Compared With Group 2a Elements Group 6a Elements Have
+are+called+alkaline+earth+metals..jpg)
Hey there, science fans! Ever get a little bored with the usual suspects? You know, the elements everyone talks about. Today, we're going to dive into a couple of awesome groups from the periodic table that you might not think about much. But trust me, they’ve got some seriously cool stuff going on.
We’re going to compare and contrast Group 2A elements with Group 6A elements. Think of it like comparing two different kinds of superheroes, each with their own unique powers and personalities. One group is all about being super friendly and ready to share, while the other is a bit more mysterious and ready for adventure.
Let's start with our first crew, the Group 2A elements. These guys are also known as the Alkaline Earth Metals. You've probably heard of a couple of them, like Magnesium. Yep, that’s the stuff in your Epsom salts and also what makes fireworks sparkle!
These elements are like the popular kids at the party. They're naturally inclined to be social and give away their electrons. It’s their happy place, you know? They just want to make friends and form bonds. It’s all about being generous.
They’re quite reactive, which means they like to jump into action. Think of them as always ready to lend a hand, or in this case, an electron. This makes them super useful in lots of everyday things.
When they do decide to team up with other elements, they usually form pretty stable compounds. It’s like they found their perfect match and are ready to settle down. This is why they’re found in so many minerals and rocks.
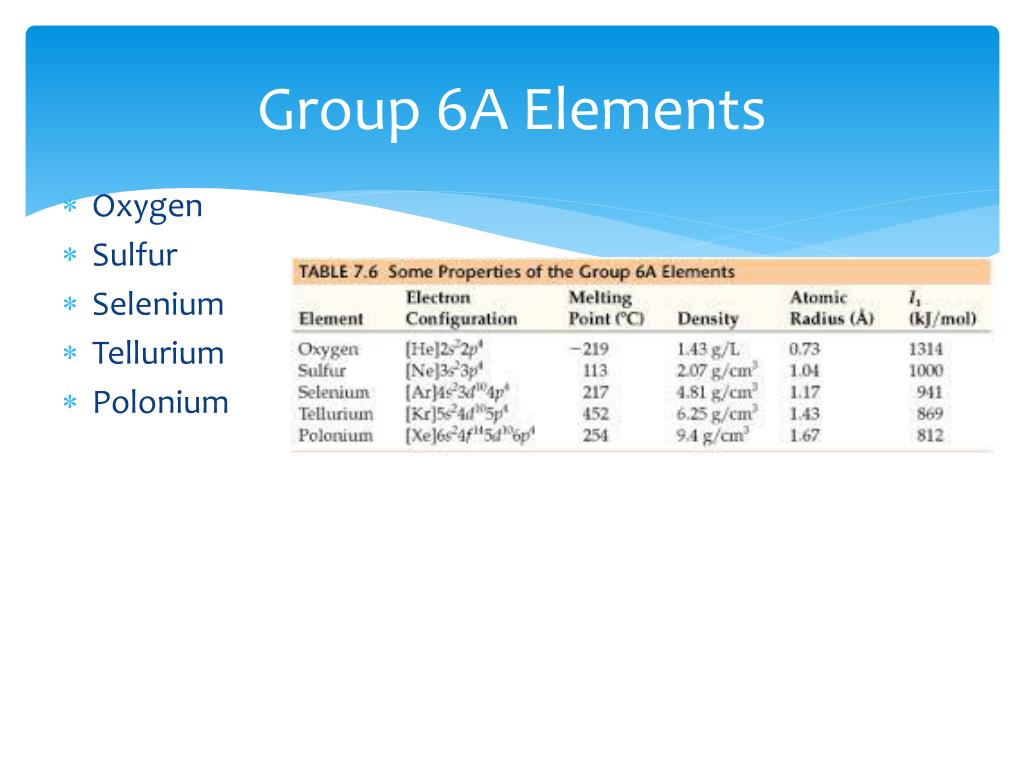
Now, let's switch gears and meet the totally different vibe of the Group 6A elements. These are often called the Chalcogens. This name sounds a bit dramatic, doesn't it? Like something out of a fantasy novel! It actually comes from Greek words meaning "ore-forming."
And why? Because these elements are super important in forming lots of important ores. They’re like the quiet but incredibly influential artisans of the element world, helping to build the foundations of so many materials.
The most famous member of this group is probably Oxygen. We all know Oxygen, right? It’s what we breathe! It’s absolutely essential for life as we know it. Talk about a superstar element!
Another member is Sulfur, which you might know from its distinctive smell. It's used in making matches and also has some cool industrial applications. It’s like the quirky artist with a very memorable signature scent.
Unlike the Group 2A elements, the Group 6A elements are a bit more reserved when it comes to giving away electrons. They’re more likely to want to take electrons from others. It’s a different strategy for making friends, but it works!

They’re also quite diverse in their personalities. Some, like Oxygen, are super reactive and eager to bond. Others, like Selenium, are a bit more laid-back. It's like having a whole spectrum of characters in one group.
So, what's the big difference that makes this comparison so entertaining? It's all about their electron-sharing tendencies. Group 2A elements are all about giving electrons. They’re the generous donors of the periodic table.
Group 6A elements, on the other hand, are more interested in taking electrons. They are the eager receivers. This fundamental difference in how they interact dictates their entire behavior and the kinds of compounds they form.
Think about it: when a Group 2A element meets a Group 6A element, it’s like a perfect match made in chemical heaven. The generous Group 2A element has electrons to spare, and the receptive Group 6A element is looking for them. Bam! A stable bond is formed. It’s a classic case of opposites attracting.
For example, Magnesium (from Group 2A) loves to give away its two outer electrons. Oxygen (from Group 6A) loves to grab two electrons. Put them together, and you get Magnesium Oxide. This is a common compound, very stable, and found in many things.
The reactivity is also a key differentiator. The Alkaline Earth Metals are pretty reactive, but generally less so than the alkali metals (Group 1A). They're enthusiastic, but not wildly so. They have a good amount of energy for bonding.
The Chalcogens have varying levels of reactivity. Oxygen is incredibly reactive, driving reactions all over the place. Sulfur is also quite reactive. But as you go down the group, the reactivity tends to decrease.
It’s this difference in electron configuration and their resulting drive to gain or lose electrons that makes them so distinct. It’s not just about what they are, but what they do that’s so fascinating.
The way these elements interact leads to a huge variety of compounds. Group 2A elements form ionic compounds, where one atom essentially gives an electron to another. It’s a clean transfer.
Group 6A elements, by taking electrons, also contribute to forming these ionic compounds. But they can also form covalent bonds, where electrons are shared. This happens more when they interact with other non-metals.
The difference in their electronegativity is a big part of this. Electronegativity is basically an atom's "pull" for electrons. Group 6A elements have much higher electronegativity than Group 2A elements. They’re the strong magnets in the electron world.
So, while both groups are essential parts of the chemical universe, they play very different roles. One is the eager giver, the other is the capable receiver. This simple difference leads to a world of different chemical reactions and materials.
It's kind of like comparing a baker who loves to give away free samples (Group 2A) to a chef who is an expert at incorporating amazing ingredients into a dish (Group 6A). Both are crucial to a delicious outcome, but their methods are distinct.
Understanding these fundamental differences helps us appreciate why certain elements combine the way they do. It explains the formation of minerals, the reactions that sustain life, and the creation of all sorts of materials we use every day.
It’s this underlying chemistry, this dance of electrons, that makes the periodic table so incredibly engaging. These seemingly simple rules lead to a complex and beautiful world of compounds.
So next time you hear about Magnesium or Oxygen, remember their elemental families and their unique ways of interacting. It’s a little peek into the amazing forces that shape our universe, all thanks to the way atoms decide to share (or not share!) their electrons.
It’s this constant interplay of giving and taking, of attraction and repulsion, that makes chemistry so dynamic. The Group 2A and Group 6A elements are perfect examples of this fundamental principle at work. They’re not just dots on a chart; they’re active participants in a grand cosmic ballet.
The fact that elements can be categorized by such fundamental behaviors—like how readily they give up electrons—is truly remarkable. It allows us to predict their behavior and understand their roles. It's like having a cheat sheet for the universe's building blocks.
So, while Group 2A elements might be the friendly sharers and Group 6A elements the keen collectors, both are indispensable. They represent two sides of the same coin, crucial for the formation of the world around us. It’s a beautiful chemical relationship.
I hope this little dive into the world of Group 2A and Group 6A elements has sparked your curiosity. It's a reminder that even the most basic components of our world have incredible stories to tell. Keep exploring, and you might be surprised by what you discover!
So, what makes it so entertaining? It's the simple yet profound difference in how they interact with other elements! One group is the generous giver, the other, the eager receiver. This fundamental difference drives the formation of countless compounds, from the air we breathe to the minerals in the earth. It's a constant cosmic dance of electrons, and it's utterly captivating!
There’s a certain elegance to it, isn’t there? These elements, just by having a different number of electrons in their outer shells, behave in completely different ways. It's a testament to the power of fundamental principles in chemistry.
The Alkaline Earth Metals are always ready to shed their two valence electrons to achieve a stable electron configuration. This makes them electropositive and excellent reducing agents. They’re the helpful friends who always have your back (and your extra electrons).
On the other hand, the Chalcogens are known for their high electronegativity. They have a strong desire to gain two electrons to complete their outer shell. This makes them excellent oxidizing agents and allows them to form a wide range of compounds, often with metals.
When you think about it, this sets up a predictable pattern. You can often guess what kind of compound will form based on which groups are involved. It’s like knowing that if you put salt and pepper together, you’ll get seasoning! It’s that foundational.
The Group 2A elements are shiny, silvery metals. They're relatively soft and can be cut with a knife, though not as easily as the alkali metals. They are always found in nature combined with other elements, as they are too reactive to exist in their pure form.
The Group 6A elements, however, are much more diverse in their physical states. Oxygen is a gas. Sulfur is a yellow solid. Selenium is a reddish-brown solid. Tellurium is a silvery-white solid. And Polonium is a rare, radioactive metal.
This diversity within Group 6A adds another layer of fascination. It shows how even within a group defined by similar electron configurations, subtle differences can lead to vastly different properties and appearances. It’s like having siblings who all look alike but have completely different personalities and talents.
The bonding behaviors are key. The Group 2A elements primarily form ionic bonds. For instance, Calcium reacts with Chlorine (a Group 7A element) to form Calcium Chloride. This is a strong, crystalline solid.
Group 6A elements can form ionic bonds, especially with metals. But they also readily form covalent bonds with non-metals. Consider water, H₂O. Here, Oxygen (Group 6A) forms covalent bonds with Hydrogen (a non-metal). This is a completely different type of interaction!
This ability to form both ionic and covalent bonds makes Group 6A elements incredibly versatile. They are the chameleons of the chemical world, adapting their bonding style to suit their partners. It’s truly amazing to consider the range of chemistry they enable.
So, while the Group 2A elements are the steadfast givers, forming stable ionic compounds with their electron-hungry partners, the Group 6A elements offer a broader palette of interactions. They are essential for life, for industrial processes, and for a whole spectrum of chemical phenomena.
It's this interplay of electron donation and acceptance, of forming ionic or covalent bonds, that makes comparing these two groups so incredibly rewarding. It's a fundamental lesson in chemistry, illustrated by the very building blocks of our universe. And that, my friends, is pretty darn cool.
