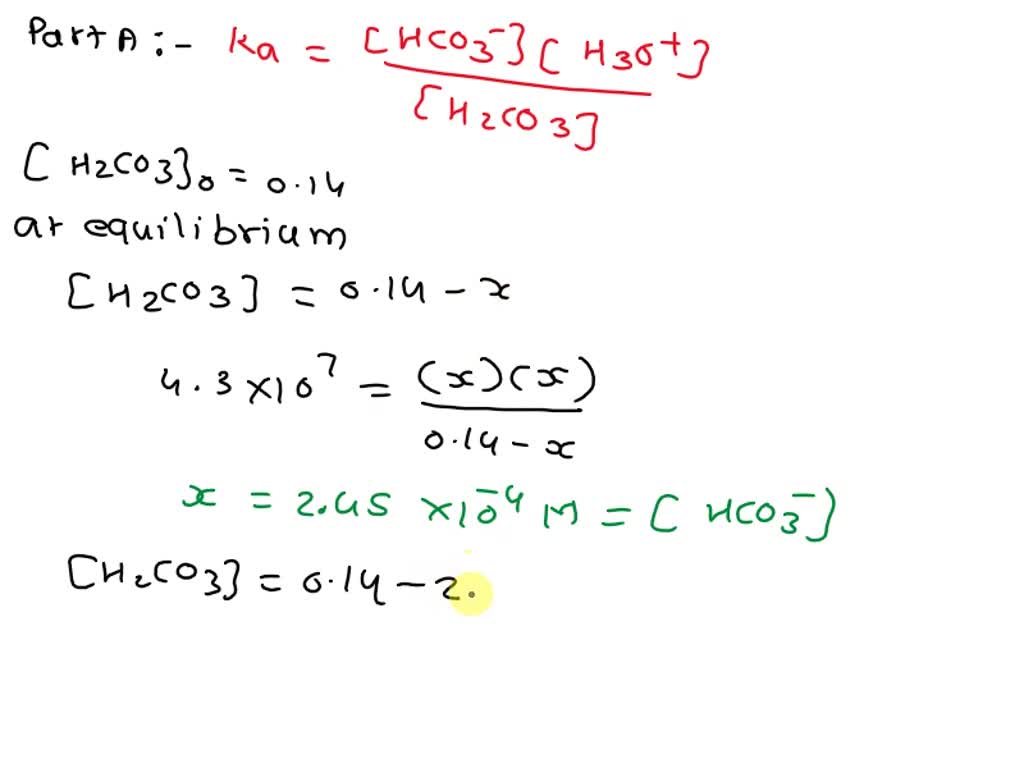
It’s a beautiful equation that encapsulates a dynamic process. It’s a snapshot of the balance at a given moment. It’s a story told in symbols, but with immense meaning.
This concept is so much more than just a formula. It’s the foundation for understanding acidity in solutions. It helps us predict how a substance will behave and what kind of environment it will create.
Carbonic acid is special because it’s a weak acid. This means it doesn’t dissociate completely. It’s not a dramatic, all-or-nothing event. It's a gentle unfolding.
The Ka1 expression perfectly reflects this weak acid nature. It acknowledges that there will still be a significant amount of undissociated carbonic acid left. It's not just the split pieces; the original molecule is still very much present.
Why is this so captivating? Because it’s relatable! Think about how sometimes you might be hesitant to do something, while other times you're all in. Carbonic acid has its own personality, and the Ka1 expression helps us understand it.
It's also entertaining because it’s connected to the real world. Carbonic acid is formed when carbon dioxide dissolves in water. This happens everywhere, from your breath exhaled into the air to the vast oceans.
The oceans, for instance, use carbonic acid to regulate their pH. This is crucial for marine life. The Ka1 expression plays a silent, yet vital, role in keeping the ocean’s delicate balance.

So, when you see that H2CO3 and the Ka1 expression, don't just think of abstract chemistry. Think of the fizz, the life in the sea, the very breath you take. It's all interconnected by these fundamental chemical principles.
What makes it particularly special is how it highlights the power of equilibrium. Things aren't always just one way or the other. There's a constant interplay, a back-and-forth, that creates stability and allows for life as we know it to thrive.
The Ka1 expression is our window into that equilibrium for carbonic acid's first dissociation. It's a testament to how much we can learn by observing and quantifying the tiny, invisible world of molecules.
It's like peering into a microscopic dance floor, where molecules waltz and sway. The Ka1 expression is the choreography that describes a specific move in that dance. And it's a move that has enormous consequences.
The simplicity of the equation, with just a few key components, is deceptive. It hides a universe of chemical behavior and environmental relevance. That's what makes it so compelling.
So, next time you encounter the Ka1 expression for H2CO3 in an aqueous solution, I hope you’ll see it not as a dry scientific fact, but as a story. A story of molecular interactions, of chemical balance, and of the hidden wonders of our world.
It’s an invitation to explore further, to wonder about the other steps in carbonic acid’s journey, and to appreciate the elegance of chemistry. It’s a tiny piece of a vast and fascinating puzzle.
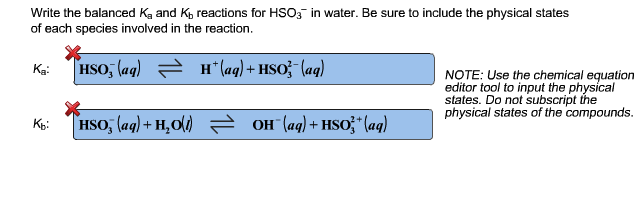
It truly is a delightful subject, a little peek into how the world works at its most fundamental level. And that, my friends, is a wonderfully entertaining and special thing!
This expression, Ka1 = ([H+][HCO3-]) / [H2CO3], is your ticket to understanding the initial steps of carbonic acid's chemical adventure in water.
The journey of carbonic acid in water is a captivating one. It’s a dance of dissociation and recombination. And the Ka1 expression is our guide through the first, crucial step of this dance.
It’s a fundamental concept in chemistry, but it’s also a gateway to understanding many natural processes. From the fizz in your drink to the health of our planet’s water bodies, carbonic acid plays a role.
So, don’t shy away from these scientific terms. They are keys that unlock deeper understanding and appreciation for the world around us. The Ka1 expression is just one such key.
It’s a reminder that even the seemingly simple act of dissolving something in water can involve complex and beautiful chemical choreography. And we get to peek behind the curtain and learn about it!
The enduring appeal of such expressions lies in their ability to simplify complexity. They allow us to grasp intricate relationships with a clear, concise notation. The Ka1 expression is a perfect example.
It’s a story of transformation, of how one substance can change and interact with its surroundings. And it’s a story that unfolds with every bubble, every pH change, and every drop of rain.
So, embrace the curiosity! The world of chemistry is full of these exciting discoveries. The Ka1 expression for H2CO3 is a fantastic starting point for your own exploration.
