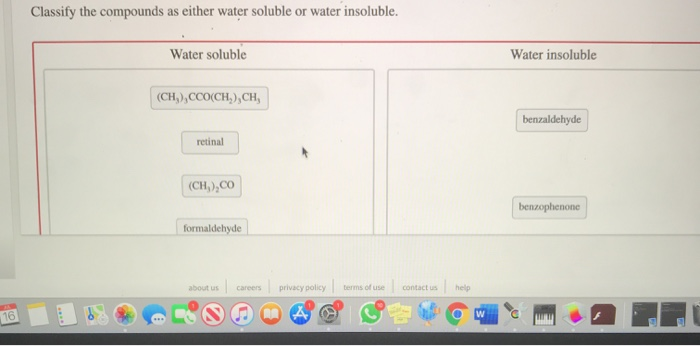
Classify These Compounds As Soluble Or Insoluble.
Hey there, science enthusiasts (and those who just stumbled in looking for a little sparkle)! Ever looked at a recipe and wondered why some things just mix and others just… well, don't? Or maybe you've tried to dissolve a stubborn sugar cube in iced coffee and ended up with a sugary surprise at the bottom? We've all been there, right? Well, get ready, because we're about to dive into a topic that sounds a bit… chemical, but I promise you, it's a whole lot of fun and can seriously up your kitchen game and even your understanding of the world around you. We're talking about solubility! And today, we're going to play a little game: classifying compounds as either soluble or insoluble.
Now, before you picture dusty textbooks and complicated formulas (don't worry, I’m not going to unleash any of those on you today!), let's break down what "soluble" and "insoluble" actually mean in a way that makes sense for our everyday lives. Think of it like making friends. Some people just click instantly, right? They mingle, they chat, they become part of the same lively group. That's like our soluble compounds – they readily mix with a solvent (usually water in our context, because, well, water is everywhere and it's pretty darn awesome) and disappear into it, creating a uniform solution. Easy peasy!
Then you have those folks who are a bit more… independent. They prefer to do their own thing, maybe they're a little shy, or just have a different vibe. They don't really blend in with the crowd. These are our insoluble compounds. When you try to mix them with water, they stubbornly refuse to dissolve. They might float around, or sink to the bottom, creating distinct little clumps or layers. Think of oil and water – no matter how much you shake it, they’re not going to become best buddies.
Why Should You Care About This Whole Soluble/Insoluble Shenanigan?
You might be thinking, "Okay, interesting, but how does this make my life more fun?" Oh, my friends, the possibilities are endless! Let’s start with the kitchen, a place where solubility reigns supreme. Ever made Jell-O? That's all about dissolving gelatin in hot water. What about that perfectly clear cup of tea or coffee? The flavors and colors have dissolved into the water, creating that delightful beverage. Without solubility, our morning brew would be a whole lot less… brewy.
And what about baking? When you cream butter and sugar, you’re not really dissolving the sugar, but you’re getting it distributed really well. But think about making frosting – you’re dissolving sugar in a liquid, and the magic happens! Even making a simple salt and pepper shaker is a lesson in solubility. Salt? Usually soluble. Pepper? Much less so. See? You’re a scientist every time you season your food!
But it’s not just about yummy treats. Think about cleaning! Soap is a fantastic example of a compound that loves to interact with both water and grease. That’s why it works so well to get rid of dirt and grime. It's a clever little molecule that bridges the gap between things that normally wouldn't mix. Pretty cool, right?
Let's Play a Little Game: Soluble or Insoluble?
Alright, are you ready to put on your imaginary lab coat? Let's try classifying some common compounds. Remember, for our general purposes, we're mostly thinking about their behavior in water. It's like a popularity contest for molecules!
The "Absolutely, Let's Be Friends!" Club (Soluble)
These guys are the life of the party. They’re eager to mingle and make new solvent friends. Here are some common members:

- Table Salt (Sodium Chloride, NaCl): Yup, the stuff you sprinkle on your fries. Dissolves in water like a charm. It’s practically begging to be dissolved.
- Sugar (Sucrose, C12H22O11): Another kitchen staple! Your tea, your coffee, your cookies – all thanks to sugar being super soluble in water. It’s the sweet deal everyone wants.
- Vinegar (Acetic Acid, CH3COOH): That tangy stuff you put on salads. It's mostly water with a bit of acetic acid, and they happily mix. No drama here.
- Rubbing Alcohol (Isopropyl Alcohol, C3H8O): If you’ve ever disinfected a cut, you’ve seen this in action. It mixes with water really well. It’s a cooperative compound.
These are your go-to's when you want a smooth, uniform mixture. They’re reliable, they’re easy, and they get the job done with minimal fuss. It’s like having a friend who always agrees to your plans without any hesitation!
The "Nah, I'm Good Doing My Own Thing" Crew (Insoluble)
These compounds are the independent spirits. They’re not saying they’re bad, they just prefer their personal space when it comes to water. They tend to stick together.

- Sand (Silicon Dioxide, SiO2): Ever tried to dissolve sand in water? It just sits there, doesn't it? It’s there for the beach vibe, not for mixing.
- Oil (various types, e.g., Vegetable Oil): The classic example! Oil and water – they just don't mix. You’ll get little oily droplets floating around or sitting on top. They're like two distinct social circles.
- Most Metal Oxides (e.g., Iron(III) Oxide, Fe2O3 - aka Rust): While some metal compounds are soluble, many, like rust, are not. They form solid particles.
- Rocks and Minerals (generally): Most rocks you find on the ground aren’t going to dissolve in your water bottle. They’re built to last and stay solid.
These are the compounds that remind us that not everything is meant to blend seamlessly. Sometimes, separation is the natural order of things. It’s like having a friend who enjoys a quiet evening at home while you’re out at a loud party – different preferences, and that’s perfectly okay!
A Little Science Trick: The "Like Dissolves Like" Rule
Now, for a little peek behind the curtain! There's a super handy saying in chemistry that helps us predict solubility: "Like dissolves like." What does this mean? It means that polar solvents (like water, which has a slightly positive and a slightly negative end) tend to dissolve polar solutes (compounds with similar charge distributions). And nonpolar solvents (like oil, which has no distinct positive or negative ends) tend to dissolve nonpolar solutes.
So, when you see water (polar) and salt (an ionic compound that behaves like a polar solute), they’re “like,” so they dissolve! When you see oil (nonpolar) and water (polar), they’re “unlike,” so they don’t dissolve. It’s a simple concept, but it unlocks a whole world of understanding about why things mix or don’t.

Understanding this simple rule can make you feel like you have a secret superpower. You can start predicting how things might behave, not just in the kitchen, but in nature, in cleaning products, and even in your own body! It’s all about the molecular dance.
The Uplifting Bit: You're a Natural Scientist!
See? You’ve just navigated the fascinating world of soluble and insoluble compounds! You’ve learned about kitchen magic, cleaning secrets, and even a fundamental scientific rule. You’ve done this with your brain and maybe a little imagination, proving that science isn’t just for people in labs. It’s in everything around us, and you are already a participant!
Don't stop here! This is just the tip of the iceberg. The more you look around, the more you'll see these principles at play. So go ahead, be curious! Ask why things happen. Experiment (safely, of course!). You’ve got the smarts, you’ve got the curiosity, and I have a feeling you’re going to have a lot of fun discovering all the amazing scientific wonders that make our world so interesting.
