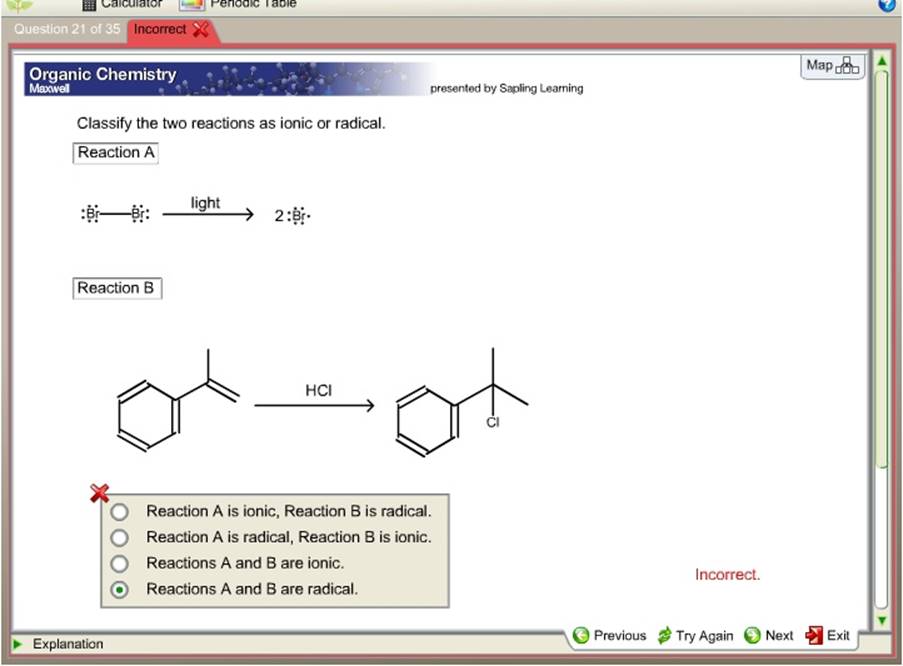
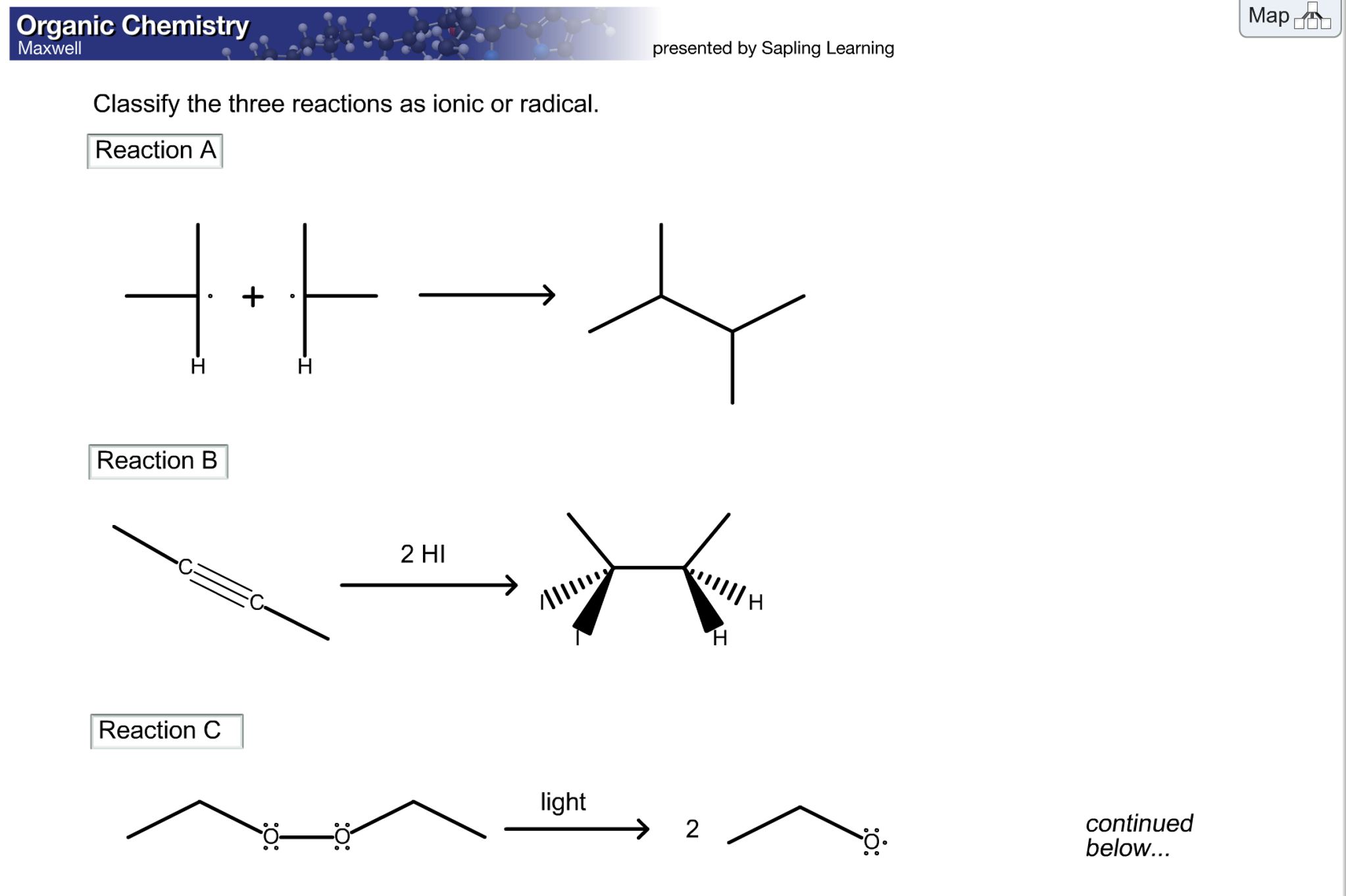
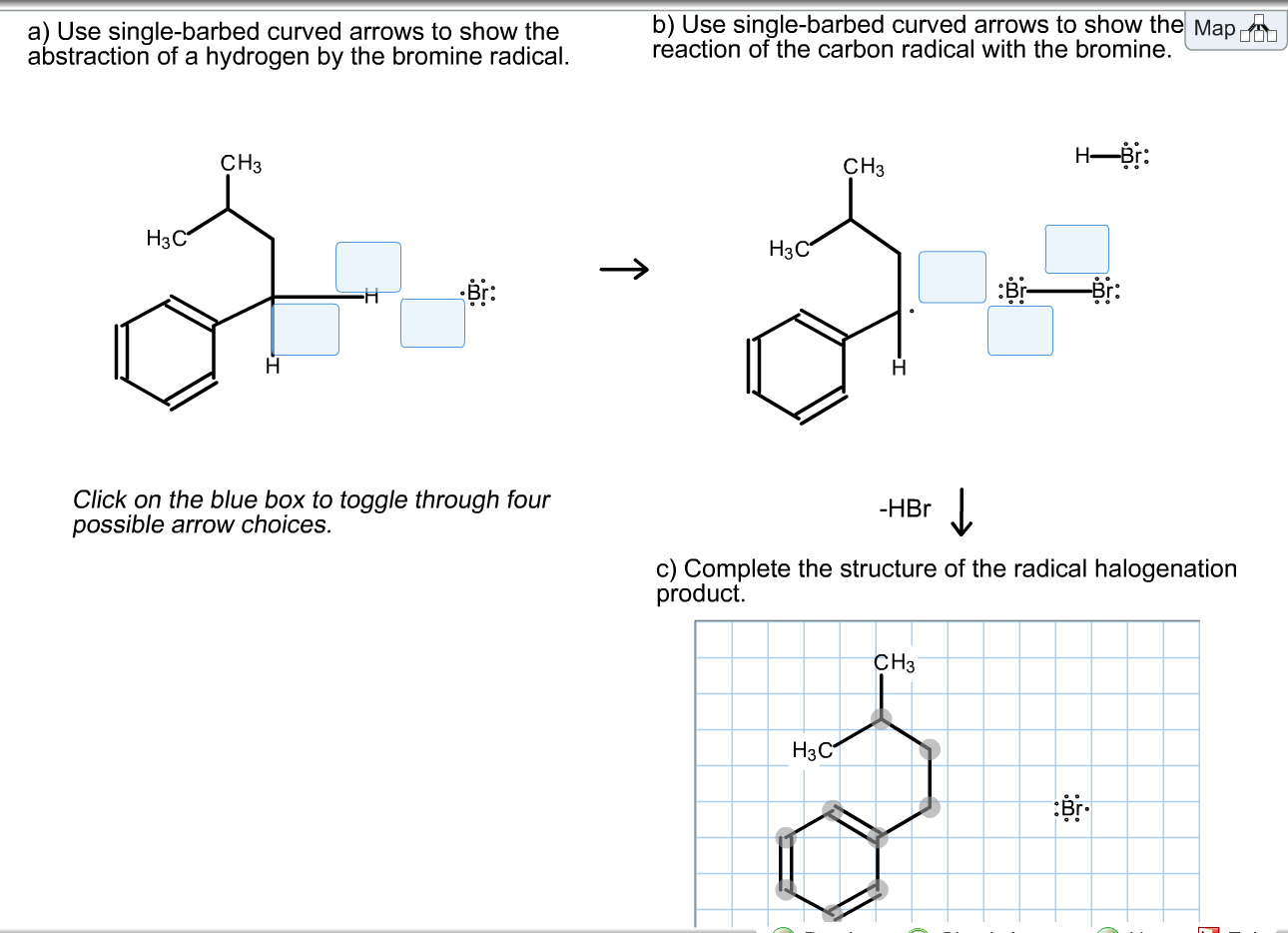
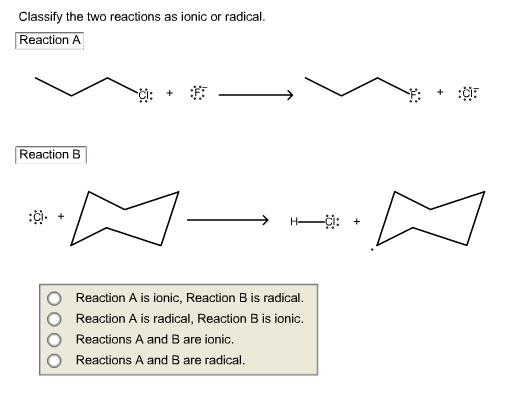
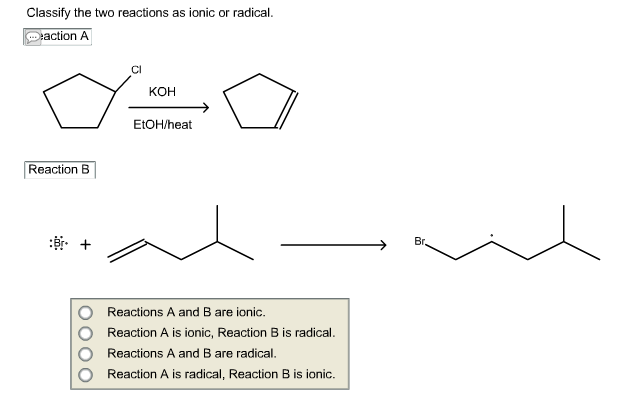
Classify The Two Reactions As Ionic Or Radical
Hey there, chill vibes only! Ever feel like the world of chemistry is a bit, well, intense? Like those old sci-fi movies with dramatic explosions and bubbling beakers? Sometimes, it feels like it’s all about precise measurements and complicated jargon. But what if I told you that even in the nitty-gritty of chemical reactions, there's a certain rhythm, a certain flow that’s not so different from how we navigate our own lives? Today, we’re going to dive into two fundamental ways chemical reactions play out, and classify them as either ionic or radical. Think of it as understanding different communication styles – some are direct and clear, others are a bit more… spontaneous and free-wheeling.
This isn't about acing a final exam (unless that's your jam, in which case, go you!). This is about appreciating the underlying principles that make things happen, from the way your avocado ripens to the magic of a perfectly baked cookie. We’re going to break it down, keep it light, and hopefully, add a splash of fun to your understanding of the universe’s tiny dances.
The Direct Approach: Ionic Reactions, All About the Ions!
Let’s start with what we’ll call the "Ionic" way of doing things. Imagine you’re at a swanky cocktail party. There are people who are definitely looking for a partner, and they’re not shy about it. They have a clear charge, a distinct purpose. In the chemical world, these are our ions. Ions are atoms or molecules that have gained or lost electrons, giving them a positive or negative electrical charge. Think of them as having a very specific vibe, a strong pull towards opposite charges.
So, an ionic reaction is like a polite, yet determined, handshake between these charged particles. One ion with a positive charge (a cation – remember, cats pawsitively have fur!) is looking for a friend with a negative charge (an anion – think of an annoying negative sibling). They’re drawn to each other like magnets, and when they meet, they readily exchange partners, forming new, stable compounds. It’s all about that electrostatic attraction, that undeniable pull.
Think about table salt, NaCl. That’s a classic example of an ionic compound. Sodium (Na) loses an electron to become a positively charged sodium ion (Na+), and chlorine (Cl) gains that electron to become a negatively charged chloride ion (Cl-). They’re a perfect match, sticking together thanks to their opposite charges. It’s a beautiful, stable relationship, much like a perfectly balanced relationship in real life – lots of give and take, and a strong foundation.
In ionic reactions, the action happens super fast. There’s no hesitation, no second-guessing. It's like when you see your favorite dessert on the menu – you know you want it, and you’re going to get it. The transfer of electrons is immediate, and the resulting ions are quite stable. You won’t find a lot of loose, unpaired electrons hanging around, causing trouble. It’s all very proper and predictable, which, let’s be honest, can be a relief sometimes!
Fun Fact: The term "ion" comes from the Greek word "ienai," meaning "to go." These charged particles are literally on the move, seeking their perfect ionic soulmate!
Practical Tip: Think about how you organize your closet. Ionic reactions are like putting your clothes away neatly in their designated drawers or hangers. Everything has its place, and there’s a clear order. When things are organized, it’s easier to find what you need and maintain harmony, just like in an ionic compound.
The Spontaneous Crew: Radical Reactions, Free Radicals on the Loose!
Now, let’s switch gears and talk about the "Radical" approach. If ionic reactions are the sophisticated cocktail party, radical reactions are more like a spontaneous jam session in a garage. Things are a bit looser, a bit more unpredictable, and definitely more energetic. Here, we’re dealing with free radicals.
What’s a free radical? Imagine an atom or molecule that’s feeling a bit… incomplete. It has an unpaired electron, like someone showing up to a dance with only one shoe. This unpaired electron makes it incredibly reactive, desperate to find a partner to pair up with. It’s like that friend who’s always up for anything, a little bit wild, and can initiate some serious change.
These free radicals are not about gentle exchanges. They’re about stealing an electron from another molecule. This theft then creates a new free radical in the molecule that lost the electron, setting off a chain reaction. It’s like a game of tag, where once you’re tagged, you’re “it” and you have to tag someone else. This chain reaction can continue for a while, leading to significant transformations.
Think about what happens when you leave an apple out on the counter. Over time, it browns. That’s a radical reaction happening! Oxygen in the air, combined with some light and enzymes in the apple, can create free radicals. These free radicals then start to break down the apple’s cells, causing oxidation and that familiar brown color. It’s a gradual but unstoppable process, a cascade of events.
Radical reactions are often initiated by energy, like UV light (hello, sunscreen!), heat, or even other highly reactive molecules. They can be a bit messy, leading to the formation of different products than you might expect from a purely ionic pathway. But they are absolutely crucial for many biological processes, from how our bodies generate energy to how our immune system fights off invaders.
Fun Fact: The term "radical" in chemistry refers to the unpaired electron. In a way, these molecules are "radical" because they’re a bit of an outlier, not fitting neatly into the paired-up world.
Cultural Reference: Think of the punk rock movement. It was often characterized by its rebellious, energetic, and somewhat unpredictable nature. Radical reactions have a similar vibe – they’re the rebels of the chemical world, initiating change in a less conventional way.
Practical Tip: We encounter free radicals all the time in our environment. Antioxidants, found in foods like berries and leafy greens, are our allies in neutralizing free radicals. So, when you’re choosing what to eat, think of it as arming yourself and your body with defense mechanisms against these reactive little rascals!
Putting It All Together: Recognizing the Difference
So, how do we tell the difference between an ionic and a radical reaction? It’s all about the players involved. If you see a reaction involving the transfer of electrons between ions with distinct positive and negative charges, you’re likely looking at an ionic reaction. It's clean, it's direct, it’s all about attraction.
If, however, the reaction involves species with unpaired electrons – those free radicals on the prowl – then you’re witnessing a radical reaction. This is where things get interesting, with chain reactions and a more dynamic, less predictable flow.
One way to think about it is like this: Ionic reactions are like a planned dinner party. You invite specific people, you have a menu, and you expect a certain outcome. Radical reactions are more like a pop-up festival. You don't always know who will show up or exactly what will happen, but there's an undeniable energy and a potential for something truly unique.
Sometimes, reactions can have elements of both. The world of chemistry isn’t always black and white, just like life. But understanding these two core pathways helps us appreciate the diverse ways molecules interact and transform. It’s about recognizing the fundamental forces at play, the underlying mechanisms that drive everything around us.
A Little Reflection: Finding Our Own Reaction Styles
Thinking about ionic and radical reactions can be a neat little metaphor for how we approach life, can’t it? We all have our preferred ways of interacting. Some of us are like ionic reactions: we thrive on clarity, on established structures, on direct communication. We know what we want, and we’re pretty good at getting it through clear, intentional actions. We value stability and predictability.
Then there are those moments when we embrace our inner radical. We’re open to spontaneous adventures, to unexpected turns. We’re adaptable, energetic, and perhaps a little more willing to dive into the unknown. These are the times when we’re more reactive, more prone to initiating change, and sometimes, creating a beautiful kind of organized chaos.
Most of us, of course, are a blend of both. We have our organized, structured moments, and then we have our free-spirited, go-with-the-flow times. Understanding these different "reaction styles," both in chemistry and in ourselves, can help us appreciate the diversity of approaches and the beauty of different ways of being. It’s not about being one or the other, but about recognizing the spectrum and finding our own comfortable rhythm within it. So, the next time you see something change, whether it’s a chemical reaction or a shift in your own plans, take a moment to appreciate the underlying flow. It's all part of the grand, fascinating dance of existence.
