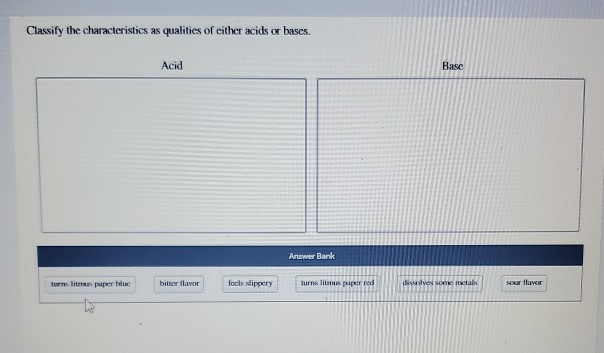
Classify The Characteristics As Qualities Of Either Acids Or Bases

Hey there, science curious folks! Ever wonder what makes things like lemon juice so tart or soap feel so slippery? Well, it all boils down to some fundamental properties that chemists love to chat about: acids and bases. Think of them like two different personalities in the chemical world, each with their own quirks and ways of interacting. It's not as intimidating as it sounds, promise! We're just going to dip our toes in and see what makes them tick.
So, What's the Deal with Acids?
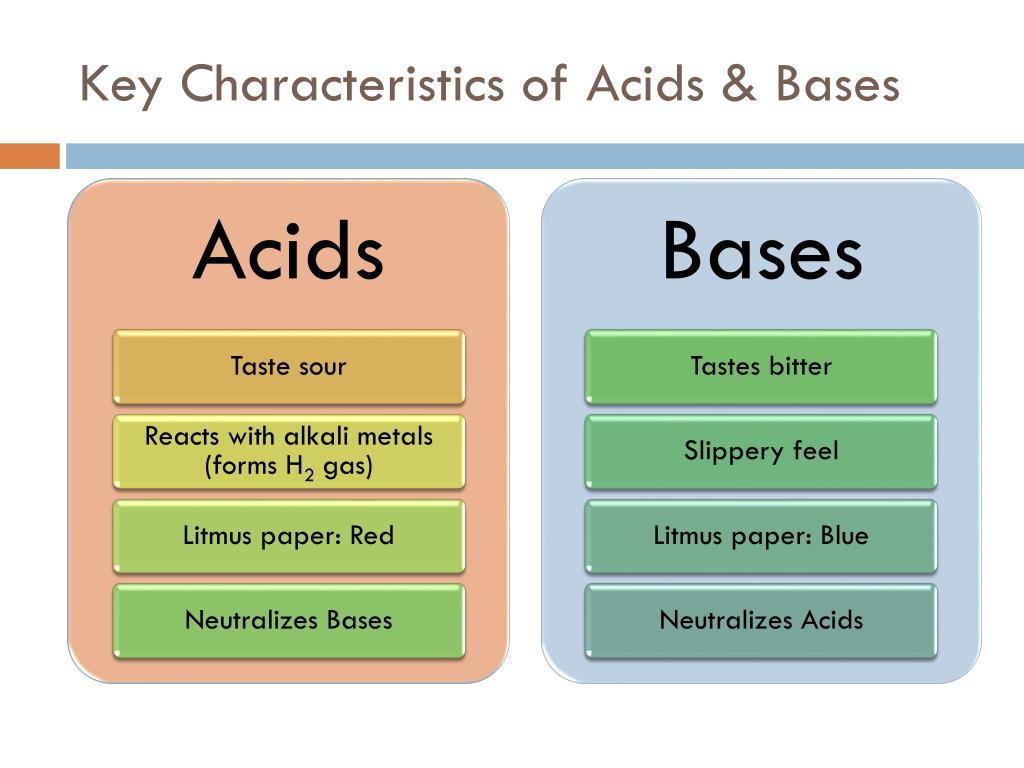
Imagine a really sour candy. That zingy, mouth-puckering sensation? That's often the work of an acid! Acids are like the bold, sometimes feisty characters. They tend to be sour tasting (though, please, don't go around tasting random chemicals to check – not a good idea!).
One of their most defining features is their ability to donate a tiny little particle called a proton (which is basically just a hydrogen atom without its electron). It's like they're always ready to share something. This proton-donating action is what makes them, well, acidic!
Think about it this way: when you squeeze a lemon, you're releasing citric acid. That acid interacts with your taste buds, giving you that sharp, tangy flavor. It's a chemical reaction, and it's happening right there on your tongue!
Acids also have a knack for corroding metals. Ever seen an old, rusty pipe? Acids can be a part of that story. They're not always aggressive, but they have the power to break down certain materials over time. It's like they're a little bit of a force of nature.
And here's a cool trick: acids often react with things called carbonates. What happens when you mix baking soda (a base) with vinegar (an acid)? Fizzing, right? That bubbly release is carbon dioxide gas, and it’s a classic sign that an acid is doing its thing. It’s like a little chemical party happening in your beaker!
Chemists have a handy way to measure how acidic or basic something is, called the pH scale. On this scale, numbers from 0 to 7 are acidic. The lower the number, the stronger the acid. So, battery acid (yikes!) is way down at the bottom, while something like milk is only mildly acidic.
So, to recap the acid club: sour taste (in safe, edible things!), a tendency to donate protons, the ability to corrode metals, and a love for reacting with carbonates to make bubbles. Pretty neat, huh?
Now, Let's Meet the Bases!
If acids are the bold ones, bases are often the smoother, more soothing characters. Think about how soap feels on your hands. It's that slippery or soapy feel. That's a classic base quality! Bases are the ones that often feel a bit luxurious, in a chemical way.
Instead of donating protons, bases are more about accepting protons. It's like they're looking for something to hold onto. Or, another way to think about them is that they often release something called hydroxide ions (OH-). It's like they're exhaling a different kind of chemical particle.
You know that feeling when you get soap in your eye? Ouch! Bases can feel quite bitter. Again, do not taste random things, but in general, if something tastes bitter, it's a good hint it might be a base. They're not as universally appealing in taste as a tart lemon might be!
Bases are also fantastic at neutralizing acids. Remember that vinegar and baking soda reaction? Baking soda is a base, and it loves to calm down the fizziness of the acid. It's like they have this yin-and-yang relationship, always trying to bring each other into balance.
This neutralization is super important. It's why antacids work! When you have too much stomach acid, you take an antacid (which is a base) to calm things down. It’s a real-life application of acid-base chemistry happening in your own body!
On our pH scale, bases hang out from 7 to 14. The higher the number, the more basic (or alkaline) something is. So, baking soda is around a pH of 8 or 9, while drain cleaner (definitely don't touch!) is way up there, super basic.
So, the key traits of bases: a slippery, soapy feel, a tendency to be bitter, the ability to accept protons (or release hydroxide ions), and a talent for neutralizing acids. They're the calmers of the chemical world!
Putting It All Together: The Classification Game
Let's play a quick game. Imagine we have a list of characteristics. Can you guess if it's an acid or a base we're talking about? It's like a chemical personality quiz!
Characteristic: Sour taste. Hmmm, tart lemons, sour candies... that sounds like our friend, the acid!

Characteristic: Feels slippery. Like washing your hands after doing some gardening... definitely a base!
Characteristic: Donates protons. This is the core definition of our energetic acid.
Characteristic: Accepts protons. The opposite of donating, this is the defining action of a base.
Characteristic: Bitter taste. Not as pleasant as sour, this points towards a base.
Characteristic: Corrodes metals. While both can do this to some extent, strong acids are often the culprits here.
Characteristic: Neutralizes acids. This is the peacemaker role, the job of a base.
Characteristic: pH below 7. That's the zone for our acids on the pH scale.
Characteristic: pH above 7. The territory for the smooth operators, the bases.
It's pretty cool how these simple differences in how they interact with other molecules give them such distinct personalities and uses. From making food taste exciting to cleaning our homes, acids and bases are all around us, doing their chemical thing.
So next time you're enjoying a squeeze of lime or washing your hands, you can think, "Ah, that's the work of an acid!" or "Hello, familiar base!" It's a little peek into the amazing, invisible world of chemistry that makes our everyday lives possible. Pretty fascinating, right?
