Collisions Covalent Bonding Answer Key Level 15
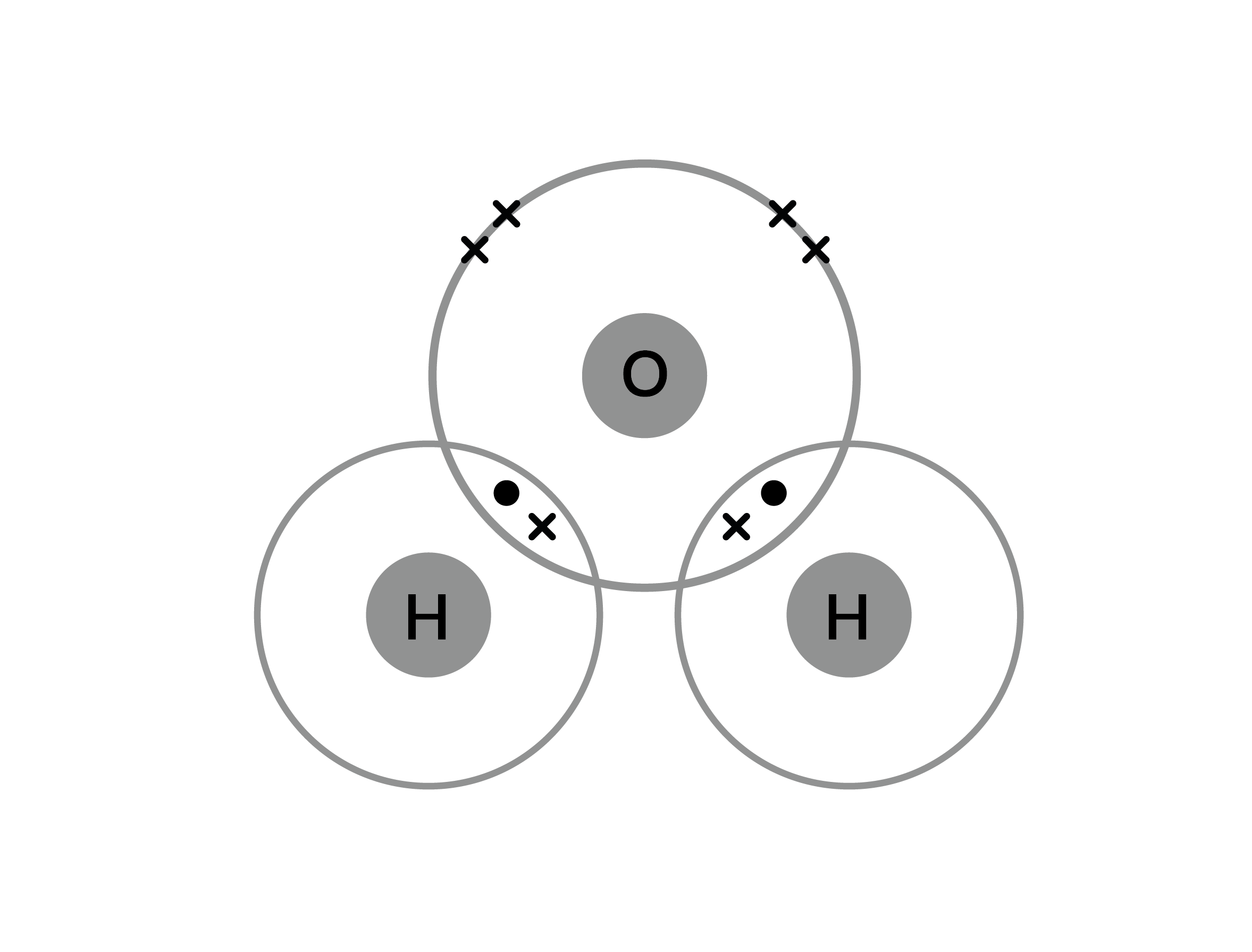
Okay, so picture this: you're casually scrolling through some super intense science quiz, probably late at night, fueled by questionable snacks. Suddenly, you hit a wall. Not a literal wall, thankfully. It’s a covalent bond. And then BAM! You're staring at a question about Collisions Covalent Bonding Answer Key Level 15. It sounds like a secret handshake for a really exclusive club, doesn't it?
I’m pretty sure Level 15 is where the real magic, or maybe just the real head-scratching, happens. It’s like unlocking a new level in a video game, but instead of dragons, you’re fighting with electron sharing. And the "collisions"? Well, in the wacky world of chemistry, "collisions" often means the humble atoms bumping into each other. Sometimes they just bounce off, like when you try to have a deep conversation with someone after they've had three coffees. Other times, if the conditions are just right, and maybe they've had a good chat about sharing their toys (electrons, in this case), they decide to stick together. That's where the covalent bond swoops in, like a superhero holding them in a permanent, electron-powered embrace.
And the answer key? Ah, the mythical creature whispered about in hushed tones. It's the golden ticket, the cheat code, the magical map to understanding why certain atoms are just so keen to hold hands. You know, the ones that decide sharing is caring when it comes to those tiny, zippy things called electrons. They’re not just passing acquaintances; they're practically roommates, sharing that valuable electron space. It’s a whole different vibe from those ionic bonds where one atom is like, "Here, you take it!" and the other is like, "Thanks, I'll have it!"
Honestly, sometimes I think Level 15 of Collisions Covalent Bonding is designed by someone who just really enjoyed making people sweat. It’s like they said, "Let’s take the concept of atoms holding hands and make it sound like a demolition derby. And then, let's hide the instructions for how it all works!" And we, the brave adventurers of learning, are left pondering the deep mysteries of these microscopic mosh pits. We’re wondering, was it a glancing blow? A full-on smash-up? Did they exchange electrons like precious gifts or just sort of nudge them closer?
The idea of electrons "colliding" and then deciding to form a covalent bond is, I feel, an unpopular opinion of mine but a profoundly beautiful one. It’s not just a sterile chemical reaction; it’s a microscopic drama. Imagine two electrons, swirling around their respective nuclei, going about their business. Then, whoops, they get a little too close for comfort. Instead of a cosmic "excuse me!", they find themselves drawn together, sharing their space. It’s like two strangers on a bus accidentally bumping knees and then striking up the best conversation of their lives. They realize they have so much in common, so many electrons to share, that they decide to stick together and become a molecule. A molecule that, I might add, probably wouldn't exist without that initial, awkward electron shuffle.

And when you’re staring at that Level 15 question, and you’re trying to figure out the exact angle of the collision, or the precise energy involved, you just want to scream, "Can’t we just say they got along and bonded?!" But no, the science gods demand precision. They want to know if the collision was energetic enough to overcome the repulsion, but not so energetic that they just blast each other into oblivion. It’s a delicate dance, a scientific tightrope walk, and Collisions Covalent Bonding Answer Key Level 15 is your slightly cryptic guide to not falling off.
Think about it. The very existence of water, of the air we breathe, of the complex organic molecules that make us us, all hinge on these tiny, energetic bumps and subsequent electron-sharing arrangements. It's a testament to the universe's ability to create order and beauty from what, on the surface, might seem like pure chaos. A chaotic collision leading to a stable, functional bond. It’s like a messy artist creating a masterpiece. Or a toddler making a glorious mess that somehow ends up looking like art. The outcome is what matters, and in the case of covalent bonding, the outcome is everything.
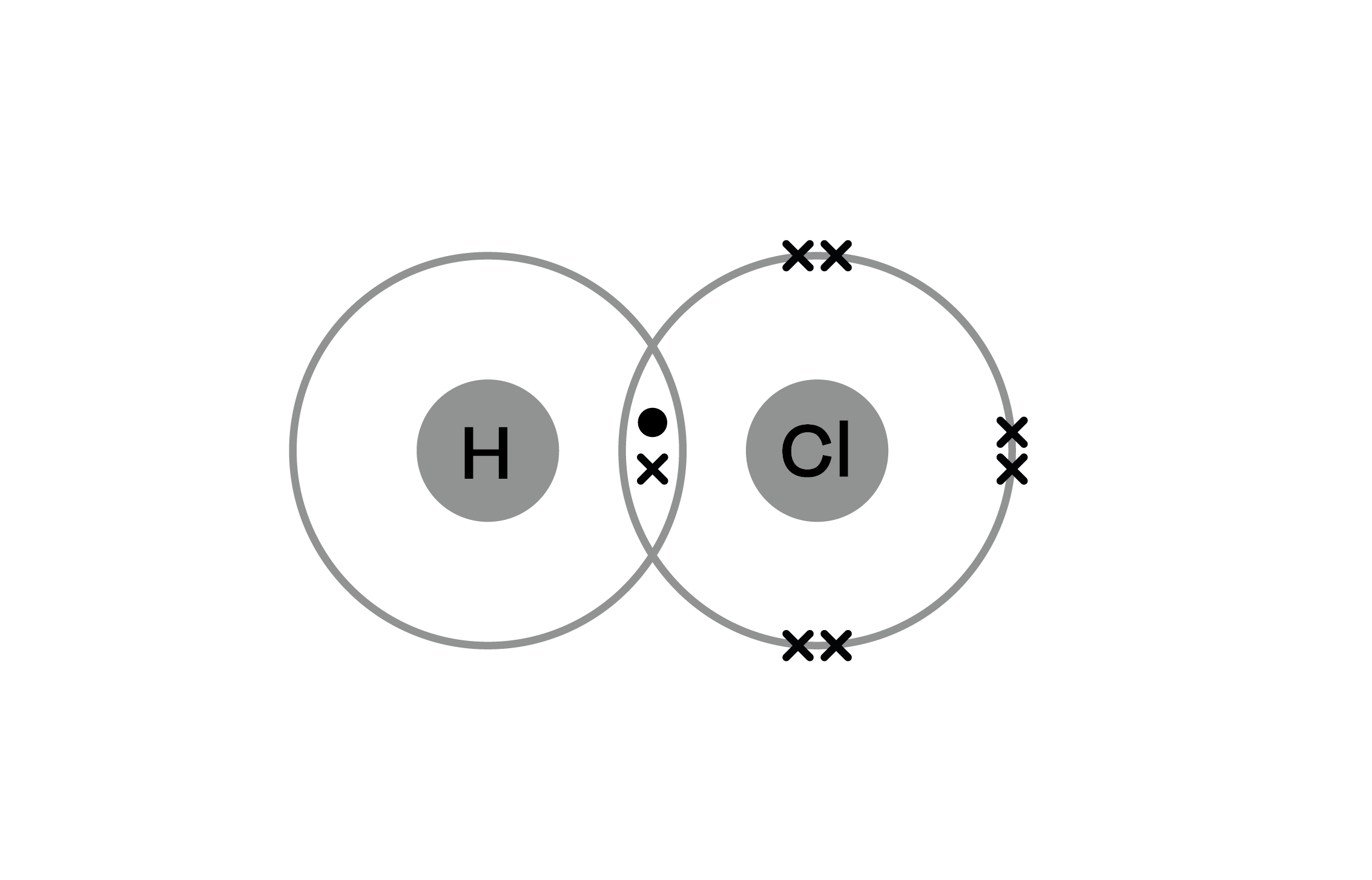
So, the next time you encounter Collisions Covalent Bonding Answer Key Level 15, take a moment. Smile. Acknowledge the absurdity and the elegance of it all. Because while the answer might be a specific number or a chemical formula, the journey to that answer involves microscopic events that are, in their own way, quite spectacular. It’s not just science; it’s a tiny universe of interactions, and we're just trying to decode its language. And sometimes, that language sounds suspiciously like atoms having a really good electron-sharing party after a bit of a fender-bender.
Maybe Level 15 is just the universe's way of telling us that sometimes, the best things happen after a little bit of a bump.
It’s a thought, isn’t it? This whole idea of how seemingly random events, like atomic collisions, can lead to incredibly stable and fundamental structures. It’s like finding that perfect parking spot right in front of the store after circling the block for ten minutes. A moment of chance, a bit of maneuvering, and poof, you're connected. And that answer key? It’s just the little note that tells you, "Yep, that collision was just the right kind of important." Don't let it intimidate you; let it amuse you. Because understanding the universe, one electron at a time, is a pretty hilarious journey.
