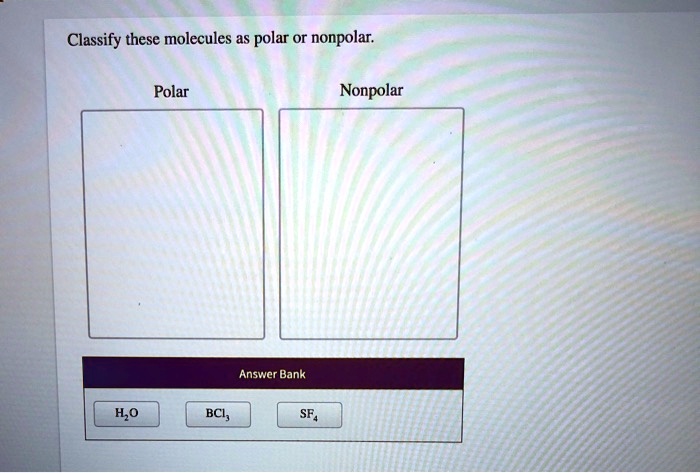
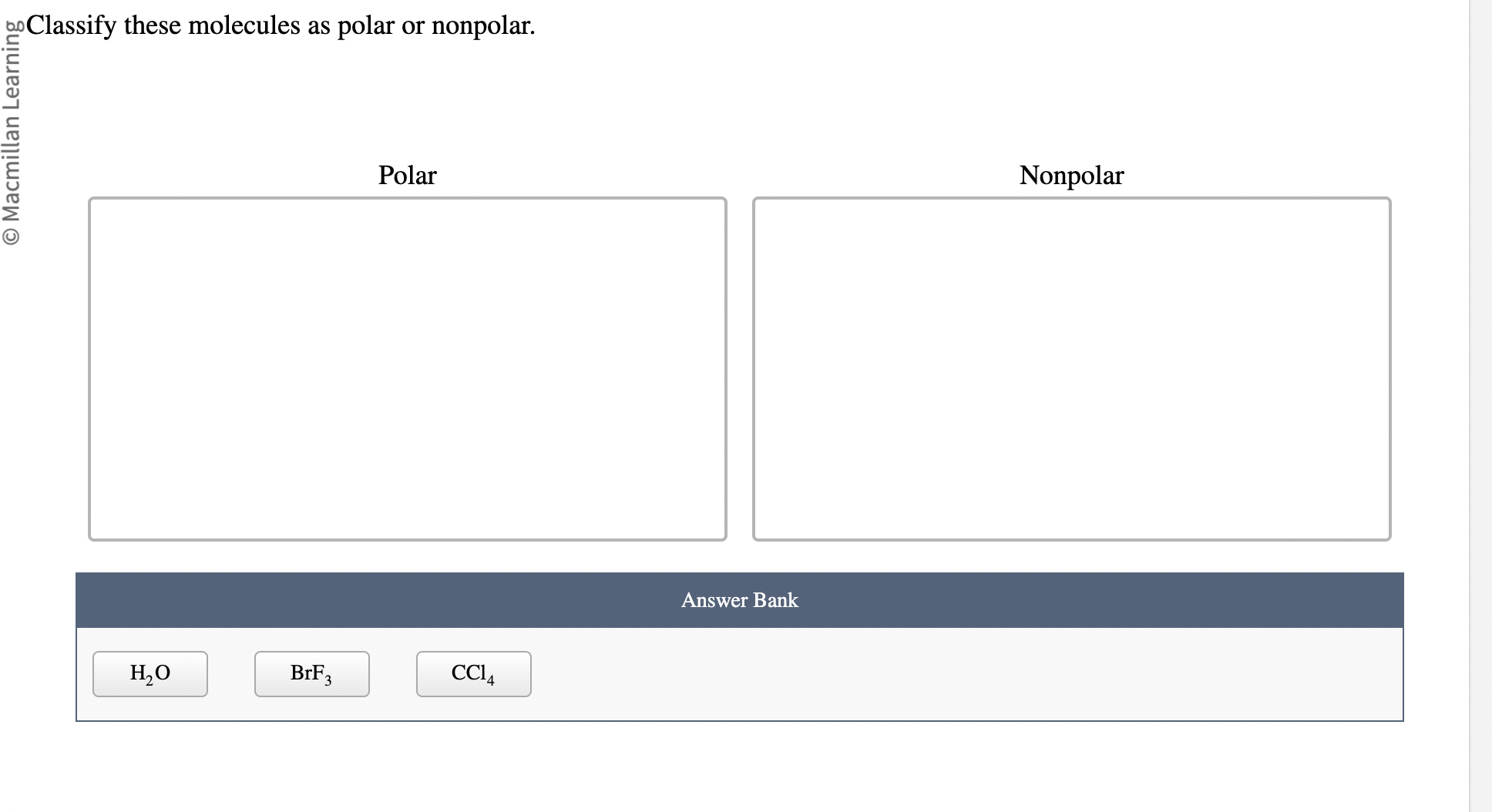
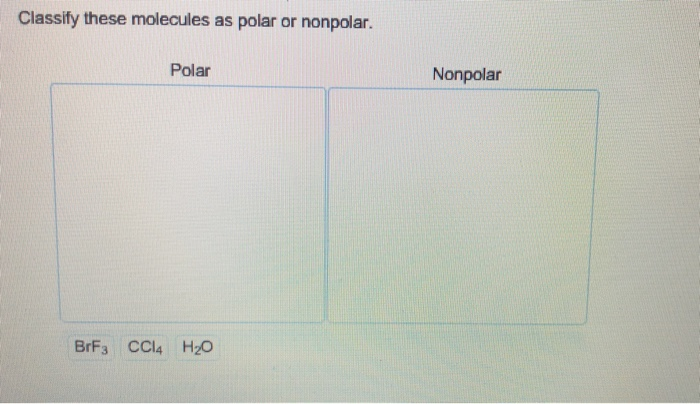
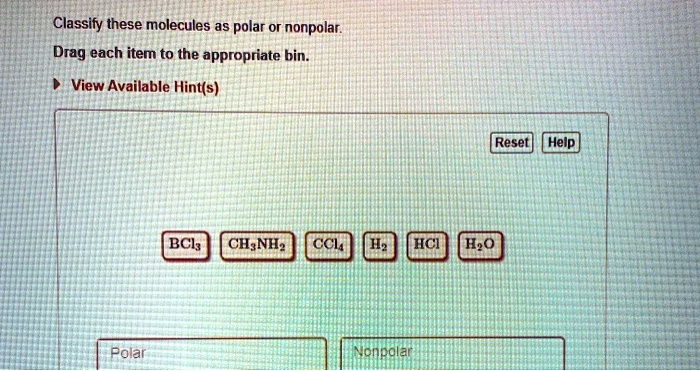
Classify These Molecules As Polar Or Nonpolar.
Hey there, science explorers! Ever looked at a molecule and wondered, "Is this little guy a diva or a chill dude?" Well, buckle up, because we're about to dive into the fascinating world of molecular polarity. Think of it like this: some molecules are super dramatic, pulling electrons this way and that, while others are just happy to go with the flow. It’s all about whether they’re polar or nonpolar, and understanding this can unlock some seriously cool chemistry secrets. Don't worry, we'll keep it light and breezy, no need for a lab coat and goggles unless you really want to!
So, what’s the big deal with polar and nonpolar anyway? Imagine a tug-of-war. In a nonpolar molecule, it's like everyone’s pulling with equal strength. The electrons are shared perfectly, no drama, no favorites. Everything is balanced and symmetrical. On the other hand, a polar molecule is like a tug-of-war where one side is just a little bit stronger. They hog the electrons, creating a slight charge difference. One end of the molecule becomes a bit negative, and the other end becomes a bit positive. Think of it like a tiny, invisible battery!
This whole electron-sharing business comes down to something called electronegativity. Don't let the fancy word scare you. It's basically just an atom's attraction for electrons. Some atoms are electron-magnets, while others are more laid-back. When atoms bond together to form a molecule, their electronegativity differences play a huge role in determining if the bond, and subsequently the molecule, will be polar or nonpolar.
Let’s start with the chill guys: nonpolar molecules. These are your laid-back, go-with-the-flow types. They either have identical atoms bonded together, or the atoms have very similar electronegativities. In these cases, the electrons are shared so evenly, there’s no permanent charge separation. It's like a perfectly balanced seesaw. Nobody's dipping lower than the other!
A super common example of a nonpolar molecule is H2 (hydrogen gas). It's literally just two hydrogen atoms hugging each other. Since both hydrogen atoms are identical, they have the same electronegativity. Their tug-of-war is a tie! So, the electrons are shared perfectly. No poles, no fuss, just happy hydrogen.
Another classic nonpolar is O2 (oxygen gas). Same deal here, two identical oxygen atoms. They're best buds, sharing electrons equally. Imagine them holding hands and spinning around. It's a beautiful, symmetrical dance.
What about molecules with different atoms? Well, it depends on how similar their electron-hogging tendencies are. Take methane (CH4). This is a big one in the chemistry world! It has a carbon atom in the center and four hydrogen atoms surrounding it. Now, carbon is a little more electronegative than hydrogen. So, in the C-H bonds, the electrons are pulled slightly towards the carbon. This sounds polar, right? Ah, but here's where geometry comes into play. Methane has a beautiful, symmetrical tetrahedral shape. Think of a tripod with a ball on top. All the hydrogen atoms are equally spaced around the carbon. Even though each C-H bond has a slight polarity, because of this perfect symmetry, the polarities of the four bonds cancel each other out. It's like having four people pushing a box in perfectly opposite directions – the box doesn't move!
So, even if individual bonds have a bit of a charge difference, if the molecule's overall shape is symmetrical, the polarities can cancel out, making the whole molecule nonpolar. This is a super important concept. We call this the molecular geometry or molecular shape. It’s like the blueprint of the molecule, and it dictates how those electron pulls actually add up.
Now, let’s talk about the divas: polar molecules. These are the ones with a bit of a personality! They have a significant difference in electronegativity between the atoms, and their shape isn't perfectly symmetrical. This means the electrons are pulled more towards one atom, creating a partial negative charge (often shown as δ-) on that end, and a partial positive charge (δ+) on the other end. Voila! You've got a dipole – a molecule with two poles.
The most famous polar molecule in the universe (or at least in our everyday lives) is water (H2O). You’ve probably heard of it! Water is made of one oxygen atom and two hydrogen atoms. Oxygen is a big electron-magnet. It's way more electronegative than hydrogen. So, in the O-H bonds, the electrons are pulled strongly towards the oxygen. This makes the oxygen end of the water molecule slightly negative.
But here's the twist: water isn't a straight line. It's bent! The two hydrogen atoms are attached to the oxygen at an angle. Imagine a Mickey Mouse head – the oxygen is the head, and the two hydrogens are the ears. Because of this bent shape, the partial negative charge on the oxygen and the partial positive charges on the hydrogens don't cancel out. The whole molecule has a distinct positive end (where the hydrogens are) and a negative end (where the oxygen is). This makes water a super strong polar molecule, and it’s why it’s such a fantastic solvent for so many things!
Think about why oil and water don't mix. It's all about polarity! Polar molecules like water tend to stick together because of their opposite charges. Nonpolar molecules like oil also stick together, but for different reasons (we’ll get to that, maybe). Because water is polar and oil is nonpolar, they just can't find common ground. They’re like oil and water, you know? 😉
Another good example of a polar molecule is ammonia (NH3). Similar to water, nitrogen is quite electronegative. It pulls the electrons from the three hydrogen atoms towards itself. Ammonia has a pyramidal shape, kind of like a little three-legged stool. Again, this shape isn't symmetrical enough to cancel out the polarity of the N-H bonds. The nitrogen end is partially negative, and the hydrogen ends are partially positive. So, ammonia is a polar molecule.

What about diatomic molecules where the atoms are different? Like hydrogen chloride (HCl). We have hydrogen and chlorine. Chlorine is much more electronegative than hydrogen. So, the electrons in the H-Cl bond are pulled way over to the chlorine side. This creates a significant partial negative charge on chlorine and a partial positive charge on hydrogen. Since it's a simple two-atom molecule, there's no fancy geometry to cancel things out. HCl is definitely a polar molecule.
Let's think about some slightly more complex ones. Take carbon dioxide (CO2). This one is a bit of a trickster! Carbon dioxide has a carbon atom in the middle and two oxygen atoms on either side, like O=C=O. Now, oxygen is more electronegative than carbon, so each C=O bond is polar, with the electrons being pulled towards the oxygen atoms. So, you have a partial negative charge on each oxygen and a partial positive charge on the carbon. HOWEVER, carbon dioxide is a linear molecule. The two oxygen atoms are on opposite sides of the carbon, like this: O---C---O. The polarities of the two C=O bonds are pointing in exactly opposite directions and are equal in magnitude. Because of this perfect linear symmetry, the polarities cancel each other out. So, even though the individual bonds are polar, the CO2 molecule as a whole is nonpolar. Mind-bending, right? It's all about the overall shape!
Okay, so how do we figure this out? Here’s the super-simplified, no-sweat checklist:
The Super-Easy Polarity Checklist:
Step 1: Look at the atoms in the molecule. Are they the same? If yes, usually it's nonpolar (unless there are some weird lone pairs that break symmetry, but let's keep it simple for now!).
Step 2: If the atoms are different, check their electronegativity. Is there a big difference? If yes, the bonds are likely polar.
Step 3: Consider the molecular geometry (shape). Is the molecule symmetrical? Think of it like this: if you could put it in the middle of a perfectly balanced scale, would it tip? If the polar bonds are arranged symmetrically, they cancel out, and the molecule is nonpolar. If the polar bonds are not arranged symmetrically, or if there's a strong electronegativity difference and no cancellation, then the molecule is polar.
Let's try a few quick classifications. Imagine:
N2 (Nitrogen gas): Two identical nitrogen atoms. Same electronegativity. Nonpolar. Easy peasy!
HF (Hydrogen Fluoride): Hydrogen and Fluorine. Fluorine is a super electronegativity hog! Big difference. Linear molecule. Polar. Definitely!
CCl4 (Carbon Tetrachloride): Carbon and Chlorine. Chlorine is more electronegative than carbon. But, CCl4 has a tetrahedral shape. All the C-Cl bonds are pulling electrons towards the chlorine atoms, but in a perfectly symmetrical way. The polarities cancel out. Nonpolar! See, shape is a big deal!
SO2 (Sulfur Dioxide): Sulfur and Oxygen. Oxygen is more electronegative than sulfur. The molecule has a bent shape. This bent shape means the polar S-O bonds don't cancel out. Polar! You can think of it as having a slightly negative oxygen side and a slightly positive sulfur side.
CH4 (Methane): We already talked about this one. Tetrahedral, symmetrical. Nonpolar, despite slightly polar C-H bonds.
NH3 (Ammonia): We did this too. Pyramidal, not symmetrical enough to cancel. Polar.
Why does all of this even matter, you ask? Well, the polarity of a molecule dictates a LOT about its behavior. Polar molecules interact differently with other molecules than nonpolar ones do. This affects things like:
- Solubility: "Like dissolves like." Polar solvents (like water) dissolve polar solutes. Nonpolar solvents (like hexane) dissolve nonpolar solutes. This is why you can wash your hands with soap (which has polar and nonpolar parts) to get rid of greasy, nonpolar dirt!
- Boiling and Melting Points: Polar molecules often have stronger intermolecular forces (attractions between molecules) because of their charges, leading to higher boiling and melting points.
- Biological Systems: So much of life is based on the interactions of polar and nonpolar molecules. Cell membranes, for example, have parts that are polar and parts that are nonpolar, creating barriers that control what gets in and out.
So, next time you see a chemical formula, don't just see letters and numbers. See a tiny world with its own personalities, its own little dramas and harmonies. Some are the life of the party, attracting all sorts of other charged molecules, while others are happy in their own little club, only hanging out with their own kind.
The amazing thing is, this simple concept of polarity underpins so much of the complex beauty we see in the world, from the way plants grow to how our bodies function. You've just unlocked a secret code to understanding the molecular dance happening all around us, all the time. Give yourself a pat on the back – you're officially a molecule detective, and the mysteries of chemistry are now a little less mysterious, and a whole lot more fun!
