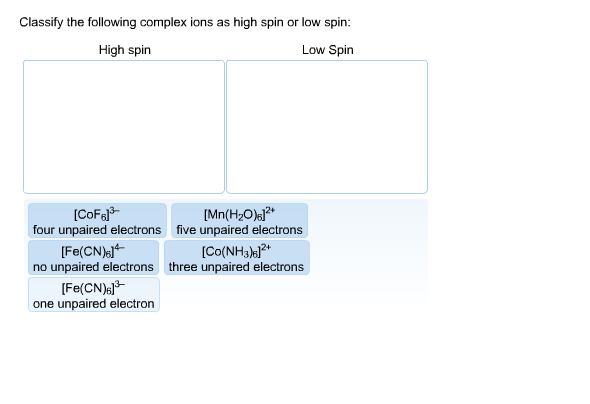
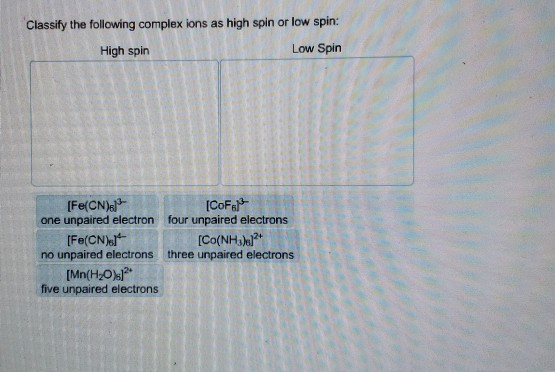
Classify The Complex Ions As High Spin Or Low Spin

Ever wonder what happens when certain atoms decide to team up and form a super-group? It’s like a rock concert, but with molecules! These cool crews are called complex ions, and they’ve got a secret life that’s surprisingly fun to explore. Think of them as tiny, colorful characters playing a game of musical chairs with their electrons. And the way they arrange themselves? That’s where the real party starts!
Now, these complex ions aren't all the same. They have different personalities, and one of the coolest ways to see this is by looking at how they handle their electrons. It’s like choosing between a chill, laid-back vibe or a super-energetic, all-out dance-off. We're talking about the difference between high spin and low spin complex ions. It sounds a bit technical, but trust me, it’s easier and way more interesting than it sounds!
Imagine you have a bunch of friends (electrons) and some cozy spots to sit (orbitals). In the world of complex ions, these friends are a bit picky about where they park themselves. Sometimes, they all want their own comfy chair, spreading out as much as possible. This is the high spin scenario. It’s like everyone claiming their own sofa during a movie marathon – no one wants to share too soon!
In a high spin complex ion, the electrons are spread out to occupy as many individual orbitals as possible before they start pairing up. It’s a strategy of “more space, please!” They’re a bit like a group that prefers to explore every corner of a new city before deciding to hang out in the same cafe. This means they’re not as “packed in.” It's a more relaxed arrangement, almost like a leisurely stroll through a park rather than a sprint.
But then, there are the other guys. These are the low spin complex ions. They’re the ones who are all about getting cozy, fast! They’d rather pair up in the same spots if it means a more “organized” or, dare I say, “efficient” setup. Think of them as the friends who immediately start playing board games in pairs, even if there are plenty of single seats available. They’re not afraid of a little closeness!

In a low spin complex ion, the electrons try to pair up in the lower energy orbitals first, even if it means sharing. They're playing a different game. This often results in fewer unpaired electrons. It's like a dance floor where people are forming couples quickly, minimizing the number of solo dancers. They’re more about teamwork and filling up the available spots in a specific, more crowded way.
What makes this so special? Well, it’s not just about how the electrons are arranged. This seemingly small difference can have a huge impact on the entire complex ion. It’s like changing the band members in a rock group – the sound can totally transform! The color you see, the way it reacts to magnetic fields, even how it behaves in chemical reactions – it can all be different!
It’s like the difference between a quiet acoustic set and a full-on electric guitar solo! Both are music, but boy, are they different!
This high spin versus low spin drama is a huge deal in chemistry. It helps scientists predict and understand how these complex ions will behave. It’s the secret sauce that makes some compounds colorful and others not. It explains why some materials are attracted to magnets and others aren't. It’s the backstage pass to understanding the personalities of these tiny molecular stars!
Think about it: you have a central metal atom, like a star performer. Then, you have ligands, which are like the bandmates or backup dancers, surrounding it. These ligands influence how the electrons in the metal atom arrange themselves. Some ligands are pushy and energetic, forcing the electrons to spread out (high spin). Others are more persuasive, encouraging the electrons to pair up (low spin).
The energy difference between these arrangements is key. When the electrons have to jump to a higher energy level to avoid pairing up, that requires more energy. If the ligands don't provide enough of a push to overcome that pairing energy, the electrons will just pair up in the lower energy levels, leading to a low spin configuration. It’s all about the energy budget!
And the colors! Oh, the colors! The way electrons in these complex ions absorb and reflect light is what gives them their vibrant hues. The specific arrangement of electrons – whether they are spread out in a high spin state or paired up in a low spin state – dictates which wavelengths of light are absorbed and which are reflected. So, that dazzling blue of a copper compound or the deep green of a nickel complex? That’s the high spin or low spin dance playing out!

It’s a bit like choosing an outfit. Do you go for a loose, flowing style (high spin) or a more form-fitting, sleek look (low spin)? Both have their own aesthetic appeal and functionality. And in the world of complex ions, this choice profoundly affects their properties.
This concept is a cornerstone for understanding the behavior of transition metals, which are the real divas of the periodic table when it comes to forming these complex ions. They have a knack for juggling electrons in these different orbital arrangements, leading to a rich tapestry of chemical behaviors.
So, the next time you see a colorful chemical or hear about a material’s magnetic properties, remember the hidden drama of high spin versus low spin. It’s a fascinating peek into the energetic world of complex ions, where electrons play musical chairs and the arrangement of the players determines the show!
