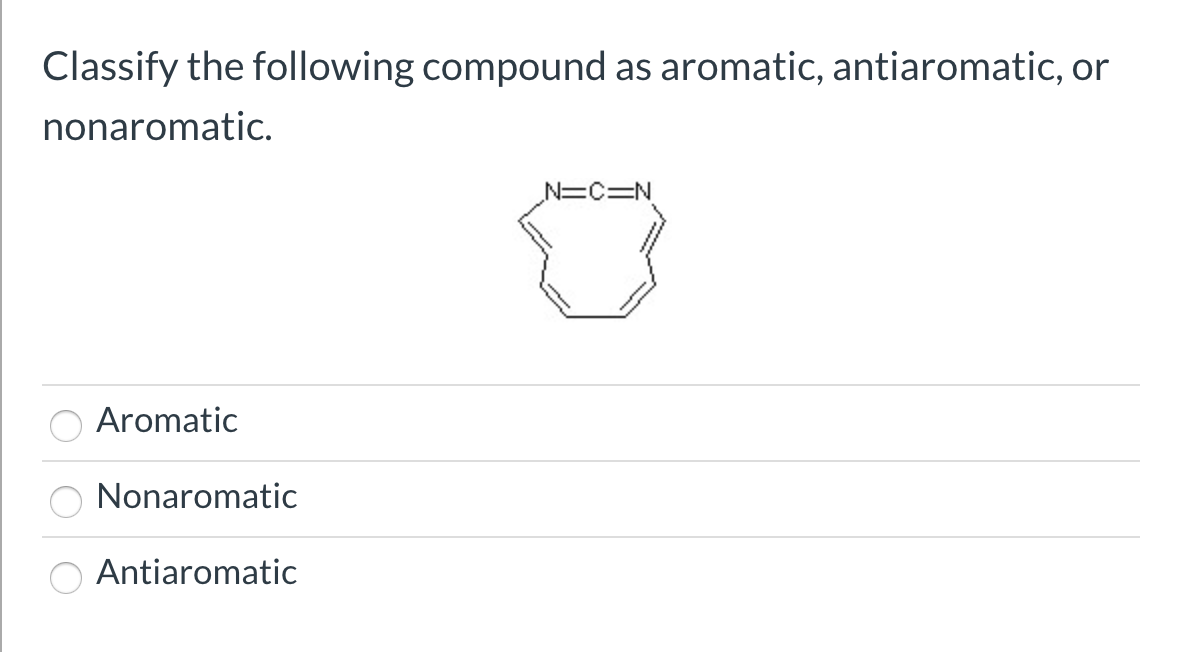
Classify The Following Compound As Aromatic Antiaromatic Or Nonaromatic

Hey there, coffee buddy! So, we're diving into the wild and wonderful world of organic chemistry today, huh? Don't panic! It's not as scary as it sounds. Think of it like trying to figure out if your new friend is the super popular kid everyone flocks to, the one who's kinda intense and maybe a little too exciting, or just, you know, chilling. That's basically what we're doing with these molecules: figuring out their vibe. We're classifying them as aromatic, antiaromatic, or nonaromatic. Easy peasy, right?
Seriously, it's all about a few key things. And once you get them, it's like unlocking a secret code. You'll be spotting these molecular personalities from a mile away. We're talking about structure, electrons, and a little bit of magic. Okay, maybe not magic, but some pretty cool rules that make these compounds behave the way they do.
So, grab another sip of that latte. We're going on a little journey. And by "journey," I mean we're gonna look at some pictures and ask some questions. The best kind of journey, if you ask me.
What's The Deal With Aromaticity Anyway?
Alright, first things first. What even is aromaticity? It's not like smelling a rose, though that's a nice thought. In chemistry terms, aromatic compounds are the rockstars. They're super stable. Like, ridiculously stable. They just don't want to react with anything. They're too cool for school, you know?
Think of benzene. You've probably seen it. It's like the poster child for aromaticity. Six carbons in a ring, alternating double and single bonds. Looks simple, right? But oh, it's so much more than that. It's all about those delocalized electrons. They're not stuck in one place, like a shy kid hiding in the corner. Nope, they're floating around, sharing the love, making the whole molecule happy and stable. It's like a giant, happy dance party happening all over the ring.
And that's the key. This electron dance party is what makes them so darn stable. They've got this special electron configuration that just makes them feel good. So, when you're trying to classify a compound, you're looking for signs of this electron party.
The Holy Trinity of Aromaticity
Now, to be considered a true aromatic king or queen, a compound needs to tick three boxes. Think of them as the membership requirements for the super-exclusive aromatic club. You gotta have all three, or you're out.
Box number one: It needs to be cyclic. So, it's gotta be a ring. No straight lines allowed in this club. Gotta be a closed loop, like a necklace. Easy enough to spot, right? If it's a chain, it's probably not our aromatic friend.
Box number two: It needs to be planar. This means all the atoms in the ring have to lie flat, like a pancake. No lumpy, bumpy rings. They have to be in the same plane so those electrons can do their thing without bumping into anything. Think of it like a perfectly smooth surface for that electron dance party. If the ring is all twisted up, the party gets awkward. And nobody likes an awkward party.
Box number three: This is where it gets a little spicy. It needs to have (4n + 2) pi electrons in its system. Now, don't let the "n" scare you. It just means you need to count the electrons in those double or triple bonds (the ones involved in the ring system, mind you!) and see if the total number fits this magical formula. The most common numbers you'll see are 2, 6, 10, 14... basically numbers that fit that (4n + 2) pattern. If you have 4, 8, 12, or any multiple of 4, well, that's a different story. We'll get to that.
So, cyclic? Check. Planar? Check. (4n + 2) pi electrons? Check. Congratulations, you've found an aromatic compound! Give it a virtual high-five. It deserves it.
What About The Other Guys? Antiaromatic and Nonaromatic
Okay, so we've met the aromatic superstars. But what about the rest of the molecules? They're not all gonna be the life of the party, are they?
The Antiaromatic Antagonists
Let's talk about the antiaromatic compounds. These guys are the opposite of aromatic. If aromatic is stable, then antiaromatic is... well, unstable. Like, really, really unstable. They're the ones who are always getting into trouble, always wanting to react. They're the molecular equivalent of that friend who always suggests doing something slightly dangerous on a whim.
They also have to follow some rules, but they twist them a little. So, they're still cyclic. That's a requirement for both aromatic and antiaromatic. They also have to be planar. Again, same as aromatic. The electrons need that flat surface to do their thing. You can't have a dance party on a bumpy floor, right?
But here's the kicker, the thing that makes them antiaromatic and totally unhappy: they have 4n pi electrons. Not (4n + 2). Just plain old 4n. So, 4, 8, 12, 16... you get the picture. When you have this arrangement of electrons, instead of being stable and delocalized like in aromatics, the electrons get all jumbled up and stressed. They're basically fighting each other. It's like a mosh pit, but way less fun and way more chemically reactive. These compounds are so unstable, they often just rearrange themselves into something else to escape the misery. It's dramatic, honestly.
So, if you see a cyclic, planar molecule with 4 pi electrons in its ring system, give it a wide berth. It's probably not going to end well for you if you try to mess with it. It's the chemical equivalent of a ticking time bomb.
The Nonaromatic Neutrals
And then we have the nonaromatic compounds. These are the chill ones. They're not particularly stable like aromatics, but they're not a chaotic mess like antiaromatics either. They're just... there. They don't have a strong opinion either way. They're the neutral observers of the molecular world.
Why aren't they aromatic or antiaromatic? Well, they usually fail one of the key requirements. The most common reason is that they aren't planar. The ring is all twisted and bent out of shape. It's like trying to hold hands with someone when you're both on different ends of a seesaw. It just doesn't work. The electrons can't delocalize properly. They're kind of stuck where they are, not doing the exciting ring-wide dance.
Sometimes, they might be cyclic and planar, but they just don't have the right number of pi electrons to make a decision. Or, they might have some atoms in the ring that are sp3 hybridized. You know, the ones with four single bonds. These guys are like the party poopers, breaking up the continuous pi electron system. They prevent the delocalization. It's like a gatekeeper who won't let everyone into the dance floor.
So, if a compound is cyclic but not planar, or it has sp3 hybridized atoms in the ring that break the pi system, or it just doesn't have the correct number of pi electrons for aromaticity or antiaromaticity, it's usually nonaromatic. They're the bystanders. They're not bad, they're just not participating in the main event.
Let's Classify Some!
Alright, theory is great and all, but let's put it into practice. Imagine we've got a few molecules in front of us. Grab your imaginary clipboard. We're going to classify them.
Example 1: Benzene, The King!
We already talked about benzene. It's got six carbons in a ring. Cyclic? Check! All those carbons are sp2 hybridized, meaning the ring is nice and flat. Planar? Check! And how many pi electrons? There are three double bonds, so that's 3 x 2 = 6 pi electrons. Does 6 fit the (4n + 2) rule? Yep! If n=1, then (41 + 2) = 6. So, benzene is definitely, unequivocally, AROMATIC. Told you it was a superstar.
Example 2: Cyclobutadiene, The Troublemaker
Now, let's look at cyclobutadiene. This is a four-membered ring with two double bonds. Cyclic? Yes. Planar? It *can be planar, which is important for antiaromaticity. Now, the pi electrons. We have two double bonds, so that's 2 x 2 = 4 pi electrons. Does 4 fit the (4n + 2) rule? Nope. Does it fit the 4n rule? Yep! If n=1, then 41 = 4. So, cyclobutadiene is ANTIAROMATIC. It's that unstable, reactive one we talked about. It hates being a square like this. It's actually really hard to isolate because it wants to do something else!
Example 3: Cyclohexene, The Chill Dude
Okay, let's try cyclohexene. This is a six-membered ring with just one double bond. Cyclic? Yes. But is it planar? Not really. A six-membered ring with one double bond is more likely to be in a "chair" or "boat" conformation, which isn't flat. Even if it *could be planar, let's count the pi electrons. We only have one double bond, so that's 2 pi electrons. Does 2 fit (4n + 2)? Yes, if n=0. So it ticks the electron box. But because it's not planar, the electrons can't delocalize properly. It's not really in a stable ring system. Therefore, cyclohexene is NONAROMATIC. It's just a regular old alkene hanging out in a ring.
Example 4: Cyclopentadienyl Anion, The Surprising Star
This one looks a little tricky. It's a five-membered ring, and it has a negative charge (an anion). Let's break it down. Cyclic? Yes. Planar? Yes, it can be. Now for the pi electrons. We have two double bonds, which is 4 pi electrons. But wait! That negative charge? It actually means there are two extra electrons. So, the total pi electron count is 4 (from the double bonds) + 2 (from the anion) = 6 pi electrons. Does 6 fit (4n + 2)? Yes! (n=1). So, the cyclopentadienyl anion is AROMATIC. How about that? Who knew a negative charge could make something so stable?
Example 5: Cyclopentadienyl Cation, The Unhappy Sibling
Now, what if we made that five-membered ring lose an electron? That's the cyclopentadienyl cation. Cyclic? Yes. Planar? Yes. Pi electrons? We start with two double bonds (4 pi electrons), but with a positive charge, we lose two electrons. So, 4 - 2 = 2 pi electrons. Does 2 fit (4n + 2)? Yes! (n=0). So, it's cyclic, planar, and has (4n+2) pi electrons. It should be aromatic, right? WRONG! This is where it gets weird. The cyclopentadienyl cation actually has a significant positive charge localized on one carbon atom, which destabilizes it. It's NONAROMATIC. It's like it wants to be aromatic, but something just isn't right. The electron counting rules aren't the only thing, but they're a really good starting point!
Example 6: Pyridine, The Heterocycle Hero
Let's look at pyridine. This is a six-membered ring, just like benzene, but one of the carbons is replaced by a nitrogen atom. Cyclic? Yes. Planar? Yes, it's planar like benzene. Now, the pi electrons. The nitrogen atom in pyridine is sp2 hybridized and contributes one electron to the pi system, just like carbon would. So, we still have three double bonds in the ring, which means 6 pi electrons. And 6 fits the (4n + 2) rule! So, pyridine is also AROMATIC. See? It's not just about carbon rings!
Example 7: Cyclooctatetraene, The Wiggly One
Last one for now, cyclooctatetraene. It's an eight-membered ring with four double bonds. Cyclic? Yes. Pi electrons? 4 double bonds means 4 x 2 = 8 pi electrons. Does 8 fit (4n + 2)? No. Does it fit 4n? Yes! (n=2). So, by electron count, it should be antiaromatic. BUT! This big ring is actually not planar. It's all wobbly and non-planar to avoid the instability of being antiaromatic. Because it's not planar, it can't delocalize those electrons properly. So, despite having 8 pi electrons, it's NONAROMATIC. It's a clever molecule, choosing to be unreactive by not being flat.
See? It's a bit of a puzzle, isn't it? You've got to check all the boxes: cyclic, planar, and the electron count. And sometimes, the molecule's shape itself throws a wrench in the works!
So, next time you see a ring structure, you can totally impress your friends (or just yourself!) by trying to figure out if it's a stable aromatic, a jumpy antiaromatic, or a just-chillin' nonaromatic. It's like a secret language of molecules, and now you're starting to understand it. Pretty cool, huh? Time for another coffee!
