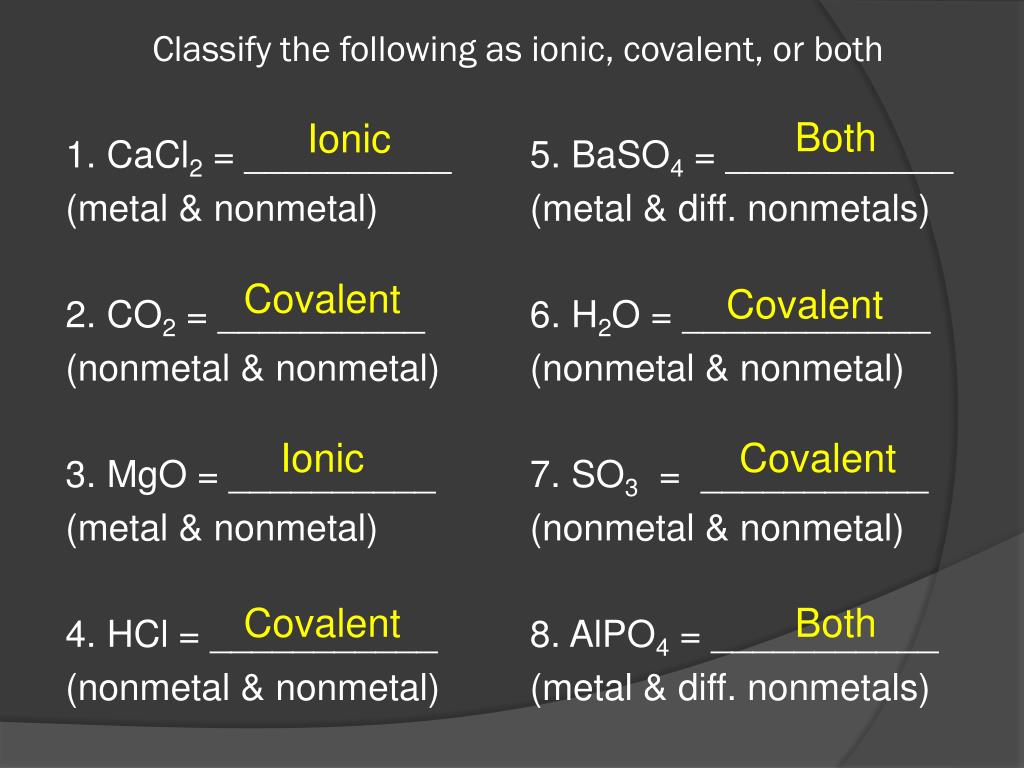
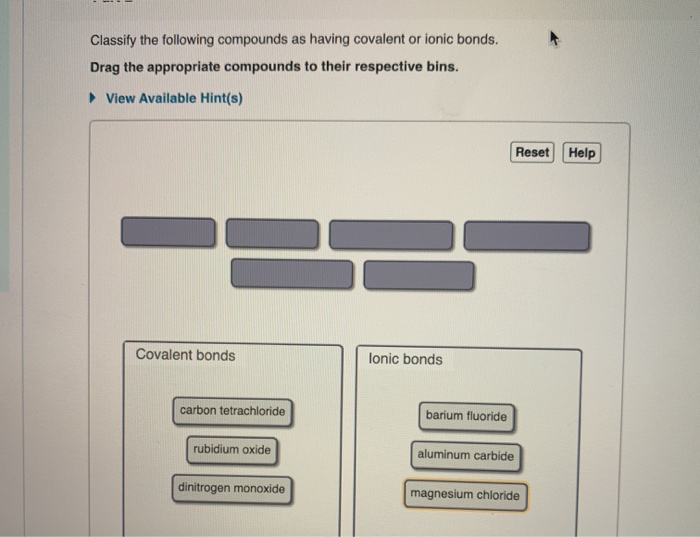
Classify The Following Compounds As Ionic Covalent Or Both
Alright, settle in, grab your latte (or your questionable office coffee, I won't judge), because we're about to dive headfirst into the wild and wacky world of chemical compounds. Forget those stuffy textbooks and mind-numbing lectures. We're talking about classifying these little molecular critters as ionic, covalent, or, in a move that would make Dr. Frankenstein proud, both! Think of it as matchmaking for atoms, but with way more explosions. (Okay, maybe not actual explosions, but sometimes it feels like it when you're trying to remember what a valence electron is.)
So, what's the deal? Why do some compounds just click and form these beautiful partnerships, while others are like awkward teenagers at a school dance, desperately trying to share electrons? It all comes down to how their atoms decide to play nice. We've got two main scenarios here, and then, of course, the wild card. Get ready to meet the stars of our show!
The "You Take It, I'll Take This" Buddies: Ionic Compounds
First up, we have our ionic compounds. These guys are like the divorce lawyers of the chemical world. They don't share; oh no, they transfer. One atom is a bit of a diva, hoarding its electrons, and the other is like, "Fine, you keep 'em, I'll just become a positive ion and we can stare longingly at each other from a distance."
Imagine a super rich atom (usually a metal, think gold-plated everything) and a not-so-rich atom (usually a non-metal, like chlorine, who’s always a bit clingy). The rich one is like, "Here, take this electron! I've got plenty!" The poor one is overjoyed and practically does a victory dance, now sporting a negative charge. The rich one, now missing an electron, is a positively charged ion. And what happens when you have a positive and a negative? Opposites attract, my friends! It's the oldest story in the book, and it applies to atoms too. They hug it out (electrostatically speaking, of course) and form these stable, crystalline structures. Think salt, also known as sodium chloride (NaCl). Sodium, that metal dude, basically tells chlorine, "You want this electron? Take it! I'm going to be Na+ now." Chlorine, happy as a clam, becomes Cl-. Boom! Ionic bond, baby!
These compounds are usually hard, brittle, and have high melting points. They’re not the kind of guys who are going to melt into a puddle at the first sign of heat. They’re built to last, like that stubborn old rocking chair your grandma has. Plus, when they’re dissolved in water, they become little electrical conductors. It’s like they’re secretly running a tiny, chemical rave in your beaker.

The "Let's Share This, It's Awkward Alone" Pals: Covalent Compounds
Now, let’s move on to our covalent compounds. These are the true peacemakers, the hippies of the atomic community. They believe in sharing is caring, especially when it comes to those precious electrons. Instead of one atom saying "Mine!" and the other saying "Fine!", they’re like, "Hey, let’s hold hands and share these electrons. It’s less lonely this way."
These bonds happen between two non-metals. Think oxygen and hydrogen in water (H2O). Oxygen is a bit of a hog, but hydrogen is even smaller and more desperate. So, they decide to get really cozy. The oxygen atom shares one of its electrons with a hydrogen, and the hydrogen shares its one electron back. They’re basically in a permanent, electron-sharing embrace. It’s like a chemical piggyback ride that never ends. This sharing creates a stable molecule. It’s not a free-for-all; they’re sharing in specific ways, forming these distinct little units called molecules.

Covalent compounds are often gases or liquids at room temperature. They’re a bit more laid-back than their ionic cousins. They tend to have lower melting and boiling points. You can’t exactly build a sturdy castle out of water, can you? Unless it’s an ice castle, which, let’s be honest, is just a temporary, albeit beautiful, covalent creation.
And here’s a fun fact: the air you’re breathing right now? Mostly covalent compounds! Nitrogen (N2) and oxygen (O2) are happily sharing electrons, keeping us all alive. So, next time you take a deep breath, thank those electron-sharing ninjas!

The "It's Complicated" Couples: Both Ionic and Covalent!
Ah, but the universe, as we know, is rarely simple. Enter the compounds that are like that couple you know who are constantly fighting and making up, but you just can’t quit them. These are the compounds that have both ionic and covalent characteristics. They're the rebels, the rule-benders!
How does this happen? Well, imagine a compound where you have a metal atom (ready to give up electrons) and a group of non-metal atoms that have already formed a covalent bond amongst themselves. Take sodium sulfate (Na2SO4). You’ve got sodium (Na), that metal dude who’s ready to go full ionic. Then you’ve got the sulfate group (SO4). Within that sulfate group, the oxygen atoms and the sulfur atom are happily sharing electrons, forming covalent bonds. They're like a tight-knit, covalently bonded family.
But then, the sodium ions (Na+) are attracted to this whole sulfate unit. So, the sodium ions form an ionic bond with the negatively charged sulfate group. It’s a bit like a powerful, independent social club (the covalently bonded SO4) that also attracts attention from lone wolves (the Na+ ions). The overall compound is held together by ionic attraction, but inside that social club, there’s a whole lot of covalent camaraderie happening.
These compounds are often found in everyday things like fertilizers and cleaning products. They're the unsung heroes of our domestic lives, managing to be both stable and internally complex. It’s a delicate dance, a chemical tango of sharing and transferring, all happening at the atomic level. Pretty wild, right?
So, the next time you see a chemical formula, don't just glaze over. Think about the personalities of the atoms involved. Are they the transfer-happy divas of ionic bonds? The sharing, caring hippies of covalent bonds? Or are they the complicated, dramatic duos that can't decide if they want to share or transfer? It’s a whole world of atomic drama waiting to be unraveled, one compound at a time!
