Complete And Balance Each Of The Following Reactions

Ever looked at a recipe and seen those mysterious ingredient amounts? Or perhaps you've tinkered with a hobby project and found yourself with leftover bits and pieces? Well, in the world of chemistry, and even in some everyday scenarios, there's a surprisingly fun and useful concept called "completing and balancing reactions." It might sound a bit technical, but think of it like making sure you have the right amount of everything to create something perfectly balanced and whole. It’s a bit like solving a puzzle, and once you get the hang of it, it’s incredibly satisfying!
So, what's the big deal about completing and balancing reactions? Essentially, it's all about making sure that when things combine or change (that's the "reaction" part), you have the same amount of "stuff" (called atoms) on both sides of the equation. Imagine baking cookies: you need the exact right amount of flour, sugar, and eggs to end up with delicious cookies, not a gooey mess or a pile of dry crumbs. Similarly, in chemistry, balancing reactions ensures that no atoms are lost or magically created. This is fundamental to understanding how the world around us works, from the air we breathe to the food we eat.
For beginners, this is a fantastic way to start appreciating the logic and order in science. It's like learning the basic rules of a game. For families, it can be a fun, hands-on way to introduce kids to scientific thinking. You can use simple analogies, like building with LEGOs – you need all the same bricks in the final structure as you started with. And for hobbyists, whether you're into gardening (understanding nutrient cycles), DIY projects (knowing how different materials interact), or even brewing your own kombucha, grasping these basic principles can help you achieve better, more predictable results.
Let's think about some simple examples. Imagine you're making water. The basic ingredients are hydrogen and oxygen. If you just put them together haphazardly, you might not get the pure, refreshing water we know. In chemistry, this looks like:
H₂ + O₂ → H₂O

This looks unbalanced, right? We have two oxygens on the left and only one on the right. To balance it, we need to make sure every atom has a buddy. We can add coefficients (numbers in front of the chemical formulas) to make it work:
2H₂ + O₂ → 2H₂O
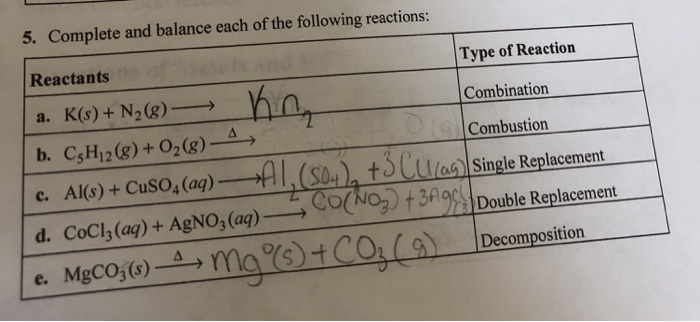
Now, we have 4 hydrogen atoms and 2 oxygen atoms on both sides. Everything is accounted for! It’s a perfect match, just like getting all your ingredients in the right amounts for a recipe.
Getting started is easier than you think. You don't need a fancy lab! Begin by understanding the symbols of common elements (like H for hydrogen, O for oxygen, C for carbon). Then, focus on the concept of atoms being conserved – they just rearrange. Look for simple, everyday reactions and try to visualize the atoms moving around. There are tons of online resources and videos that demonstrate this concept visually, which can be incredibly helpful.
Ultimately, understanding how to complete and balance reactions is about appreciating the elegance and predictability of the universe. It’s a skill that opens up a deeper understanding of science and can even make your everyday endeavors more successful and enjoyable. It’s a little bit of scientific detective work, and the satisfaction of solving the puzzle is truly rewarding!
