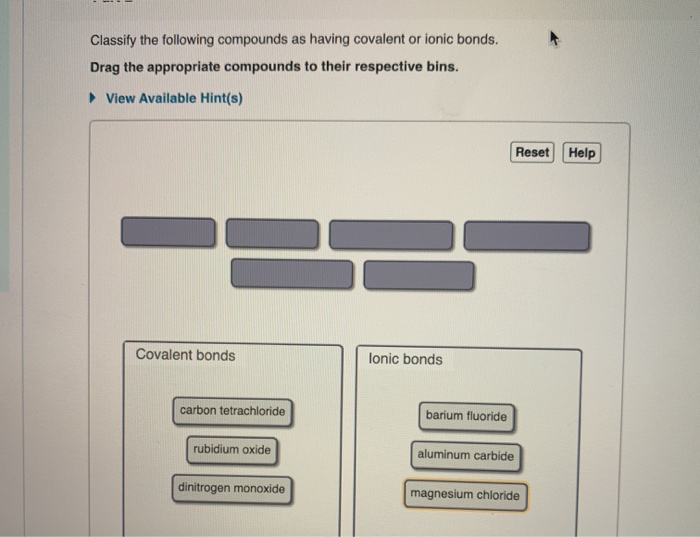
Classify The Following Compounds As Ionic Or Covalent
Hey there, chemistry adventurers! So, you've stumbled upon the wonderful world of classifying compounds, have you? Don't panic! It's not as intimidating as it sounds. Think of it like sorting your socks – you've got your comfy cotton ones, and then you've got those fancy silk ones. They're both socks, but they're pretty different, right? Well, compounds are a bit like that. They generally fall into two main camps: ionic and covalent. And today, we're going to figure out which is which, like super-sleuth chemists!
Let's be honest, sometimes chemistry can feel like trying to decipher an ancient scroll. But we're going to break it down into bite-sized, easy-to-digest pieces. No need for a lab coat, just your trusty brain and maybe a cup of something tasty. We'll make this so simple, you'll be classifying compounds in your sleep… okay, maybe not in your sleep, but you'll definitely feel confident about it!
The Great Divide: Ionic vs. Covalent
So, what's the big deal? What makes a compound ionic or covalent? It all boils down to how the atoms in the compound decide to hang out together. It's like choosing between sharing your toys or hoarding them all for yourself. Each atom has its own little electron posse, and their behavior dictates the type of bond they form.
Think of it this way: atoms are a bit like people at a party. Some are super generous and love to give things away (these are the metals). Others are a bit more possessive and really want to snatch up those goodies (these are the nonmetals). The way they interact at the party determines the vibe, and that vibe is our ionic or covalent bond.
The "Give and Take" Crew: Ionic Compounds
Let's start with the ionic compounds. These guys are all about that give and take. Imagine a metal atom and a nonmetal atom meeting. The metal atom is like, "Hey, I've got an extra electron I don't really need. Want it?" And the nonmetal atom, with its electron-hungry tendencies, is thrilled! "Yes, please! I'll take it!"
So, the metal atom transfers one or more of its electrons to the nonmetal atom. This transfer is key! When the metal atom loses an electron, it becomes positively charged (because it has more protons than electrons now – it's like a positive attitude!). We call this a cation. And when the nonmetal atom gains that electron, it becomes negatively charged (more electrons than protons – a bit grumpy, perhaps?). We call this an anion.
Now, here's the fun part: opposite charges attract! It's like those magnetic personality types. The positive cation and the negative anion are drawn to each other like magnets. This strong electrostatic attraction is what we call an ionic bond. They stick together really, really tightly!
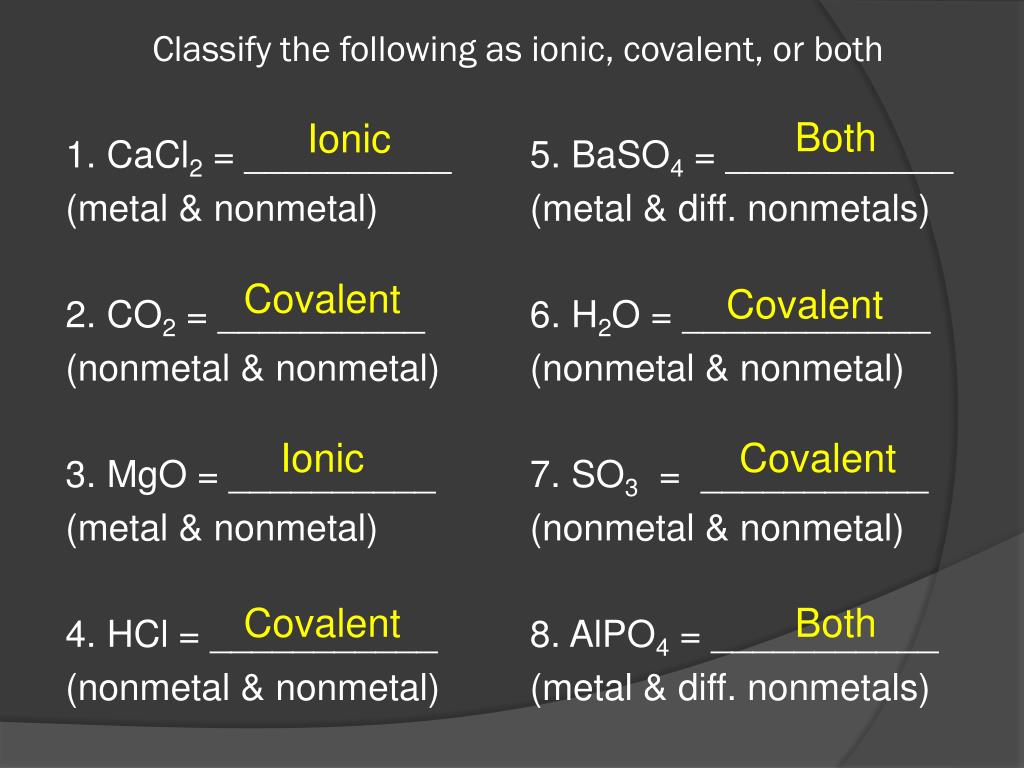
Think about table salt, NaCl (sodium chloride). Sodium (Na) is a metal, and chlorine (Cl) is a nonmetal. Sodium is happy to give away an electron, and chlorine is desperate to snatch one up. Voilà! Sodium chloride is formed. It’s a classic ionic compound. You can almost picture sodium waving goodbye to an electron and chlorine enthusiastically grabbing it.

Key takeaway for ionic compounds: We're talking about a transfer of electrons, usually between a metal and a nonmetal. This creates charged ions that attract each other. Easy peasy, right? It’s like a chemical friendship based on a very generous gift!
So, how do you spot these ionic compounds when you see them? Generally, they're formed from elements from the left side of the periodic table (the metals) and elements from the right side (the nonmetals). Look for a metal cation and a nonmetal anion hanging out together. It's like finding a king and queen ruling their little chemical kingdom.
Also, ionic compounds tend to have some cool properties. They usually form crystals (think of those sparkly salt crystals!), have high melting and boiling points (you can't melt them with a gentle breeze!), and conduct electricity when melted or dissolved in water (because those charged ions are free to zoom around!). It's like they have a built-in electrical circuit.
The "Sharing is Caring" Crew: Covalent Compounds
Now, let's switch gears and talk about the covalent compounds. These guys are the ultimate sharers. They believe in communal living and working together. Instead of one atom completely giving an electron to another, they decide to share their electrons.
Imagine two nonmetal atoms getting together. Neither of them is particularly keen on giving up their electrons completely. They're both a bit like, "Hmm, I like my electrons, but maybe I could use an extra one from you?" So, they compromise! They decide to put their electrons into a shared pool, forming a covalent bond.
This sharing creates a molecule. Think of water (H₂O). Hydrogen (H) and oxygen (O) are both nonmetals. They don't transfer electrons; they share them. The oxygen atom shares some of its electrons with the hydrogen atoms, and the hydrogen atoms share their electrons with the oxygen atom. It’s like a cooperative agreement, a true partnership. They're all holding hands, chemically speaking.
Key takeaway for covalent compounds: We're talking about the sharing of electrons, usually between two or more nonmetals. This forms molecules. It's the "sharing is caring" philosophy in action!
How do you identify covalent compounds? They're typically formed from elements that are all nonmetals. Look for compounds made up of elements from the right side of the periodic table. If you see a bunch of nonmetals huddled together, chances are you've got a covalent compound on your hands. It's like a club where everyone's a member and everyone contributes.
Covalent compounds have different properties than ionic ones. They usually have lower melting and boiling points (a gentle breeze could melt them!), and they don't conduct electricity very well (unless they're dissolved in something that helps them conduct, which is a whole other adventure!). They're more like your everyday, easy-going substances.
Putting Your Detective Skills to the Test!
Alright, enough theory! Let's get our hands dirty (metaphorically, of course – no actual dirt required!). Here are some compounds, and we’re going to classify them. Remember the golden rules: transfer = ionic, sharing = covalent. And the players: metals tend to transfer, nonmetals tend to share or receive.
Compound #1: Potassium Chloride (KCl)
Okay, let's look at KCl. We've got Potassium (K) and Chlorine (Cl). Is Potassium a metal or a nonmetal? Yep, it's a metal (found on the left side of the periodic table). Is Chlorine a metal or a nonmetal? It's a nonmetal (on the right side). So, we have a metal and a nonmetal. What do they do? They transfer electrons! Therefore, KCl is an ionic compound. Potassium gives an electron to chlorine, they become charged, and they're attracted. Boom!
Compound #2: Carbon Dioxide (CO₂)
Next up, CO₂. We have Carbon (C) and Oxygen (O). Are these metals or nonmetals? Both Carbon and Oxygen are nonmetals. So, what happens when two nonmetals meet? They share electrons! They form a molecule by cooperating. Therefore, CO₂ is a covalent compound. It's all about that teamwork!
Compound #3: Sodium Oxide (Na₂O)
Let's tackle Na₂O. We have Sodium (Na) and Oxygen (O). Sodium is a metal, and Oxygen is a nonmetal. Metal + Nonmetal = Transfer of electrons. That means Na₂O is an ionic compound. Sodium is generous with its electrons, and Oxygen is a willing recipient. A classic ionic pairing!
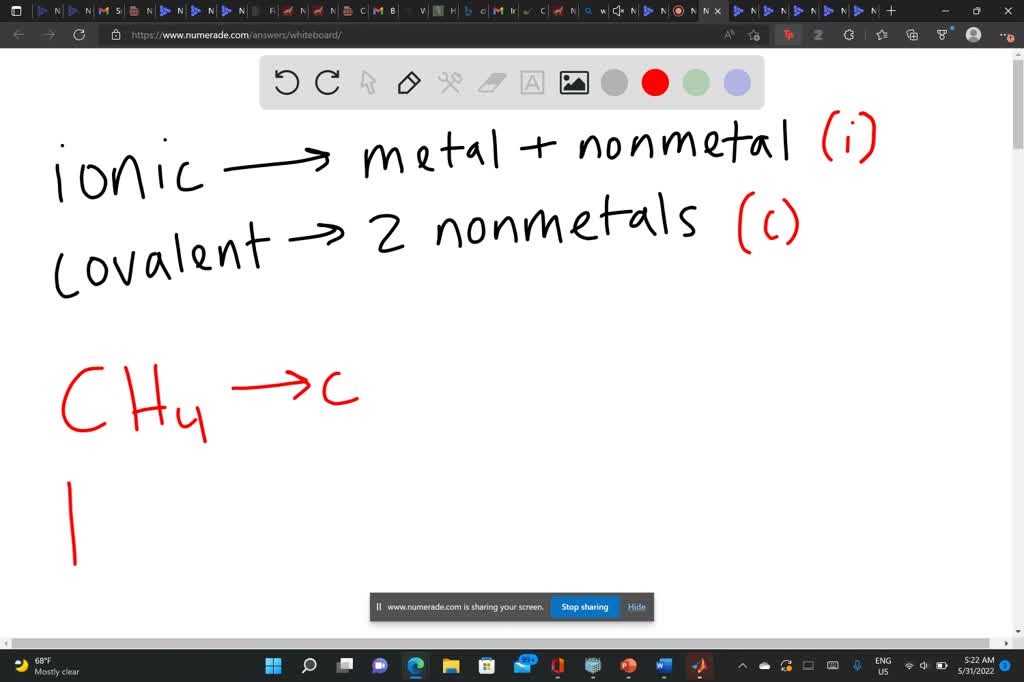
Compound #4: Methane (CH₄)
How about CH₄, which is methane? We've got Carbon (C) and Hydrogen (H). Are these metals or nonmetals? Both are nonmetals. So, what's the deal? They share electrons! They form a molecule. Therefore, CH₄ is a covalent compound. They’re sharing their electron toys!
Compound #5: Calcium Bromide (CaBr₂)
Let's analyze CaBr₂. We have Calcium (Ca) and Bromine (Br). Calcium is a metal, and Bromine is a nonmetal. Metal + Nonmetal = Transfer of electrons. You know the drill! CaBr₂ is an ionic compound. Calcium hands over some electrons, and Bromine happily accepts.
Compound #6: Sulfur Dioxide (SO₂)
Moving on to SO₂. We have Sulfur (S) and Oxygen (O). Are these metals or nonmetals? Both Sulfur and Oxygen are nonmetals. So, what's the outcome? They share electrons to form a molecule. Thus, SO₂ is a covalent compound. Sharing is caring, indeed!

See? It's not rocket science… well, it's chemistry, but you get the idea! Just remember: Metal + Nonmetal = Ionic (transfer). Nonmetal + Nonmetal = Covalent (sharing).
Now, a little word of caution. Sometimes, there are exceptions or more complex situations, like polyatomic ions (groups of atoms that act as a single charged unit). But for the most part, our simple rule of thumb will get you pretty far. Think of it as your trusty compass in the vast ocean of chemical compounds.
There are also compounds that don't fit neatly into these two boxes, but for the fundamental classification, ionic and covalent are your main players. It's like having your main characters in a story – there might be supporting roles, but these two are the stars!
And remember, understanding this is a HUGE step in your chemistry journey. You're not just memorizing; you're understanding why things behave the way they do. You're looking at the fundamental interactions that build the world around us. From the salt on your table to the air you breathe, these basic principles are at play!
So, take a moment and pat yourself on the back. You've just conquered a fundamental concept in chemistry! You’ve gone from wondering to knowing, from confused to confident. That’s pretty awesome, if you ask me. Every new piece of knowledge you gain is like adding a beautiful new color to your mental palette. Keep exploring, keep asking questions, and never let the perceived difficulty of a subject dim your curiosity. The world of science is vast and exciting, and you’re perfectly equipped to explore it!
Go forth and classify with confidence, my friends! You’ve got this!
