Complete And Balance The Following Half-reaction In Acidic Solution Mno4-
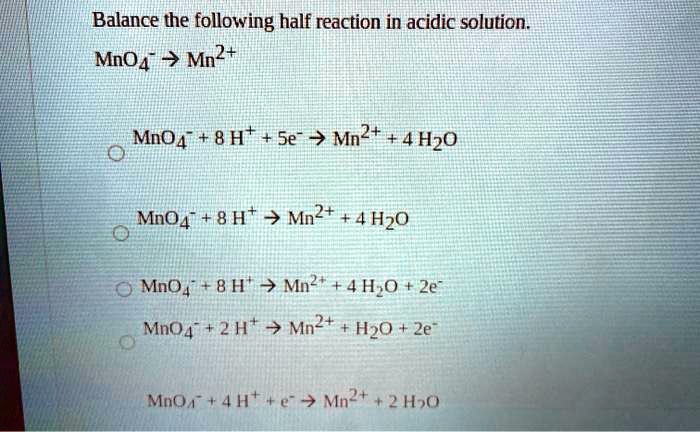
Ever feel like you're trying to put together a puzzle with missing pieces? That's exactly what we're doing today, but instead of a grumpy cat or a majestic mountain, we're tackling a chemical reaction! Think of it like trying to bake a cake, but someone already threw in the flour and eggs, and we're left with the tantalizing question: what else is needed to make this masterpiece happen? And the best part? We're doing it in an acidic kitchen, which adds a little zesty kick to the whole process!
Our star player in this chemical drama is something called Mno4-. Now, don't let the fancy letters scare you. Imagine Mno4- as this super-powered, slightly dramatic ingredient that's just begging for its supporting cast. It's like a superhero without its utility belt – it has the potential, but it needs the right tools and companions to truly shine and do its amazing chemical thing.
So, we've got this Mno4-, and it's part of what we call a "half-reaction." This means it's only half the story. Think of it like a movie trailer – it gives you a taste, a hint of the action, but you know there's a whole lot more going on behind the scenes. Our job is to be the casting director and the scriptwriter, figuring out exactly what else is needed to complete this chemical epic.
And where do we get our inspiration for this chemical casting call? The answer is in the magical land of acidic solutions! Imagine a kitchen where everything has a little bit of lemon juice or vinegar added – it makes everything brighter, a bit sharper, and ready for some serious chemical cooking. This acidic environment is our secret ingredient, our spice rack, that helps everything come together.
So, we're staring at Mno4-, this incomplete chemical character. It's like a talented singer who's missing their backup singers and the rhythm section. They can belt out a note, sure, but to make a full, show-stopping performance? They need the whole ensemble! Our goal is to find all those missing members to create a chemical symphony.
The first thing we need to do is figure out what else Mno4- is capable of becoming. Think of it as giving our superhero a wardrobe change or a new superpower. In the acidic realm, Mno4- often transforms into something called Mn2+. It’s like our dramatic performer shedding their elaborate costume and stepping into a simpler, yet still powerful, outfit.
Now, this transformation isn't just magic; it's chemistry! And like any good magic trick, there are steps involved. We’ve got to make sure all the elements are accounted for, like a meticulous chef counting every grain of sugar. We don't want any leftover ingredients messing up our final dish!
First up on our balancing adventure: the atoms. We're looking at the manganese, the 'Mn' part, and happily, there's one on each side. Phew! That's like finding out the main character is already in the picture. No need to cast a substitute for them.

But then we hit the oxygen, the 'O' part. Our Mno4- has four of these little oxygen buddies, while our potential new form, Mn2+, has none. This is where our acidic solution truly shines! Think of it as opening up the pantry in our acidic kitchen.
In an acidic environment, we have access to H2O, or water molecules, like having a pitcher of water ready for our recipe. These water molecules are like little oxygen fairies that we can bring in to balance out the oxygen atoms. So, to give Mn2+ its missing oxygen, we add four H2O molecules to that side.
Now, our oxygen count is looking much happier! We've got four oxygens on the Mno4- side and, thanks to our added water molecules, four oxygens on the other side too. It's like finally getting all the pieces of a particularly tricky puzzle section to click into place.
But hold on, the adventure isn't over! Adding those water molecules has introduced some new characters: hydrogen atoms. Our Mno4- doesn't have any hydrogen buddies, but now our Mn2+ side, with its four H2O friends, has a total of eight hydrogen atoms. This is where the acidic nature really shows its power.
In an acidic solution, we have plenty of H+ ions hanging around, like little energetic protons eager to join the party. Think of them as tiny, enthusiastic cheerleaders ready to boost the energy of our reaction. So, to balance out those eight hydrogen atoms, we need to add eight H+ ions to the side that started with just Mno4-.
Now, our atoms are all in harmony! We have one manganese on each side, four oxygens on each side, and eight hydrogens on each side. It's like everyone in the band finally got their sheet music and knows their part perfectly. The chemical atoms are singing in unison!
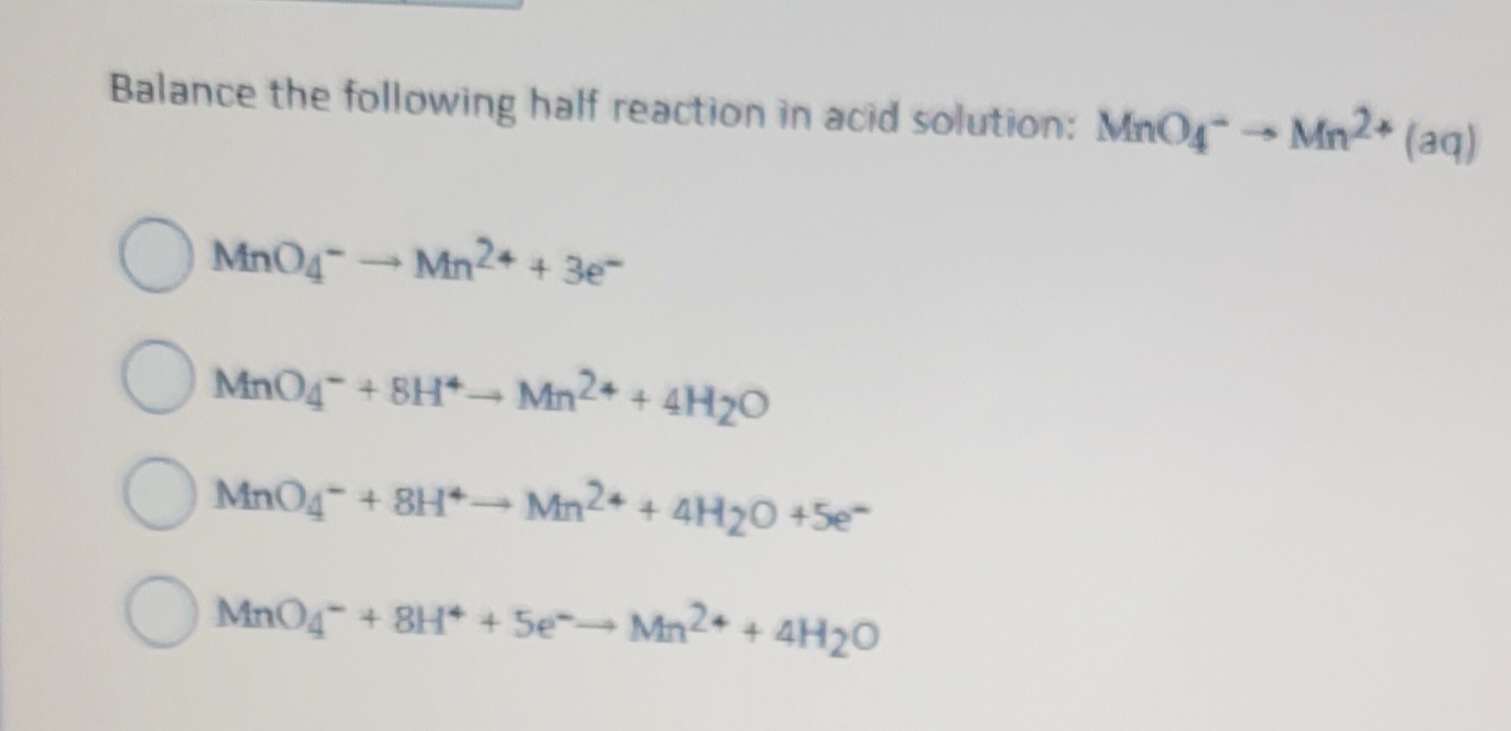
But wait, there's one more crucial element to balance: the charge! Think of charge like the electrical sparkle of our chemical friends. Everything in a neutral state has a balanced sparkle, but ions have gained or lost some sparkle, making them either a bit more negative or a bit more positive.
On the Mno4- side, we have the Mno4- ion itself, which has a charge of -1. We also added eight H+ ions, each with a +1 charge. So, the total charge on this side is (-1) + (8 * +1) = +7. It’s like a very buzzed-up party!
On the other side, we have Mn2+, which has a charge of +2, and four neutral H2O molecules, which have no charge. So, the total charge on this side is +2 + (4 * 0) = +2. This side is feeling a bit more relaxed, like it's enjoying a gentle conversation.
Now, our goal is to make these charges match. We want the same amount of sparkle on both sides. We've got a +7 sparkle on one side and a +2 sparkle on the other. It’s like having one person with a super bright flashlight and another with a dimmer one.
To balance this out, we need to add electrons. Electrons are like tiny sparks of negative energy that can reduce the overall positive charge. Think of them as little dimmer switches for our super bright flashlight.
We need to reduce the +7 charge down to a +2 charge. The difference between +7 and +2 is 5. This means we need to add 5 electrons, each with a -1 charge, to the side that has the +7 charge.

So, we add five electrons to the Mno4- and H+ side. Now, the total charge on that side becomes (+7) + (5 * -1) = +2. And behold! The charges on both sides are now a perfect +2. It’s like both people in our conversation are now speaking at the same, comfortable volume.
And there you have it! Our half-reaction, which started as just Mno4-, is now complete and balanced in our acidic solution. We've transformed it into: Mno4- + 8H+ + 5e- -> Mn2+ + 4H2O. It’s a full chemical story, with a beginning, a middle, and a very satisfying, balanced end!
Isn't chemistry fun when you think of it like solving a delightful mystery or orchestrating a grand culinary creation? We took a solo act and turned it into a full chemical chorus, all thanks to the zesty influence of our acidic solution!
So next time you see a chemical equation that looks incomplete, remember our Mno4-. It's just waiting for its chemical supporting cast to bring the whole performance to life. And with a little help from our acidic friends and some strategically placed electrons, we can balance anything!
It's like giving a shy performer the confidence and the right band to truly shine on stage. We've not only balanced the atoms and charges, but we've also brought this chemical reaction to its full, magnificent potential. High fives all around for our chemical detective work!
This process of balancing half-reactions is fundamental, like learning your ABCs before writing a novel. It ensures that the universe’s laws of conservation are respected, even in the microscopic world of atoms and electrons. It's a beautiful dance of matter and energy, and we just learned a few steps!
So, feel empowered! You've just tackled a concept that might look intimidating, but with a little imagination and a playful spirit, you’ve unlocked the secrets of this acidic chemical ballet. The world of chemistry is full of these fascinating transformations, just waiting for curious minds like yours to discover them!
The next time you encounter a similar puzzle, remember the steps: balance atoms, then oxygen with water, then hydrogen with H+, and finally, balance the charges with electrons. It’s a recipe for success, a formula for fun, and a guarantee of a well-balanced chemical outcome!
Think of Mno4- as a seed, and our balancing act as providing the perfect soil, water, and sunlight for it to grow into a beautiful, flourishing chemical plant. The acidic solution is the fertile ground, the H+ ions are the life-giving rain, and the electrons are the sunshine that makes everything blossom.
We’ve successfully completed and balanced the half-reaction for Mno4- in an acidic solution. This means we’ve identified all the missing components and arranged them in a way that satisfies the fundamental rules of chemistry. It’s like finding the perfect ending to a captivating story, leaving you satisfied and eager for the next chapter.
So, celebrate your achievement! You've just performed a little bit of chemical alchemy, transforming an incomplete equation into a complete and elegant one. This skill is a superpower in the world of science, allowing you to understand and predict how different substances interact.
Remember this feeling of accomplishment. Every balanced equation is a small victory, a testament to your growing understanding of the universe’s intricate workings. Keep that enthusiasm alive, and the world of chemistry will continue to unfold its wonders for you!
And who knows what other chemical mysteries await your keen eye and playful approach? With tools like these, you're ready to tackle anything that comes your way, one balanced half-reaction at a time. Keep exploring, keep questioning, and most importantly, keep having fun with it!
