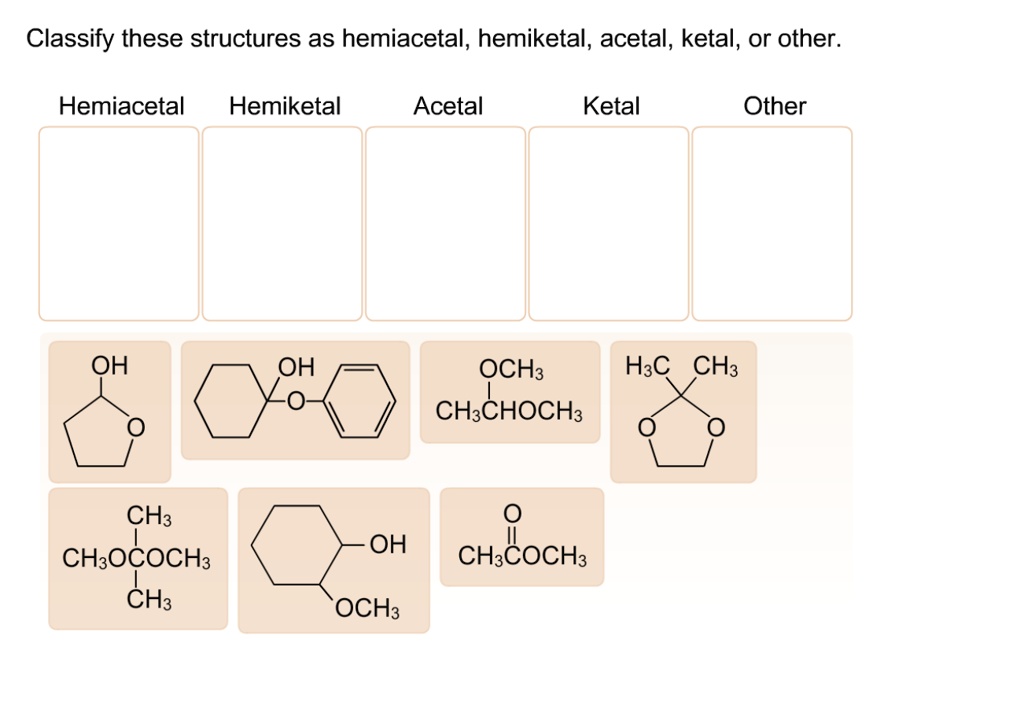
Classify These Structures As Hemiacetal Hemiketal Acetal Ketal Or Other
Ever look at a molecule and think, "Wow, that's some fancy chemical architecture!"? Well, you're not alone! The world of organic chemistry, with its intricate structures and fascinating reactions, can be surprisingly engaging. Today, we're diving into a specific type of molecular structure that pops up surprisingly often, especially when we talk about sugars and other organic compounds. Understanding these particular groupings of atoms can unlock a deeper appreciation for how molecules interact and transform. It’s like learning to spot different kinds of LEGO bricks – once you know what they are, you start seeing them everywhere, building all sorts of amazing things!
Why is this so useful, you ask? Think of it like being a detective. When you encounter a new chemical compound, being able to classify its functional groups is like finding crucial clues. It tells you a lot about the compound's potential properties and how it might behave in different situations. For instance, if you know a molecule has a certain functional group, you can make educated guesses about its reactivity, its solubility in water, or even its taste! This is fundamental in fields ranging from medicine and pharmaceuticals (designing new drugs) to food science (understanding flavor and preservation) and materials science (creating new plastics and polymers).
Learning to classify these structures is a key skill for anyone interested in chemistry, providing a solid foundation for understanding more complex chemical concepts.
So, let's get down to business. We’re going to be looking at structures that involve a carbon atom bonded to both an oxygen atom (part of an –OH group, a hydroxyl) and another oxygen atom that’s also part of a carbon chain. These are often found in cyclic molecules, especially when sugars ring-close. The magic happens when a carbon atom ends up being connected to two oxygen atoms, and at least one of those oxygens is also connected to a hydrogen atom.
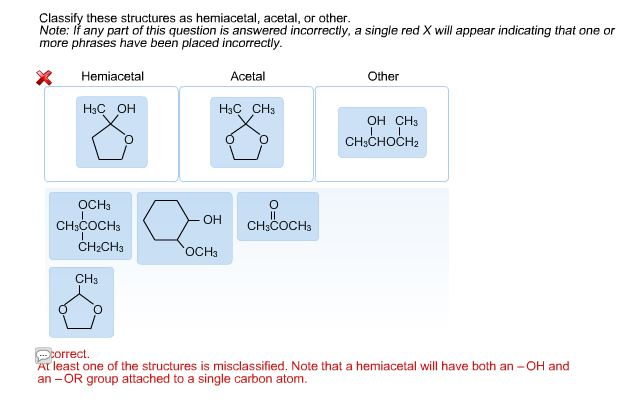
Our first category is the hemiacetal. This is where a carbon atom is bonded to an –OH group and an –OR group (where R is a carbon-containing chain or ring). Crucially, this carbon atom also needs to be bonded to at least one hydrogen atom. You often see hemiacetals when an aldehyde (a carbon double-bonded to an oxygen and single-bonded to a hydrogen) reacts with an alcohol. It's like a temporary handshake between two different functional groups.
Next up, we have the hemiketal. This is very similar to a hemiacetal, but it originates from a ketone instead of an aldehyde. So, in a hemiketal, a carbon atom is bonded to an –OH group and an –OR group, and it’s also bonded to two other carbon atoms (no hydrogen directly attached to this central carbon in the original ketone structure). Imagine a ketone – a carbon double-bonded to an oxygen – getting together with an alcohol. The result? A hemiketal!
Now, things get a little more stable with the next two. Enter the acetal. An acetal forms when the hemiacetal reacts further, typically with another alcohol. In an acetal, the central carbon atom is now bonded to two –OR groups (meaning two alkoxy groups) and two other carbon atoms. It has lost its –OH group. This is a more permanent and often more stable arrangement than a hemiacetal. Think of it as the hemiacetal fully committing to its partnership with alcohols!
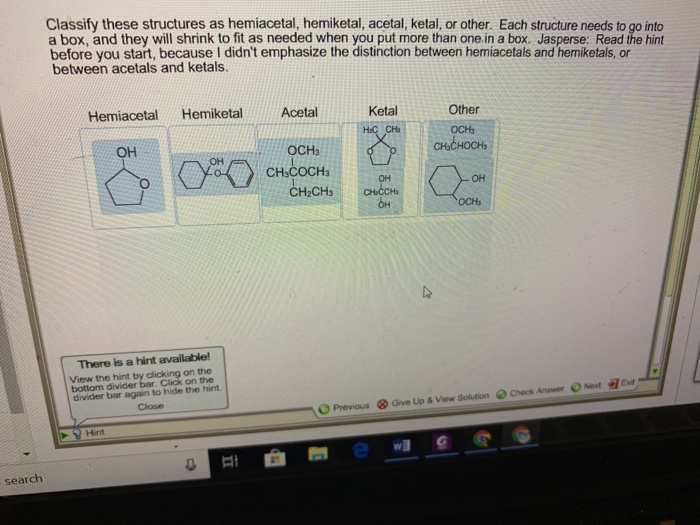
Following suit, we have the ketal. Just like the acetal is the more stable form of the hemiacetal, the ketal is the more stable form of the hemiketal. Here, the central carbon atom is bonded to two –OR groups and two other carbon atoms. It's the result of a hemiketal reacting with another alcohol, shedding its –OH group in the process. Ketals are generally quite stable molecules.
Sometimes, structures don't fit neatly into these categories. That’s where other comes in! This is our catch-all for anything that doesn't match the precise definitions of hemiacetal, hemiketal, acetal, or ketal. Perhaps the carbon is bonded to more than two other atoms, or the oxygen atoms are arranged differently, or it's a part of a larger, more complex functional group. These could be parts of esters, ethers, carboxylic acids, or a myriad of other organic structures. The key is to identify the defining characteristics of the acetal and ketal families, and if a structure doesn't fit those, it falls into the 'other' category.
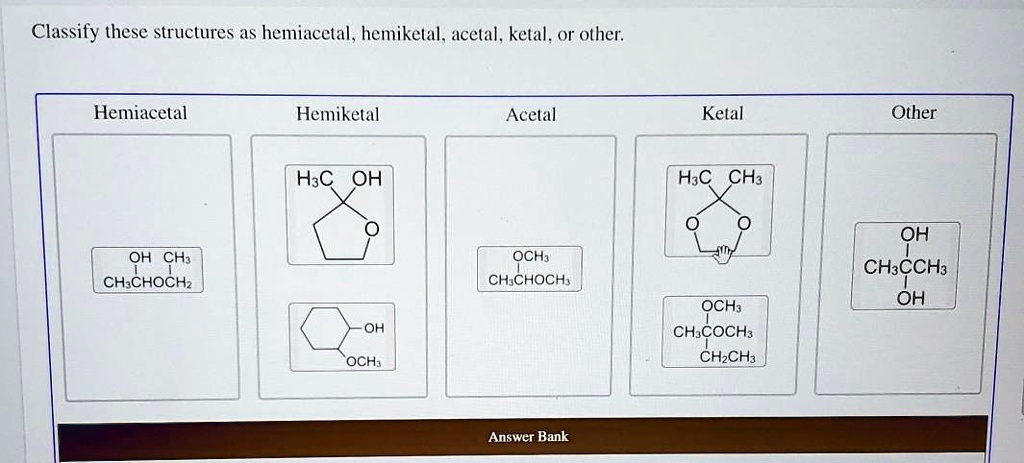
So, how do we put this into practice? Let's say you’re presented with a molecule. First, identify the carbon atom that has multiple oxygen attachments. Then, count how many –OH groups and –OR groups are attached to that specific carbon. Also, check what else is bonded to it. Is it a hydrogen and two other carbons (forming an aldehyde origin)? Or is it two other carbons (forming a ketone origin)? If it has an –OH and an –OR, it's a hemiacetal or hemiketal. If it has two –OR groups, it's an acetal or ketal. If the carbon in question has an –OH and two –OR groups, for example, or if the oxygens are structured differently, it's likely 'other'.
Mastering these classifications might seem like a small step, but it's a giant leap in understanding organic chemistry. It’s about recognizing patterns and building your chemical vocabulary. So, next time you see a chemical drawing, try your hand at classifying these structures. You might be surprised at how often you encounter these fascinating molecular building blocks!
