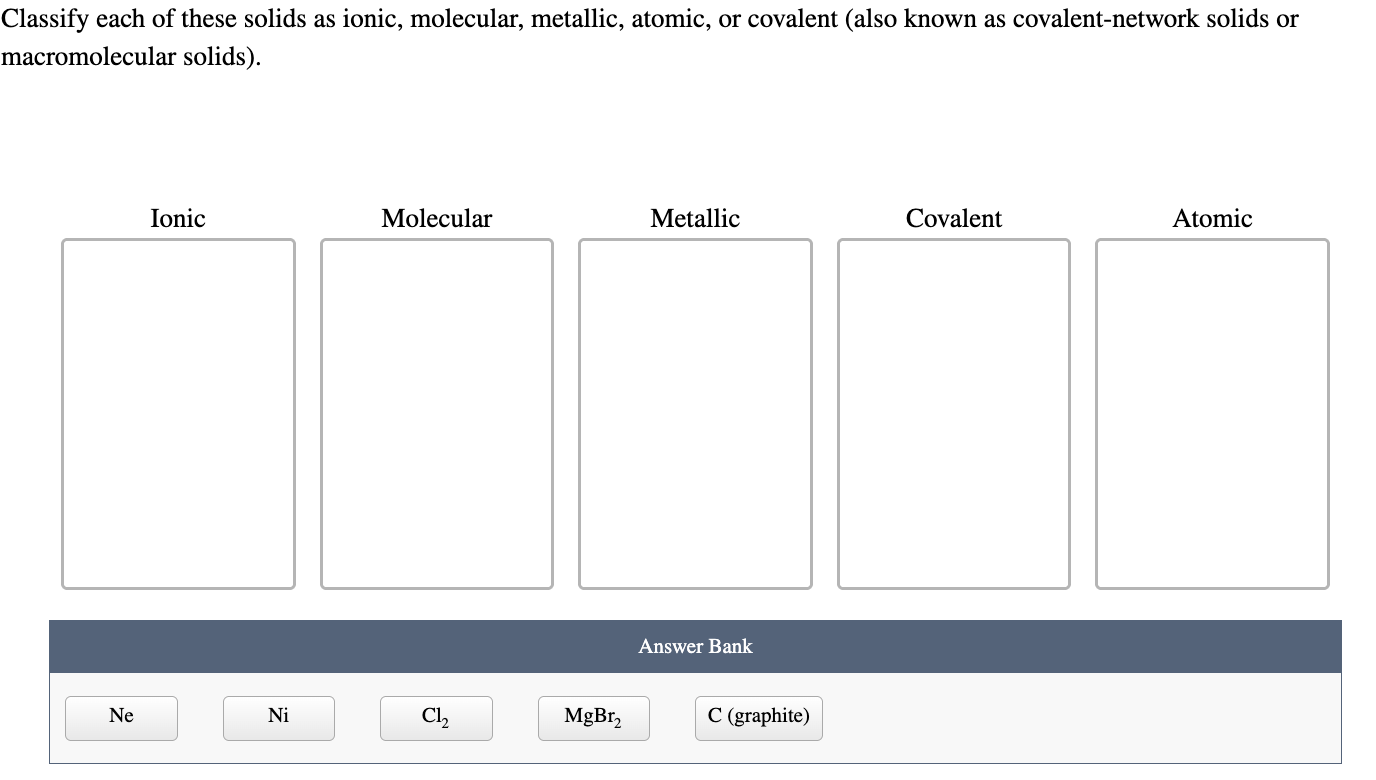
Classify These Solids As Molecular Ionic Or Atomic
Hey there, science explorers! Ever looked at different stuff around you and wondered what makes it tick? Well, get ready for a super fun journey into the world of solids. We're going to classify them, and trust me, it's way more exciting than it sounds.
Think of it like sorting your toys. Some toys are all about playing together as a team, others are like little individual action figures, and some are like building blocks that stick together in a really organized way. Solids are kind of like that!
We've got three main groups to play with: Molecular, Ionic, and Atomic solids. Each one has its own unique personality and way of doing things. It's like a science party, and you're all invited to the VIP section!
Let's dive into the first cool category: Molecular solids. Imagine tiny little LEGO bricks that are molecules. These molecules are made of atoms, but they're already buddies with each other.
In molecular solids, these happy little molecule groups stick together. They're not super strong, mind you. Think of it like a bunch of friends holding hands. It's a gentle connection.
Because the connections aren't super tight, molecular solids are usually pretty soft. You can often scratch them easily. They also tend to melt and boil at lower temperatures.
So, if you've got something that's like a soft block that melts when you leave it in the sun, chances are it's a molecular solid. Think of things like sugar or ice. Yep, that frozen water you love is a molecular solid! Pretty neat, huh?
Now, let's shift gears to our next exciting group: Ionic solids. These guys are the drama queens of the solid world, in the best possible way! They're all about opposites attracting.
In ionic solids, we have atoms that have either gained or lost electrons. This makes them have a positive or negative charge. Think of them as magnets, one positive and one negative.
These charged particles, called ions, get together in a super organized, grid-like structure. They attract each other really strongly, like a super-powered magnet hug. This makes ionic solids quite tough.
Because of these strong attractions, ionic solids usually have high melting and boiling points. They need a lot of energy to break those strong bonds. So, they generally stay solid even when things get pretty warm.
A classic example of an ionic solid is table salt, or sodium chloride. You know, the stuff you put on your fries? That's right, your favorite seasoning is an ionic solid. It's also known for being brittle, meaning it can break if you hit it hard.
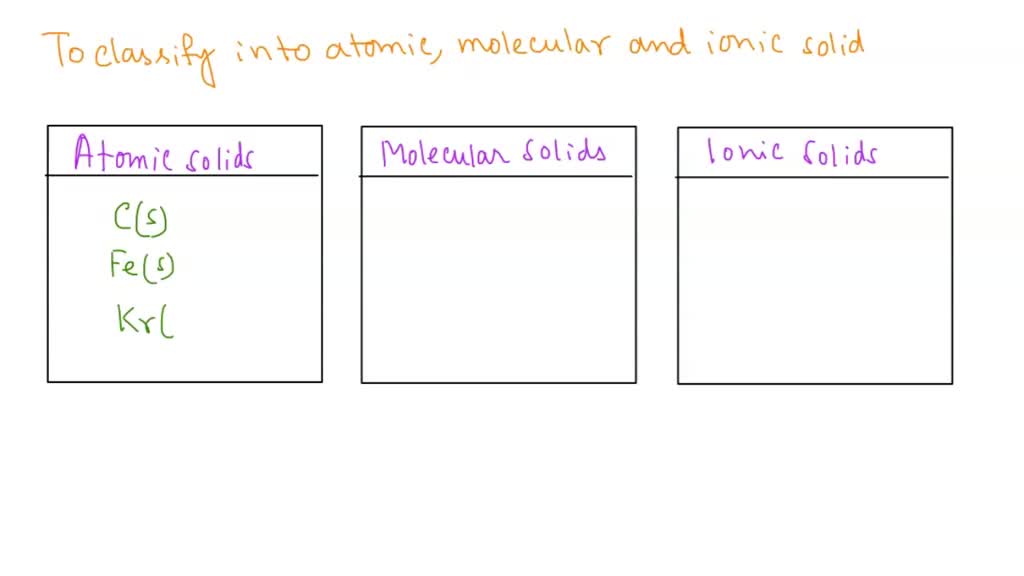
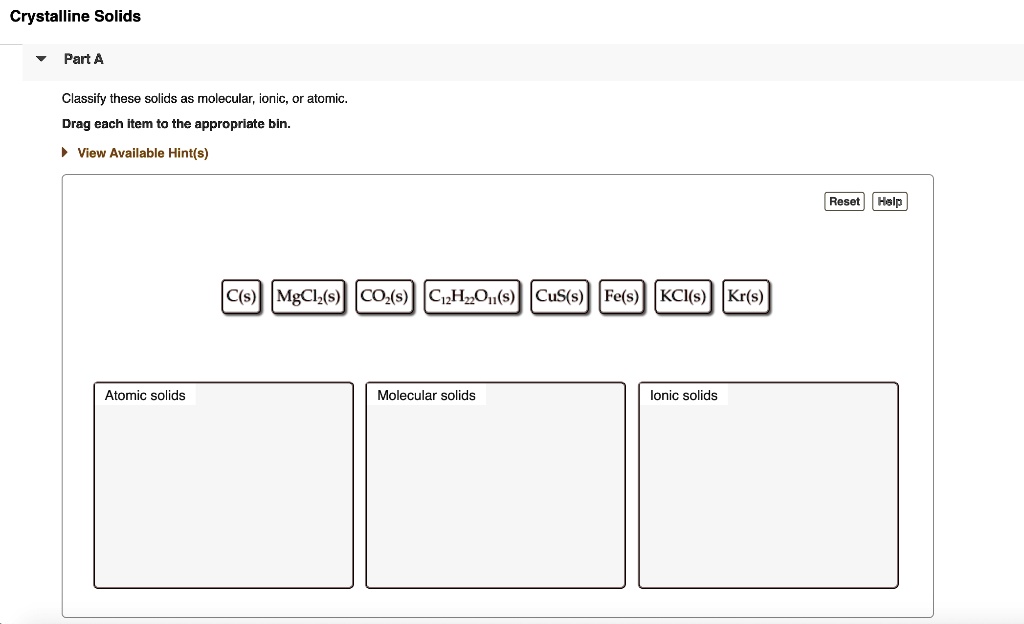
The third and final group for our classification adventure is Atomic solids. These are the ultimate individualists. They're made of atoms that are all by themselves, not really bonded to each other in pairs or groups.
Atomic solids are a bit like a crowd of people at a party, each person doing their own thing. However, these atoms are held together in a structure by very strong forces. It's like they're all standing in a very orderly line, but they're still individual atoms.
There are actually a few sub-types of atomic solids, which makes things even more interesting. It's like having different types of action figures in your collection!
First up in the atomic world are metallic solids. Think of metals like iron, copper, or gold. These are all atomic solids.
![[ANSWERED] Classify each of these solids as ionic, molecular, metallic](https://media.kunduz.com/media/sug-question/raw/59492721-1659707667.890089.jpeg?h=512)
In metallic solids, the atoms are arranged in a lattice, and their outer electrons are free to move around. This is what gives metals their awesome properties, like being able to conduct electricity and heat. It's like a tiny, organized highway for electrons!
Metals are also usually shiny and can be bent and shaped without breaking. That's why we use them to build cars, jewelry, and pretty much everything cool. They’re strong and versatile!
Next, we have covalent network solids. These are the super-tough guys. They're made of atoms bonded together in a continuous network of strong covalent bonds.
Imagine a giant, interconnected web of atoms. That's a covalent network solid! These bonds are incredibly strong, making these solids extremely hard and giving them very high melting points.
Think of diamond, the hardest naturally occurring substance on Earth. Yep, that's a covalent network solid. Or even sand, which is mostly silicon dioxide. These are the ultimate powerhouses.
Finally, we have non-bonding atomic solids. These are a bit rarer and are usually gases at room temperature. But when they do solidify, they're held together by very weak forces.
![[ANSWERED] Classify each of these solids as ionic, molecular, metallic](https://media.kunduz.com/media/sug-question/raw/57388010-1659272951.765628.jpeg?h=512)
The noble gases, like helium and neon, fall into this category when they're in their solid form. They're so content being by themselves that they barely interact. It's like they're just chilling in their own little space.
So, why is classifying these solids so entertaining? Because it's like being a detective! You get to look at a substance and figure out its secret identity based on its properties.
Is it soft and melts easily? It's probably molecular. Is it hard, brittle, and melts at a high temperature? Look out for ionic! Does it conduct electricity, is it shiny, or is it incredibly hard? It might be an atomic solid in disguise.
What makes this so special is that it helps us understand the world around us on a fundamental level. It's not just about memorizing names; it's about understanding the "why" behind everything.
It's like unlocking a secret code that explains why your ice melts, why your salt is crystalline, and why diamonds are so precious and strong. Each classification tells a story about how atoms and molecules are interacting.
And the best part? You can start spotting these solids everywhere! Next time you're in the kitchen, at the beach, or looking at jewelry, you can be the one thinking, "Aha! I know what kind of solid you are!"
It’s a little bit of science magic that you can use every day. It sparks curiosity and makes you appreciate the amazing complexity hidden within even the simplest materials.
So, are you ready to put on your detective hat? The world of solids is waiting for you to explore and classify them. It's an adventure that's both educational and incredibly fun.
Don't be shy! Grab a piece of chalk, a sugar cube, or even just a metal spoon. Start observing. Start questioning. You might be surprised at what you discover.
This classification is the first step to understanding how these materials behave and why they are useful for different purposes. It’s the foundation of material science!
So, go forth and classify! Have fun with it. Embrace the scientific curiosity. You'll be amazed at how much more interesting the solid stuff around you becomes.
It’s a game of observation and deduction, and everyone can play. So, what are you waiting for? Let the classification games begin! Happy solid sorting!
So remember, the next time you encounter a solid, think about its bonds. Are they weak and friendly like in molecular solids? Strong and oppositely charged like in ionic solids? Or are the atoms marching to their own beat, but in an orderly fashion, like in atomic solids?
Each type has its own super-powers and its own weaknesses. It’s a fascinating peek into the building blocks of our universe.
And who knows, maybe this little bit of science fun will inspire you to explore even more. The world of chemistry is full of amazing discoveries just waiting to be made by curious minds like yours.
