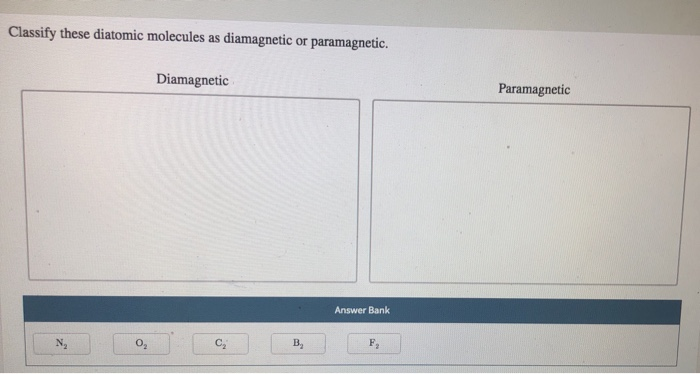
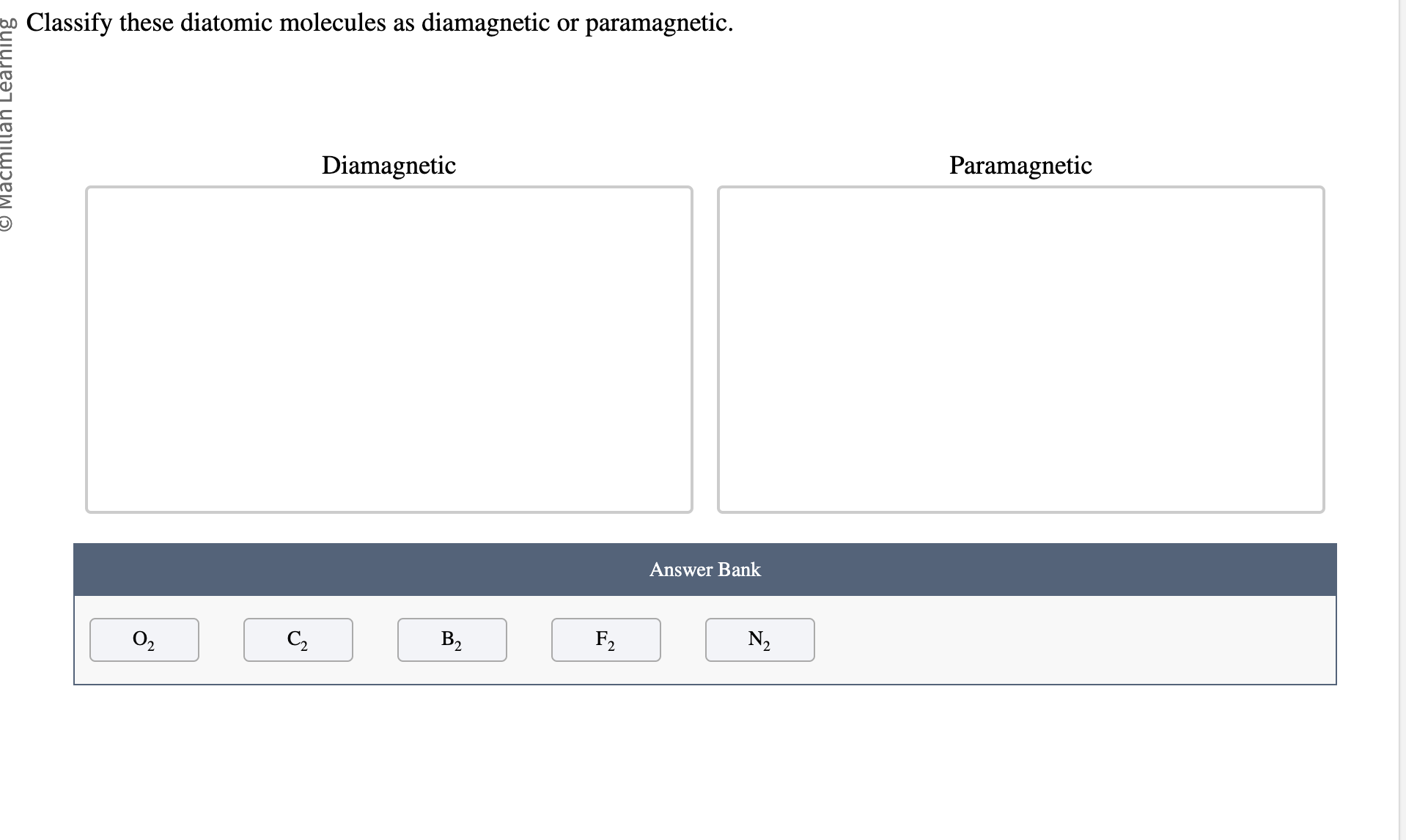
Classify These Diatomic Molecules As Diamagnetic Or Paramagnetic.
Hey there, science adventurers! Ever look at something super tiny, like a molecule, and wonder if it's got a secret magnetic personality? Well, buckle up, because today we're diving into the wonderfully weird world of diatomic molecules and their magnetic leanings. Think of them as tiny elemental buddies who like to hang out in pairs!
These little duos are everywhere, forming the very air we breathe and the water we drink. But when it comes to magnetism, some are total show-offs, while others are perfectly content to blend in. It's all about the way their electrons are behaving, and trust me, it's more exciting than a cat chasing a laser pointer!
We're going to explore two main magnetic personalities: diamagnetic and paramagnetic. Imagine these as two distinct clubs in the molecular world. One club is super chill and doesn't really care about magnets, while the other gets all excited and dances around when a magnet is nearby. It's like the difference between a sloth and a hyperactive puppy!
The Chill Squad: Diamagnetic Wonders!
First up, let's talk about the diamagnetic molecules. These guys are the introverts of the magnetic universe. They're perfectly happy with their electrons all paired up, like cozy couples on a couch. When you bring a magnet near them, they barely flinch. They might even give a tiny, polite push away. It's like they're saying, "Thanks, but no thanks, I'm perfectly comfortable here."
Think of them as the people who politely decline every party invitation, preferring a quiet night in with a good book. They have a complete set of paired electrons, which means all their electron "slots" are filled with two electrons spinning in opposite directions. This perfect pairing cancels out any magnetic effect. They are the epitome of balance and order!
One of our superstar diamagnetic duos is dihydrogen, also known as H₂. This is basically two hydrogen atoms giving each other a high-five and forming a stable bond. Each hydrogen atom brings one electron, and when they pair up, voilà! A perfect, matched set. No unpaired electrons means no magnetic drama. They're just vibing.
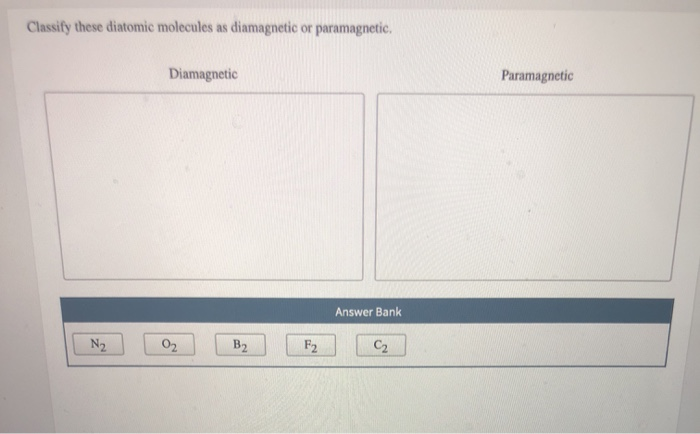
Another excellent example is dinitrogen, N₂. This is the mighty nitrogen gas that makes up a huge chunk of our atmosphere. N₂ has a triple bond, which is like a super-strong handshake between the two nitrogen atoms. All their electrons are neatly paired up, making N₂ totally indifferent to magnetic fields. It's like a perfectly organized sock drawer – no strays allowed!
Then we have difluorine, F₂. Fluorine is a bit of a dramatic element on its own, but when two of them team up, they form a stable, diamagnetic molecule. All their electrons find their perfect partners, and the F₂ molecule cruises through life, unaffected by external magnetic forces. It's like they've achieved a state of electron zen.
And don't forget dicarbon, C₂. This one's a bit of a rare bird in its elemental form, but it's a fantastic example of a diatomic molecule. C₂ has a quadruple bond (yes, really!), which means it's incredibly stable. All its electrons are paired up in this tightly knit structure, making it decidedly diamagnetic. It’s electron harmony at its finest!
So, if you encounter a diatomic molecule where all the electrons are playing nice and paired up, you can bet your bottom dollar it's diamagnetic. They are the quiet achievers, the steady Eddies of the molecular world. They might not be flashy, but they're essential!

The Energetic Ones: Paramagnetic Mavericks!
Now, let's switch gears and talk about the exciting crowd: the paramagnetic molecules! These guys are the life of the molecular party. They've got at least one electron that's flying solo, a lone wolf looking for a magnetic cuddle. When you bring a magnet near them, they get super excited and are attracted to it. It's like a magnet is their favorite toy, and they can't help but play!
Imagine these molecules as the enthusiastic friends who will always say "yes" to an adventure. They have one or more unpaired electrons, which gives them this magnetic "personality." These unpaired electrons are like tiny magnets themselves, and they line up with an external magnetic field, pulling the molecule towards it. They're basically saying, "Ooh, shiny magnet! I wanna get closer!"
Our first star on the paramagnetic stage is dioxygen, O₂. Yep, the very oxygen we breathe! This is where things get really interesting. Oxygen molecules are surprisingly paramagnetic. They have two unpaired electrons, which means they're constantly looking for a magnetic friend. This is why oxygen is so crucial for many biological processes – it’s involved in energetic exchanges!
Think of it like this: O₂ molecules are like little magnets with their north and south poles sticking out, ready to grab onto anything magnetic. This characteristic is actually super important for how oxygen behaves in our bodies and in the environment. It’s a molecule with a magnetic pulse!
Another fun example is mononitrogen monoxide, also known as NO. This is a bit of a tricky one because it’s not made of identical atoms, but it’s still a diatomic molecule. NO has an odd number of electrons, which means it's practically guaranteed to have at least one unpaired electron. Hence, it’s wonderfully paramagnetic. It's always looking for that magnetic connection!
Then we have monoboron, B₂. This diatomic molecule is a bit of a rebel. In its gaseous state, B₂ is actually paramagnetic! It has unpaired electrons that make it attracted to magnetic fields. It’s another example of how electron configuration dictates magnetic behavior, and B₂ definitely has some unpaired electrons doing their thing.
Let's consider monochlorine, Cl₂. Oops, scratch that! Cl₂ is actually diamagnetic. My apologies, sometimes even the science enthusiasts get a little flustered! But if we did have a diatomic molecule with unpaired electrons, it would be paramagnetic. The key is always those lone electrons.

The rule of thumb for paramagnetic molecules is simple: if you find even one unpaired electron, you've got a magnetic enthusiast on your hands! These molecules are often involved in reactions where they readily interact with other substances, partly due to their magnetic attraction. They're the dynamic duos, always ready for action!
The Grand Finale: Putting It All Together!
So, there you have it! The secret lives of diatomic molecules revealed through their magnetic personalities. It's all about how their electrons are arranged. Are they all happily paired up, making them diamagnetic and indifferent to magnets? Or do they have a few rogue electrons, making them paramagnetic and drawn to magnetic fields?
It’s a fascinating peek into the microscopic world, where even the tiniest particles have their own unique characteristics. So next time you’re breathing in air (mostly N₂ and O₂!), remember that one of those molecules is a chill introvert (N₂) and the other is an enthusiastic magnet-lover (O₂)! Isn't science just the coolest?
Keep exploring, keep wondering, and remember that even the smallest things can have the biggest personalities. Happy molecule-watching!
