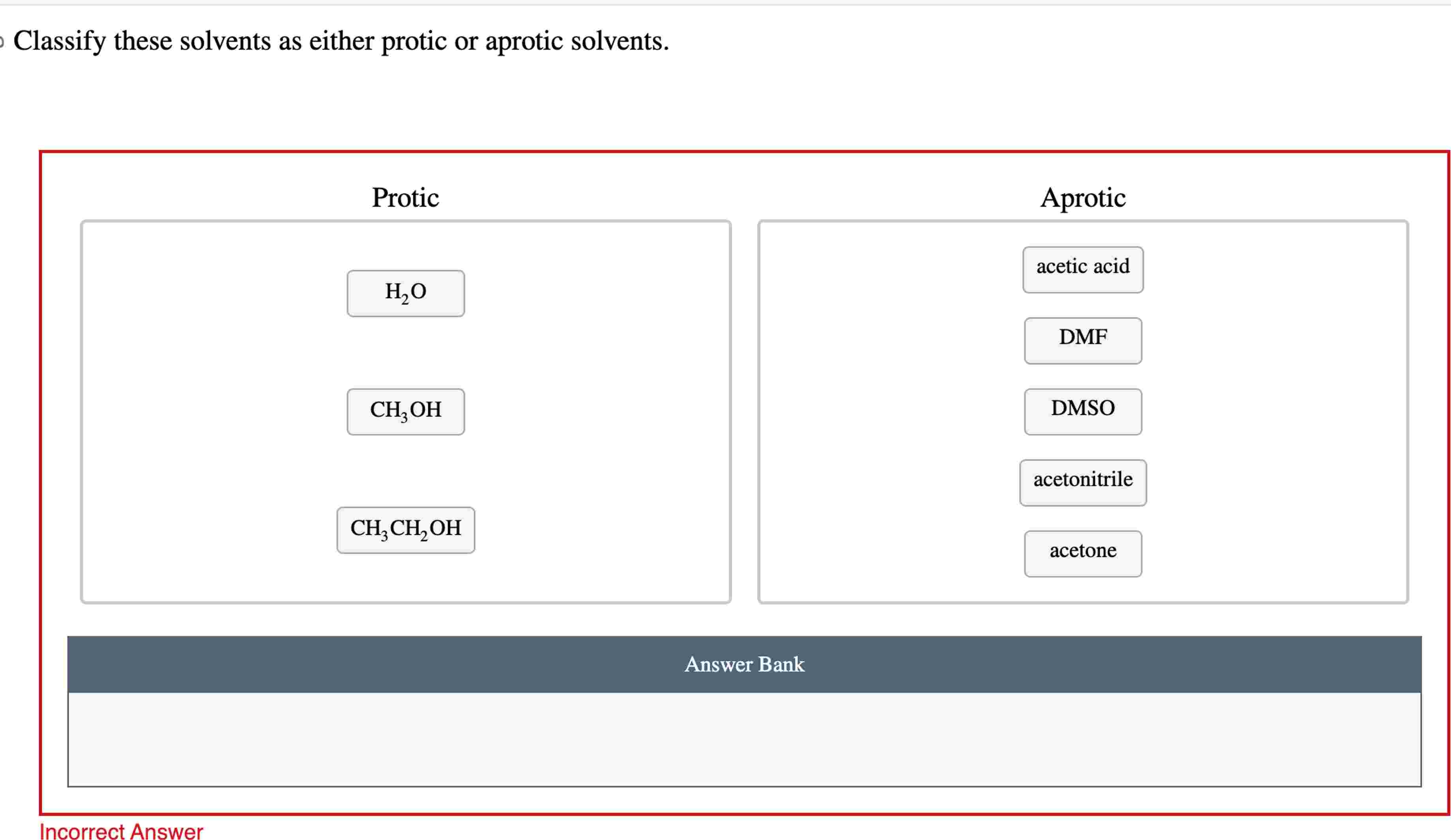
Classify These Solvents As Either Protic Or Aprotic Solvents.
Ever wondered what makes some liquids dissolve things better than others? It's a little bit of chemistry magic, and today we're going to peek behind the curtain at something called protic and aprotic solvents. Don't let the fancy names scare you; think of it like sorting LEGO bricks! It's a fun puzzle that can make your understanding of how things mix and react so much clearer, whether you're tinkering in a home lab, helping with science homework, or just curious about the world around you.
So, why should you care about protic and aprotic solvents? For the beginner chemist or science enthusiast, it's like learning your ABCs. Understanding these solvent types is fundamental to grasping many chemical reactions. For families looking to explore science together, it's a great way to demystify common household items. Think about why nail polish remover works so well, or why oil and water just don't mix! For hobbyists, from those making their own perfumes to crafters working with resins, knowing which solvent to choose can be the difference between a brilliant creation and a sticky mess.
The core idea is simple: it all boils down to whether the solvent has a hydrogen atom attached to an oxygen or nitrogen atom. If it does, we call it protic. Think of it like a tiny hydrogen "proton" that's eager to get involved in things! Water is the classic protic solvent – it’s everywhere and does so much. Alcohols, like ethanol (the kind in hand sanitizer) and methanol, are also protic. They’re great at dissolving polar substances because their hydrogen can form strong interactions.
On the flip side, aprotic solvents don't have that special hydrogen. They’re still great at dissolving things, but they do it in a slightly different way, usually by being good at dissolving non-polar substances or by supporting reactions without getting directly involved in the same way protic solvents do. Examples of aprotic solvents include acetone (the main ingredient in most nail polish removers!), dimethyl sulfoxide (DMSO), and tetrahydrofuran (THF). You might encounter these in art supplies or in more advanced DIY projects.

Want to get started? It's easier than you think! The simplest way is to look at the labels on products you use. Can you identify any of the solvents we mentioned? Water is your easiest protic example. Acetone on a nail polish bottle is aprotic. For a fun experiment, try dissolving a small amount of salt in water (protic) and then in a similar amount of vegetable oil (which is more aprotic/non-polar). See how the salt dissolves in water but not in oil? That's a little hint at solvent power in action!
Classifying solvents as protic or aprotic is a neat way to organize your understanding of chemistry. It’s a practical skill that helps explain why certain things mix and react the way they do. It’s a small piece of knowledge that opens up a bigger understanding of the material world, making everyday science more accessible and, dare we say, a bit more fun!
