Complete And Balance The Following Neutralization Reactions

Hey there, fabulous humans! Ever feel like life’s throwing a lot of… stuff at you? From that slightly too-tangy lemonade at brunch to the inevitable work deadlines that make your brain feel like an overcooked noodle, we’re all about finding that sweet spot of balance. And guess what? The universe, in its infinite wisdom, has a pretty cool way of doing that too, especially when it comes to chemistry. We’re talking about neutralization reactions – the ultimate pH balancing act of the molecular world. Think of it as nature’s way of saying, “Okay, deep breaths, let’s find equilibrium.”
You’ve probably heard of acids and bases, right? They’re those classic rivals in the chemistry classroom. Acids, with their sharp, sour tastes (think lemons, vinegar), are all about giving away hydrogen ions. Bases, on the other hand, are all about accepting those ions, often feeling a bit slippery (think soap). When these two meet in just the right way, magic happens. They neutralize each other, creating something much more chill: water and a salt. It’s like a rockstar and a librarian finding common ground at a coffee shop – a surprisingly harmonious outcome!
The Everyday Alchemy of Balancing Act
So, why should you care about this whole acid-base shindig? Because it’s happening all around you, all the time! It’s the science behind why baking soda is your best friend for soothing insect bites (your skin’s slightly acidic after being poked by a mosquito) or why antacids work their magic on that fiery heartburn. It’s all about restoring that happy medium, that comfortable pH level.
Let’s dive into some of the classic dance partners in this neutralization ballet. Our first star player is often a strong acid, like hydrochloric acid (HCl). You’ve likely encountered HCl in your stomach, diligently breaking down your lunch. But it’s also used in industries, from metal cleaning to making plastics. When HCl gets a bit too wild, it needs a counterpoint.
Enter sodium hydroxide (NaOH), a strong base. Think of it as the calm, steady presence. NaOH is also a workhorse in industries, used in everything from soap making to paper production. When HCl and NaOH decide to tango, the result is a beautiful symphony of water (H₂O) and sodium chloride (NaCl) – yep, that’s regular old table salt! How neat is that? Your stomach acid, when neutralized, essentially becomes salt and water. Pretty wild to think your internal digestive juices are just a slightly more intense version of your kitchen spice rack, eh?
The Reaction Revealed: HCl + NaOH → NaCl + H₂O
This equation is like the opening credits to our neutralization movie. It shows us the key ingredients and the equally key outcomes. Hydrochloric acid (HCl) plus sodium hydroxide (NaOH) gives us sodium chloride (NaCl) and water (H₂O). It’s a textbook example, and a really elegant one at that. The hydrogen from the acid teams up with the hydroxide from the base to form water, and the remaining ions – the sodium from the base and the chloride from the acid – pair up to form the salt. Boom! Neutral. Zero drama.
Imagine this: you’re making a super-sour pickle brine, and it’s just… too much. You want to dial it back a notch. A tiny, tiny bit of baking soda (sodium bicarbonate, NaHCO₃) can help. Baking soda is a base, and it’ll react with the acetic acid (the sour stuff) in your brine, making it more palatable. It’s like adding a sprinkle of sugar to your espresso – a subtle adjustment for a more enjoyable experience.
![[ANSWERED] Complete and balance the following neutralization reaction](https://media.kunduz.com/media/sug-question/raw/74327311-1659787475.2104254.jpeg?h=512)
When Acids Go Green (and Bases Do Too!)
Not all acids and bases are created equal. We’ve got the heavy hitters, the strong ones like HCl and NaOH, which fully dissociate in water. Then we have the milder characters, the weak acids and weak bases. These are the ones that often show up in our daily lives in gentler forms.
Take acetic acid (CH₃COOH), the star of vinegar. It’s a weak acid, meaning it doesn’t release all its hydrogen ions at once. This is why vinegar is great for cleaning and pickling, but you won’t find it being used to etch metal like its stronger cousins. Its reaction partner? Often a weak base like sodium bicarbonate (NaHCO₃), also known as baking soda. This is the dynamic duo of household hacks!
When acetic acid from vinegar meets baking soda, what do we get? It’s a bit more complex than the HCl and NaOH reaction, but the essence is the same: neutralization. The reaction produces sodium acetate (CH₃COONa), water (H₂O), and a fun little extra – carbon dioxide gas (CO₂). That’s the fizzy, bubbly action you see when you mix them! It’s like a tiny, contained science fair experiment happening right on your kitchen counter.
The Bubbly Delight: CH₃COOH + NaHCO₃ → CH₃COONa + H₂O + CO₂
This equation is a bit of a showstopper, isn't it? It’s the reason why we love doing the baking soda volcano experiment with kids. The acetic acid from the vinegar, the sodium bicarbonate from the baking soda, they get together and create sodium acetate (which is basically a salt), water, and the glorious carbon dioxide bubbles. This reaction is a fantastic visual reminder that even seemingly simple mixtures can have exciting results. Plus, who doesn't love a good fizz?

This reaction is super useful. Feeling a bit of indigestion? A spoonful of baking soda dissolved in water can help neutralize excess stomach acid (which is primarily HCl). While not as sophisticated as over-the-counter antacids, the principle is the same: a base reacting with an acid to bring relief. Just remember, moderation is key – we don’t want to over-alkalize things either!
More Than Just Molecules: Cultural Connections
The concept of balance and harmony isn’t just a chemistry lesson; it’s deeply ingrained in cultures around the world. Think about the Yin and Yang in Taoism, representing the interconnected and interdependent nature of opposites. Or the Ayurvedic principle of balancing the three doshas (Vata, Pitta, Kapha) for optimal health. These are all about finding that perfect equilibrium, much like a neutralization reaction finds its sweet spot.
Even in culinary arts, the balance of flavors is paramount. The acidity of a lemon or lime can cut through the richness of a fatty dish, creating a more dynamic and enjoyable taste experience. Too much acidity, and it’s puckeringly unpleasant. Too little, and the dish might feel heavy or one-dimensional. It’s a constant dance of complementing and contrasting elements.
And let's not forget about nature itself. The pH of soil is crucial for plant growth. If it's too acidic or too alkaline, plants struggle. Farmers often use lime (calcium oxide, a base) to raise the pH of acidic soils or sulfur to lower the pH of alkaline soils, essentially performing a large-scale neutralization to create the ideal growing conditions.

A Splash of Green: Using Bases to Tame Acidity
Let’s consider another scenario. You’re making a vibrant green smoothie, and you’ve added a bit too much spinach, making it taste a tad… earthy or even slightly bitter. Spinach, while packed with nutrients, can have a slightly alkaline nature. To counter that subtle bitterness, you might add a tiny squeeze of lemon juice – a touch of acid. This is a micro-neutralization happening in your blender! It’s about tweaking those flavors to achieve that perfect sip.
On a larger scale, think about acid rain. When pollutants like sulfur dioxide and nitrogen oxides are released into the atmosphere, they react with water to form sulfuric and nitric acids, which then fall as acid rain. This can damage ecosystems, buildings, and monuments. To combat this, sometimes limestone (calcium carbonate, a base) is used to neutralize the acidity in affected lakes and soils. It’s a way of stepping in to correct an imbalance, much like our stomach uses its own built-in systems.
When Things Get a Little… Salty
The ‘salt’ that’s produced in neutralization reactions isn't always the table salt we’re used to. There are thousands of different salts, formed from the combination of different acids and bases. For example, when sulfuric acid (H₂SO₄), a strong acid, reacts with potassium hydroxide (KOH), a strong base, you get potassium sulfate (K₂SO₄) and water.
The Potassium Tango: H₂SO₄ + 2KOH → K₂SO₄ + 2H₂O
Notice the ‘2’s in this equation? That’s stoichiometry at play, ensuring everything is balanced perfectly. For every molecule of sulfuric acid, you need two molecules of potassium hydroxide to completely neutralize it. This gives you potassium sulfate, which is actually used as a fertilizer! So, a strong acid and a strong base can combine to create something beneficial for plant life. It's a reminder that even powerful forces can yield constructive results when they work together harmoniously.
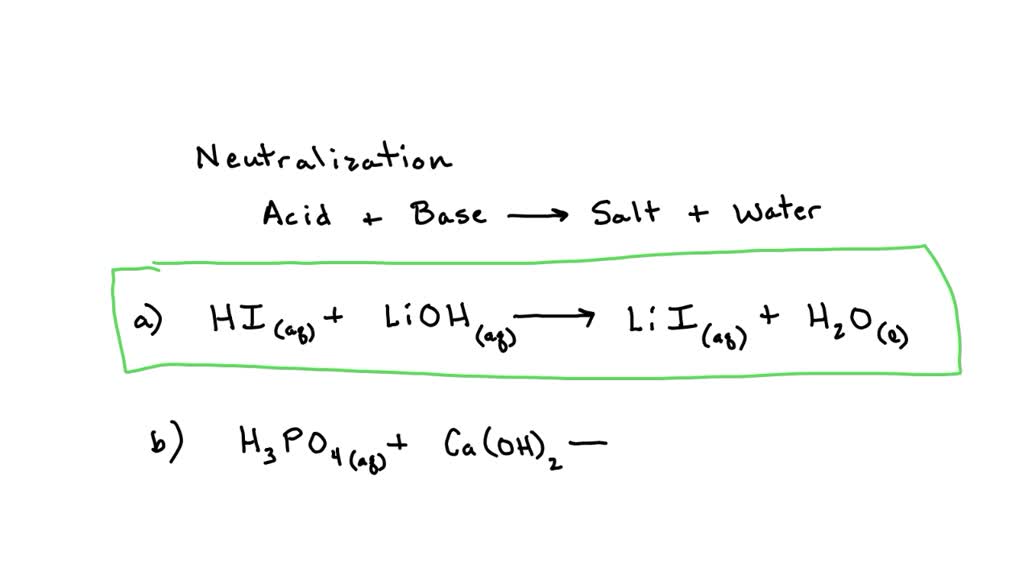
Another example: nitric acid (HNO₃) reacting with calcium hydroxide (Ca(OH)₂). This yields calcium nitrate (Ca(NO₃)₂) and water. Calcium nitrate is also used as a fertilizer, particularly in hydroponic systems. So, the next time you see lush green plants thriving, remember that some of their essential nutrients might have been born from a neutralization reaction!
Practical Tips for Your Own Little pH Balance
You don’t need a lab coat to appreciate or utilize neutralization. Here are a few everyday takeaways:
- Stomach Soothers: For occasional heartburn, over-the-counter antacids are your go-to. They contain mild bases like magnesium hydroxide or aluminum hydroxide that neutralize excess stomach acid.
- Gardening Grace: Test your soil pH. If it’s too acidic, add lime. If it’s too alkaline, add sulfur. This is literally a neutralization reaction happening in your backyard!
- Kitchen Chemistry: Baking soda is your mild base superhero. Use it to calm acidic marinades, degrease pans, or even to make your coffee taste less bitter.
- Vinegar Versatility: That trusty bottle of vinegar? It’s your mild acid ally for cleaning and descaling. Just remember to use it judiciously – you don’t want your home to smell like a salad dressing factory!
- Insect Bites: A paste of baking soda and water can help neutralize the acidity from insect stings and bites, providing relief.
It’s amazing how understanding a little bit of chemistry can empower you to handle everyday situations more effectively and even creatively. It’s about knowing the properties of the substances around you and how they interact.
A Final Thought on Finding Your Balance
We spend so much of our lives trying to find that perfect equilibrium, don’t we? Whether it’s balancing work and play, our ambitions and our well-being, or even just the flavors in our dinner plate, the quest for balance is universal. Neutralization reactions in chemistry are a beautiful, tangible illustration of this principle. They show us that even opposing forces, when brought together in the right proportions, can create something stable, useful, and harmonious.
So, the next time you feel a bit overwhelmed, a little too ‘acidic’ from stress or a touch too ‘alkaline’ from too much of a good thing, take a moment. Think about the elegant dance of acids and bases. Find your own little moments of neutralization. A deep breath, a walk in nature, a chat with a friend – these are all ways we bring balance back into our lives. And just like in chemistry, sometimes the most profound changes come from the simplest, most balanced interactions.
