Comparative Reactions Of Carboxylic Acid Derivatives
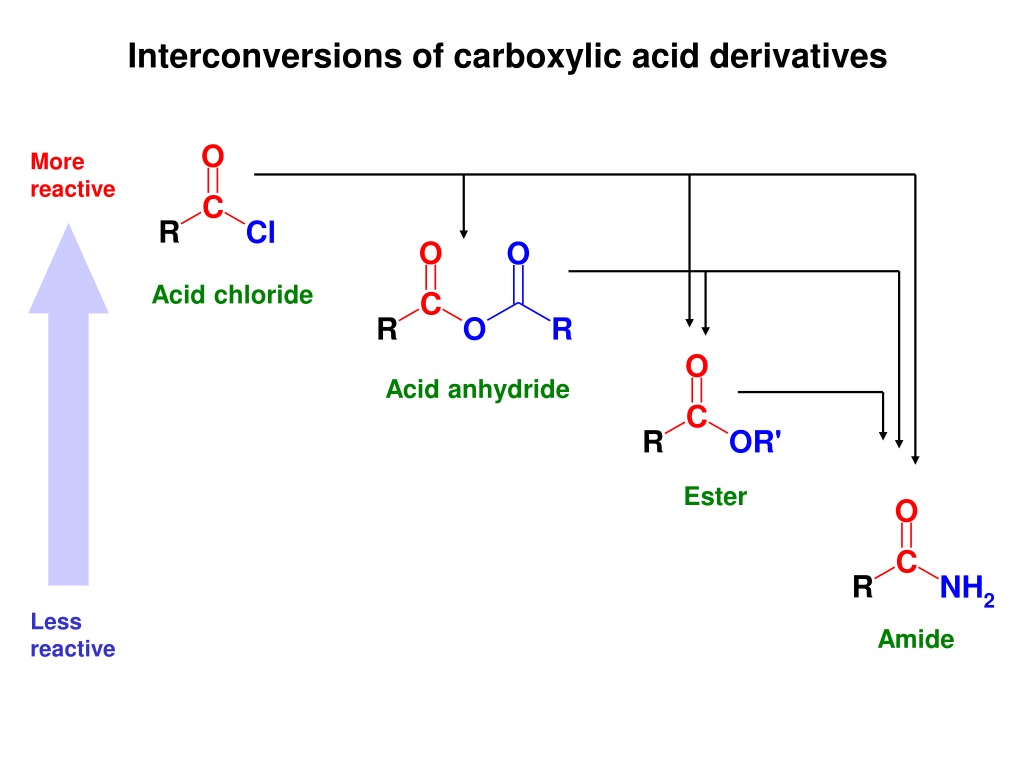
Imagine a big family reunion. You've got the sophisticated aunt, the boisterous uncle, the quiet cousin, and the enthusiastic teenager. In the world of chemistry, carboxylic acid derivatives are a bit like that. They're all related, sharing a common ancestor, but they have their own unique personalities and react in wonderfully different ways. It's like a chemistry family drama, but way more interesting and with fewer awkward silences.
At the heart of this family is our "parent molecule," carboxylic acid. Think of it as the wise grandparent, always there with solid advice and a comforting presence. It's a stable, reliable sort of character. But when it starts to mingle and make friends, things get a lot more exciting.
The Enthusiastic Newcomers: Acid Halides
First up, we have the "energetic youngsters," the acid halides. These guys are like the teenagers at the reunion, always eager for action and a bit reckless. They're incredibly reactive, jumping into situations with both feet.
If you introduce them to just about anything – a bit of water, an alcohol, even a friendly amine – they'll react almost instantly. It’s like they can’t wait to get their hands dirty. They're the life of the party, but you have to keep a close eye on them!
Their eagerness comes from a really electronegative atom, usually chlorine or bromine, hanging out with them. This atom is like a tiny, super-strong magnet, pulling all the attention and making the rest of the molecule super ready to react. It's this powerful pull that makes them so quick to change.
Think about making something fizzy. An acid halide is like the instant yeast that makes dough rise in minutes. They’re fast, they’re dramatic, and they get the job done with a flourish. They’re responsible for creating some really useful compounds, like certain plastics and medicines, all thanks to their explosive energy.

The Generous Givers: Esters
Next, we meet the "kind and giving" members of the family: the esters. These are the ones who always bring a delicious homemade treat to the reunion. They’re generally quite well-behaved, but they have a generous spirit.
Esters are responsible for some of the most delightful scents we know. Think of the sweet aroma of ripe strawberries or the intoxicating fragrance of lavender – that’s the magic of esters at work! They're the perfume bottles of the chemical world.
While they’re not as quick to react as acid halides, they're still quite amenable. When you introduce them to water, especially with a little help from an acid or a base, they're happy to break down and share their components. It's like they're willingly giving away their flavorful essence.
This ability to be broken down is key to how they function in nature. Many natural fats and oils are actually long chains of esters. Your body uses these esters as a source of energy, so in a way, they're literally giving you the fuel to live and enjoy life. It’s a heartwarming thought, isn't it?
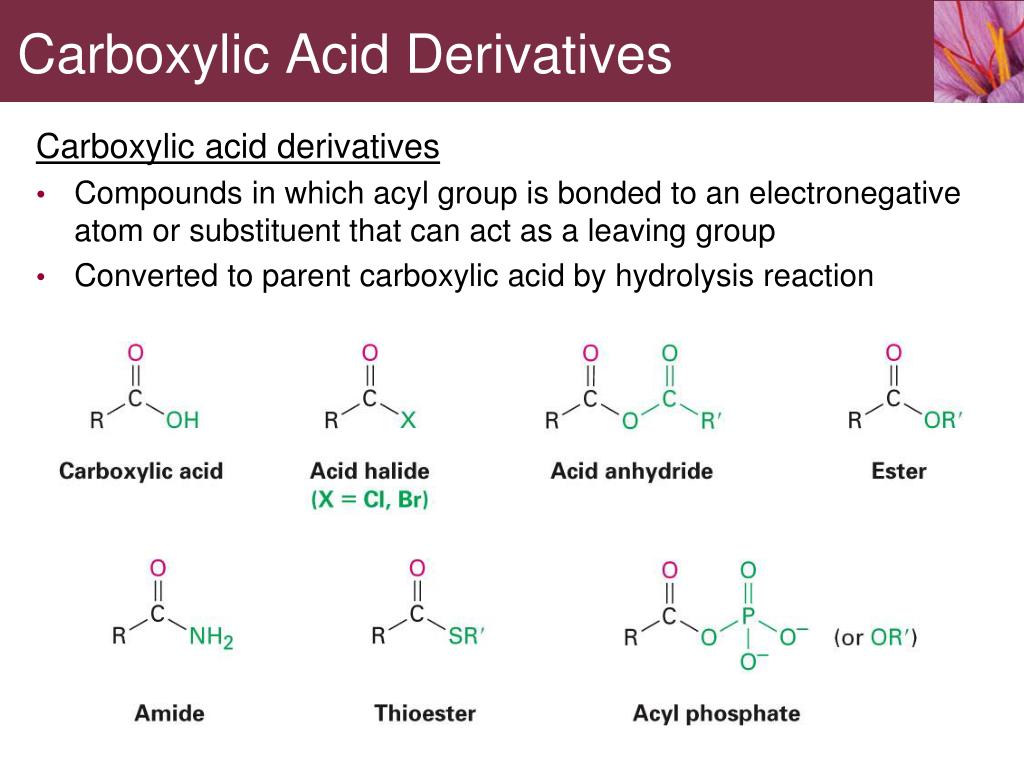
And who can forget the deliciousness? Many artificial flavorings and fragrances are also esters. That burst of fruity flavor in your candy or the pleasant scent of your soap? Thank an ester! They're the unsung heroes of sensory pleasure.
The Dependable Helpers: Amides
Now, let’s talk about the "rock-solid, dependable" members: the amides. These are the folks who are always there for you, the steady hands you can rely on. They’re not as flashy as the acid halides, nor as perfumey as the esters, but they’re incredibly important.
Amides are the building blocks of life itself! Yes, you read that right. The very proteins that make up your muscles, your hair, and your enzymes are all linked together by amide bonds. They're the fundamental connectors of the biological world.

These guys are notoriously stable. Trying to get an amide to react is like trying to get a grumpy teenager to do chores – it takes a lot of effort and strong persuasion. They don't break down easily, and that’s a good thing!
It's this strong, stable nature that makes them perfect for building complex structures like proteins. Imagine trying to build a skyscraper with flimsy materials; it wouldn't last. Amides provide the strong, reliable scaffolding for life.
When they do react, it’s often a slow and deliberate process. Think of digestion, where enzymes painstakingly break down proteins over time. It’s not a dramatic explosion, but a carefully orchestrated, life-sustaining process.
The Overworked Relatives: Anhydrides
Finally, let's meet the "always-busy, slightly stressed" relatives: the anhydrides. These are the ones who have taken on a lot of responsibility. They're essentially two carboxylic acid molecules that have gotten rid of a water molecule to bond together.
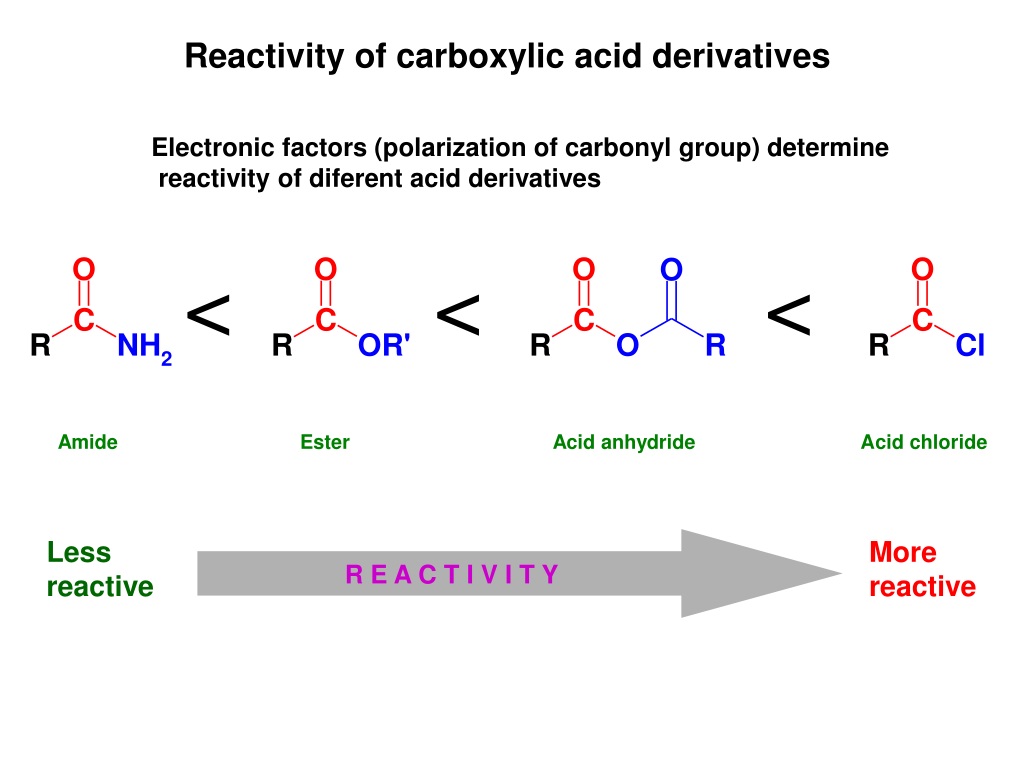
Because they’re formed by losing water, they’re eager to get it back. This makes them quite reactive, a bit like the acid halides but perhaps a tad more measured. They’re ready to spring back to their original, less stressed form.
When you introduce an anhydride to water, it’s like a sigh of relief. They happily absorb the water and split back into two carboxylic acids. It’s a reunion of sorts, but with a splash!
They’re often used in the creation of important materials, like aspirin. Aspirin, that everyday pain reliever, is made using an anhydride. It’s a powerful reminder that even the "stressed" members of the family play crucial roles in our well-being.
So, the next time you encounter a carboxylic acid derivative, whether it’s the sweet scent of an ester, the strength of a protein amide, the dramatic flair of an acid halide, or the helpful nature of an anhydride, remember their unique personalities. They’re all part of the same chemical family, but their individual quirks make the world a richer, more interesting, and often more delightful place. It's a family story written in molecules, and it’s constantly unfolding.
