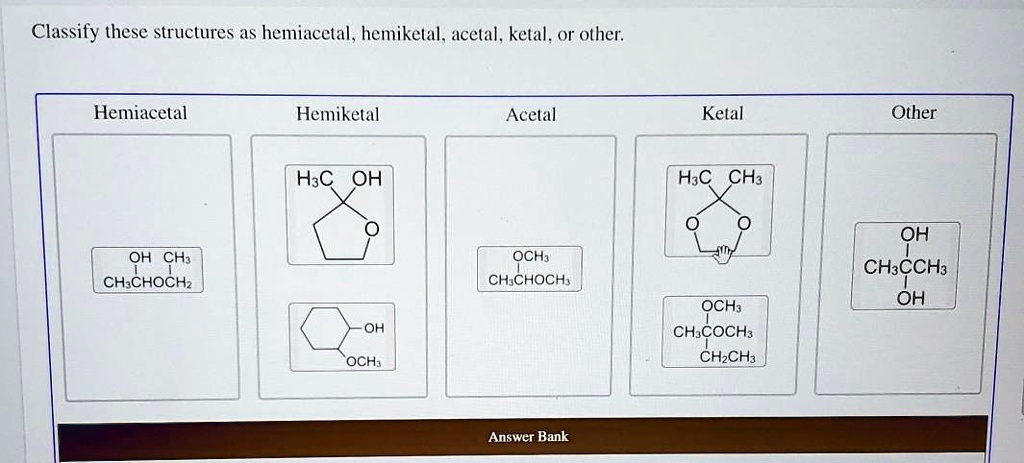
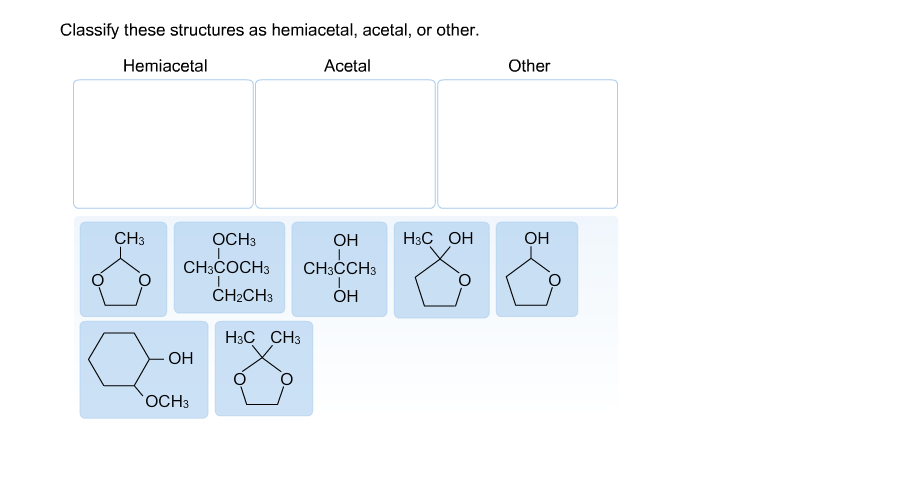
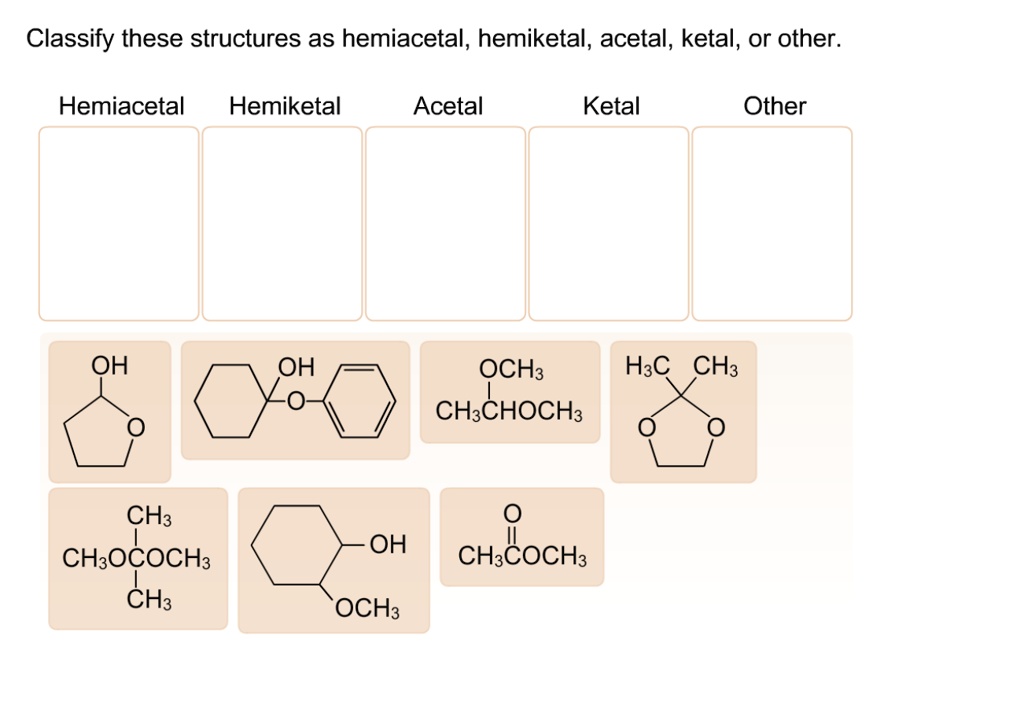
Classify These Structures As Hemiacetal Acetal Or Other
Ever found yourself staring at a molecule and wondering, "What on earth is that thing?" You're not alone! Sometimes, these tiny building blocks of life can look like they've been drawn by a very enthusiastic toddler with a penchant for loops and dots.
But fear not, aspiring chemists and curious minds! We're going on a little adventure today, a treasure hunt through the molecular world. We're going to learn to spot some familiar faces in this microscopic crowd.
Think of it like identifying your favorite characters in a cartoon. You know a superhero when you see one, right? Well, we're going to learn to recognize our own molecular superheroes: the hemiacetal, the acetal, and a few other quirky characters.
Don't worry about memorizing complicated formulas or chemical jargon. We're focusing on the vibe, the overall personality of these structures. It's all about observation and a little bit of fun.
So, grab your imaginary magnifying glass, and let's dive in. We might even discover some surprising connections to things you already love, like your morning coffee or that sweet, sweet dessert.
Meet the Family!
Our first star of the show is the humble hemiacetal. Imagine a cheerful little carbon atom. It's got a friendly oxygen atom attached, which is holding onto a hydrogen atom – like a little dangling charm.
But wait, there's more! This same carbon atom is also holding hands with another oxygen atom. This second oxygen atom is part of a bigger, longer chain, like a cozy scarf wrapped around it.
So, a hemiacetal is basically a carbon atom with three special friends: a hydroxyl group (that's the oxygen with the hydrogen charm), an oxygen that's part of a longer chain, and usually another carbon atom holding on tight. It’s like a hug that’s not quite complete.
Think of it as the "almost there" stage. It's got most of what it needs, but it's still got a little room for change. It's like the anticipation before a really good surprise!
Now, let's talk about what makes them so special. These hemiacetals are often found in sugars. Yes, those delicious things that give you energy and make your taste buds sing!
When sugars are in a solution, like in water, they often decide to curl up into a ring. And guess what forms in that process? You guessed it – a hemiacetal! It’s like the sugar molecules giving themselves a little molecular hug.
Heads up! Keep an eye out for that single oxygen with a hydrogen dangling, connected to a carbon that’s also linked to another oxygen in a longer chain. That’s your hemiacetal signal!
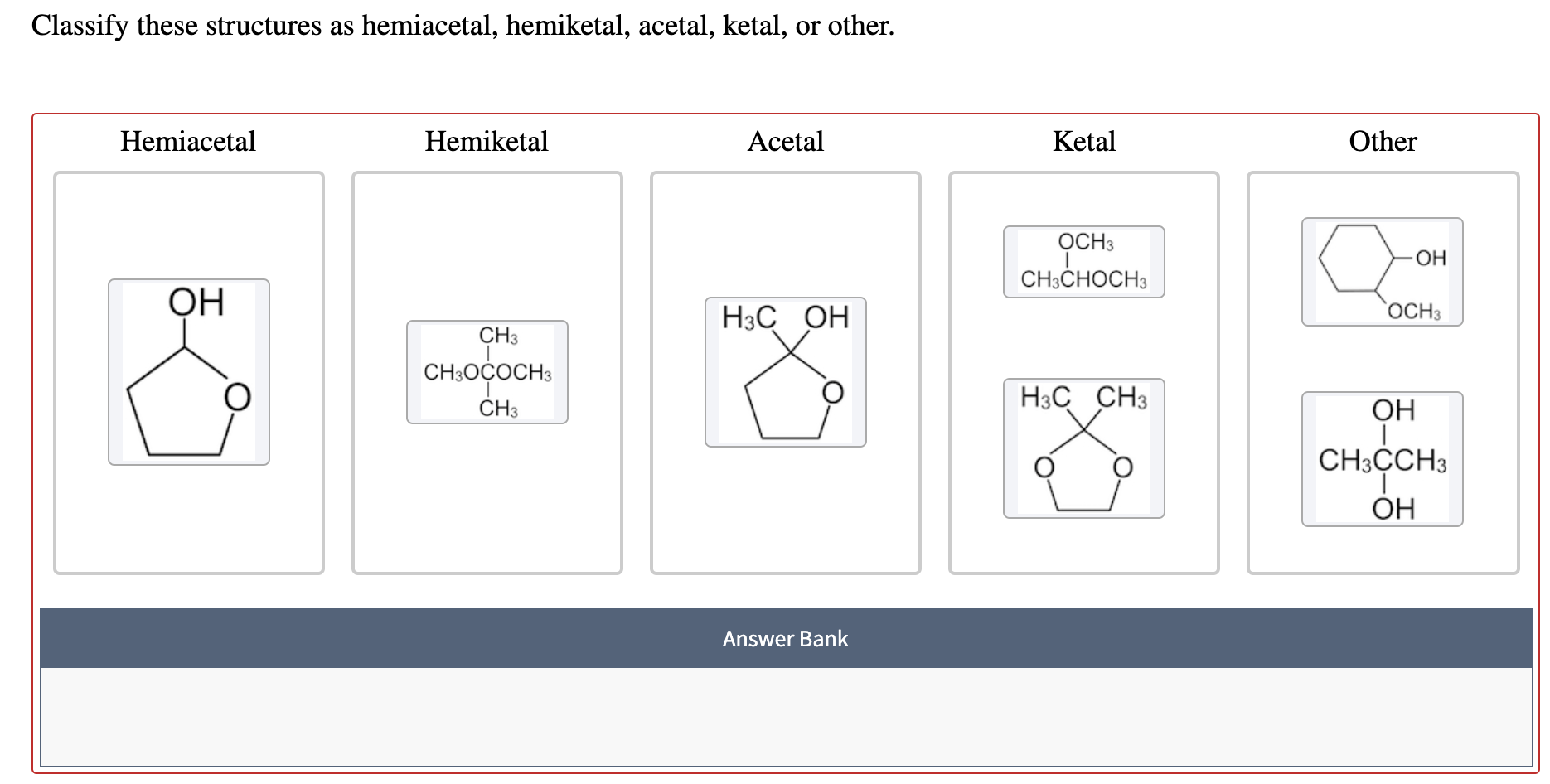
The Perfectly Formed Acetal
Now, meet the acetal. This one is the polished, finished product of our previous friend. The hemiacetal was a little shy, holding onto its hydrogen charm.
The acetal has moved on from that shy phase. It’s let go of that hydrogen charm completely. Instead, it's now holding hands with two other oxygen atoms, both of which are part of longer chains.
So, a carbon atom forms an acetal when it's connected to two oxygen atoms, and each of those oxygen atoms is part of a longer chain. It’s a double hug of molecular bonding!
This makes the acetal a bit more stable, a bit more settled. It's like a perfectly brewed cup of tea – it's reached its ideal state.
Why should you care about these stable molecules? Well, acetals are like the dependable guardians of sweetness. They're often found in sugars that are not in solution, or when they've decided to pair up with other sugar molecules.
Think of it as a sugar molecule that’s decided to settle down and get serious. It’s no longer in that "just about to react" phase. It’s ready for the long haul.
And here’s a fun fact: many of the molecules that give fruits their delightful sweetness are in this acetal form. So, next time you bite into a juicy apple, you're enjoying the perfectly formed acetal! It's a little piece of molecular perfection.
Spotting Tip: Look for a carbon atom connected to two different oxygen atoms, and make sure each of those oxygens is attached to something else, forming a longer chain. No dangling hydrogen charms here! That’s your acetal in action.
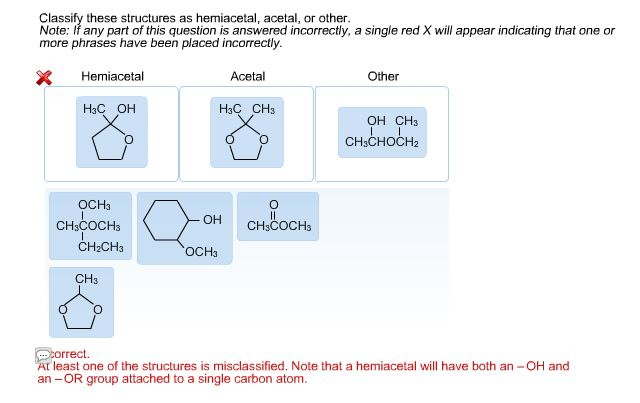
The "Other" Gang: A Quirky Bunch
Of course, the world of chemistry isn't all about perfect hemiacetals and acetals. We have a whole cast of other characters, and they can be just as fascinating, if not more so!
Sometimes, you'll see molecules that look almost like a hemiacetal or an acetal, but with a little twist. Maybe the carbon atom is holding onto something else entirely, like a chlorine atom or a nitrogen atom.
These are our "other" molecules. They're not conforming to the strict rules of hemiacetal or acetal, and that's perfectly okay! They're the rebels, the artists of the molecular world.
For instance, you might encounter a molecule where a carbon atom is bonded to an oxygen (like in a hemiacetal or acetal), but instead of another oxygen in a chain, it's bonded to a simple hydrogen atom directly. This might seem minor, but it changes the whole character of the molecule.
Or, you could see a carbon atom bonded to three oxygen atoms. That's definitely not fitting our hemiacetal or acetal mold! These are unique structures with their own special properties and reactions.
These "other" structures often have very specific jobs. They might be involved in making flavors, or they could be part of more complex biological processes. They’re the supporting actors who make the whole show work.
Don't underestimate these guys! They might not have the catchy names of their more famous cousins, but they are essential. They add variety and complexity to the chemical symphony.
The "Other" Rule: If it looks like it's trying to be a hemiacetal or acetal but is missing a key feature or has an extra one, or if the attached atoms are just plain different (like a chlorine instead of an oxygen in a chain), then you’ve likely found an "other"! Embrace the uniqueness!
Putting It All Together: A Sweet Surprise!
So, we've met the shy hemiacetal, the confident acetal, and the wonderfully diverse "other" group. Now, let's see how this plays out in something you might enjoy every day.
Consider glucose, the sugar that fuels your body. When glucose is floating around in water, like in your bloodstream, it likes to curl up into a ring, forming a hemiacetal. It's all about being ready for action, ready to provide energy.
But when two glucose molecules decide to link up, for example, to form a larger sugar like maltose (the sugar in malted milkshakes!), they do so by forming an acetal bond between them. This is a more stable connection, like a strong friendship.
And what about other sugars? Many of them, especially those found in fruits and vegetables, exist in their acetal form when they are stable and ready to be stored or consumed. It's a testament to their well-structured nature.
Even in more complex molecules, you might find segments that resemble these structures, but with slight modifications. These are the "other" types, contributing their unique flair to the overall molecule. They're the flavor enhancers, the silent workhorses.
The beauty is that these seemingly complex structures are just organized atoms doing their thing. They are the fundamental building blocks of so much that we find delicious and essential.
So, the next time you're enjoying a sweet treat or marveling at the wonders of nature, take a moment to appreciate the molecular dance happening within. You might just spot a hemiacetal, an acetal, or one of the intriguing "other" molecules at play. It's a secret world of delicious chemistry waiting to be discovered!
