Cocl2 Decomposes According To The Equation Above
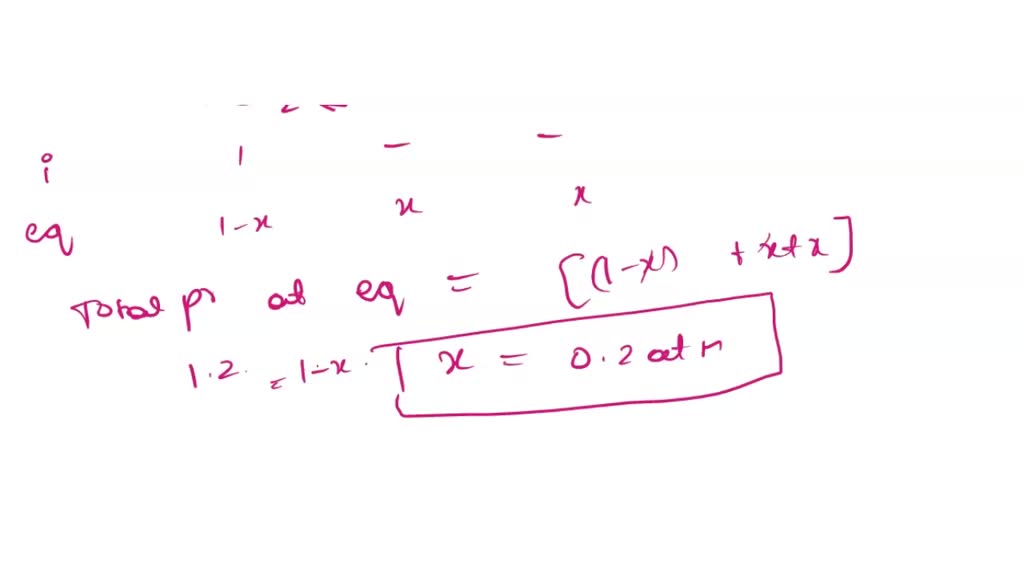
Hey there, science enthusiasts and curious minds! Ever wondered what happens when you mess with stuff? Like, what if you took a common chemical and gave it a little nudge, say, with some heat? Well, today we're diving into something pretty neat: the decomposition of cobalt(II) chloride, or CoCl₂ as the chemists like to scribble it down. You might have seen this stuff before, maybe in those cool humidity indicators that change color. Pretty neat, right?
Now, the equation for this whole shindig is apparently "above." Since I can't actually see it floating there, let's imagine it together. We've got our friend, CoCl₂, chilling out. Then, BAM! Something happens, and it breaks apart into simpler bits. It’s like when you have a perfectly built LEGO castle, and then a playful cat (or maybe just a strong gust of wind!) comes along and scatters the bricks. Nature just loves taking things apart, doesn't it?
So, What's the Big Deal?
You might be thinking, "Okay, chemicals break apart. So what?" Well, it's not just any chemical breaking apart. This is cobalt(II) chloride, and its little breakup party is actually quite informative. It tells us about the building blocks of matter and how they can be rearranged. It's like looking at the ingredients list after a cake has been baked – you can see what went into it, and how it all came together. Pretty fundamental stuff, if you ask me.
The equation, whatever it is, shows us the products. What does CoCl₂ turn into when it decides it's had enough of being a molecule? Does it become its constituent elements, cobalt and chlorine? Or perhaps it forms something else entirely, like a different compound and some stray atoms? The possibilities are endless, and that's what makes chemistry so exciting – it's like a giant puzzle where you get to figure out the pieces and how they fit.
Why Cobalt(II) Chloride?
Why are we singling out CoCl₂ for this decomposition discussion? Well, cobalt itself is a pretty interesting element. It's known for its vibrant blue color, which is why CoCl₂ is often used in those color-changing applications we mentioned earlier. Think about it: a simple chemical, exposed to moisture, changes its color. It’s like a mood ring for your surroundings!
When CoCl₂ decomposes, it's not just a random act. It's governed by the laws of chemistry. Heat is often the trigger. Imagine you're trying to get a stubborn lid off a jar. You might run it under hot water, right? Heat provides energy, and sometimes, that energy is just enough to break the bonds holding a molecule together. It’s like giving the molecule a good shake-up until it lets go.
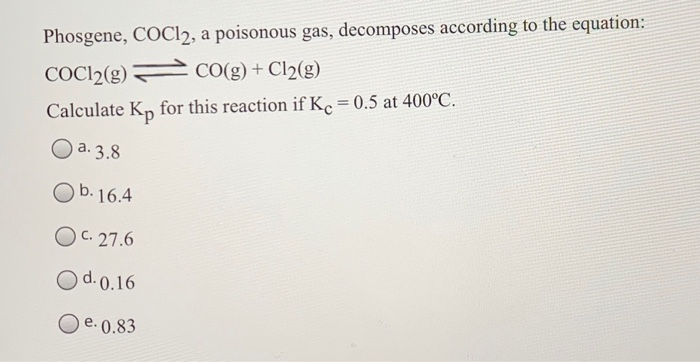
So, the equation "above" likely shows that when CoCl₂ gets sufficiently heated, it's going to undergo some kind of transformation. Maybe it releases some chlorine gas. Ever heard of chlorine gas? It’s a bit pungent, definitely not something you want to be breathing in a concentrated dose! But in controlled scientific settings, it’s a valuable byproduct. It’s like when you're baking cookies and you get that wonderful aroma filling your kitchen – a delightful side effect of a chemical process.
And what's left behind? Likely some form of cobalt oxide. Cobalt oxides are also known for their colors, often in shades of red, blue, or green, depending on the specific oxide and how it's made. So, our initial blue compound might break down and reform into something else, possibly with a different hue. It’s a chemical metamorphosis happening right before our eyes, or at least, on our lab benches!
The Chemistry Behind the Scene
Let's get a little more specific, even without seeing the exact equation. A common decomposition reaction for hydrated cobalt(II) chloride (meaning it has water molecules attached) might look something like this (and remember, this is a hypothetical representation for our discussion):

CoCl₂·6H₂O (solid, pink) + Heat → CoCl₂ (solid, blue) + 6H₂O (gas)
See? The pink hydrated form, when heated, loses its water molecules and becomes the anhydrous (water-free) blue form. This is the magic of those humidity indicators! When it’s humid, the anhydrous blue CoCl₂ absorbs water, turning pink again. When it dries out, it releases the water and turns blue. It's a reversible reaction, kind of like a chemical seesaw!
But what if we keep heating the anhydrous CoCl₂? That's where the "decomposition" part really kicks in. At much higher temperatures, the cobalt(II) chloride itself might start to break down further. A possible outcome could be something like:
2CoCl₂ (solid, blue) + Heat → 2CoO (solid, black/gray) + Cl₂ (gas)

In this scenario, our blue cobalt(II) chloride breaks down into cobalt(II) oxide (CoO), which is often a black or gray solid, and chlorine gas (Cl₂). So, our original blue compound has transformed into a different colored solid and a rather reactive gas. It’s like a magic trick where something familiar turns into something new and a bit… energetic!
Why This is So Cool
This whole process is fascinating because it demonstrates several key chemical principles. Firstly, it shows that compounds are not immutable. They can be broken down into simpler substances. This is the essence of chemical decomposition.
Secondly, it highlights the role of energy, particularly heat, in driving chemical reactions. Without enough energy, these bonds will just stay put. It’s like trying to convince someone to change their mind without giving them any good reasons – they'll probably stick to their guns! Heat provides those "good reasons" for the chemical bonds to break.
![[Solved]: Hydrogen iodide decomposes according to the e](https://media.cheggcdn.com/study/d2b/d2bc30c7-b1e0-4a32-8551-b7c49811ebdc/image.png)
Thirdly, it introduces us to the concept of different oxidation states and compounds. We’re not just dealing with cobalt and chlorine in one form, but potentially in different combinations and forms of compounds, like cobalt(II) chloride and cobalt(II) oxide. This is like learning about different members of a family – they might share some traits, but they are distinct individuals.
And let's not forget the visual aspect! The color changes associated with cobalt compounds are just stunning. From the pink of hydrated CoCl₂ to the blue of anhydrous CoCl₂, and then potentially to the black or gray of CoO, it's a whole spectrum of visual cues telling us what's happening. It’s like a chemical light show!
Think about how this applies to the real world. Understanding how substances decompose helps us in so many areas. It’s crucial for understanding how materials degrade over time, how to create new materials with specific properties, and even how to manage chemical waste. It's the bedrock of so much innovation!
So, even though the specific equation for CoCl₂ decomposition might be "above" and out of my immediate reach, the principles behind it are universal and incredibly cool. It’s a reminder that the world around us is constantly in flux, undergoing transformations that we can observe, understand, and even harness. The next time you see something change color or crumble with age, remember that there's a whole world of chemistry at play, just like our friend cobalt(II) chloride, breaking down and becoming something new. Pretty neat, right?
