Classify The Compounds As Strong Acids Or Weak Acids.
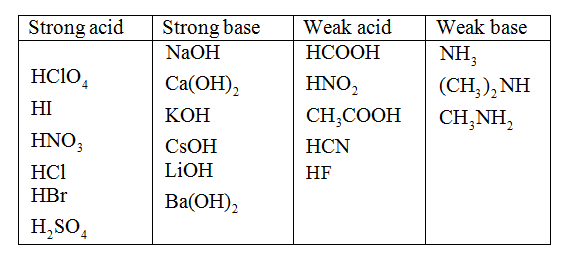
Ever found yourself staring at a list of chemical compounds and wondering, "Which ones are the real powerhouses, and which are just a little bit shy?" Well, get ready to dive into the fascinating world of acid classification! It might sound like something straight out of a science lab, but understanding the difference between strong acids and weak acids is surprisingly relevant to our everyday lives. And the best part? It can actually be a fun and rewarding little mental exercise.
So, why would anyone want to classify acids? Think of it like sorting your tools. You wouldn't use a delicate paintbrush to hammer a nail, right? Similarly, knowing whether an acid is strong or weak helps us understand its potential behavior and applications. For instance, strong acids are like the heavy-duty equipment – they tend to react more vigorously and are excellent at breaking things down. This makes them invaluable in industries like manufacturing, where they’re used to clean metals, process minerals, and even produce fertilizers. They’re the unsung heroes behind many of the products we use daily.
On the other hand, weak acids are more like your everyday utility players. They’re gentler but still incredibly useful. You’ll find them in surprising places! That tangy taste in your yogurt? That's likely lactic acid, a weak acid. The zesty kick in your lemonade comes from citric acid, another weak acid. Even our own bodies rely on weak acids for digestion. Understanding this classification helps us appreciate the delicate balance of chemistry that makes our food taste great and our bodies function smoothly. It’s also crucial in fields like medicine and environmental science, where precise control over chemical reactions is paramount.
Now, how can you get more enjoyment out of classifying these chemical critters? First, start with the common ones. You'll often encounter hydrochloric acid (HCl) and sulfuric acid (H₂SO₄) as prime examples of strong acids. For weak acids, think of acetic acid (in vinegar) and carbonic acid (in fizzy drinks). The more familiar you become, the easier it gets!
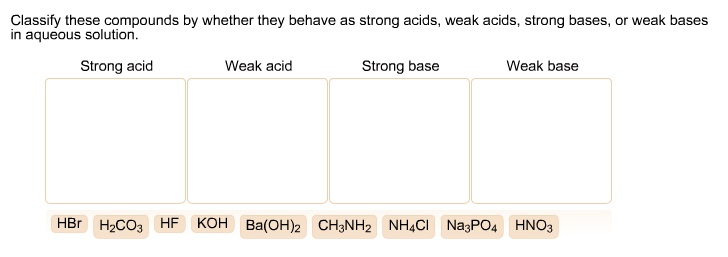
Secondly, don't be afraid of the 'why'. Strong acids completely dissociate in water, meaning they release all their hydrogen ions. Weak acids only partially dissociate. This fundamental difference explains their differing strengths. Visualizing this process, perhaps with simple diagrams or even just mentally, can be really illuminating and add a layer of understanding to your classification. It turns it from a rote memorization task into a puzzle to solve.
Finally, connect it to the real world. When you’re cooking, eating, or even cleaning, try to identify potential acids at play. Knowing that the rust remover uses a strong acid or that your stomach acid is very strong but carefully managed can make the learning process much more engaging. So, next time you encounter a chemical name, take a moment to ponder: is it a powerful strong acid or a more subtle weak acid? It’s a small step, but one that opens up a whole world of chemical understanding and appreciation!
