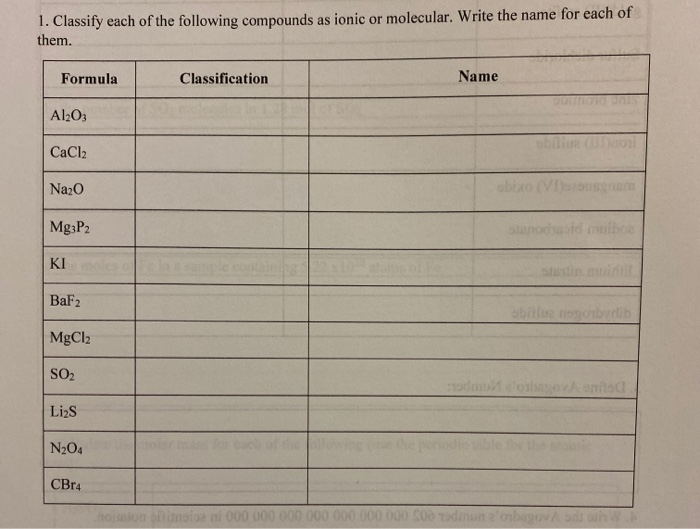
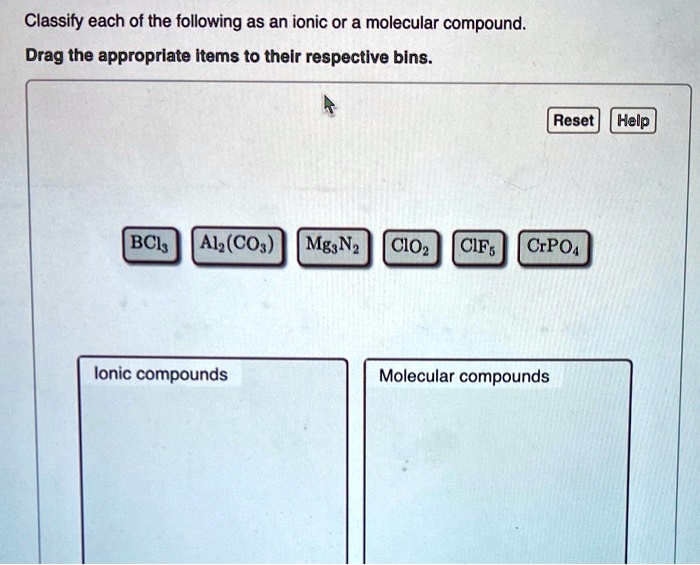
Classify Each Of The Following Compounds As Ionic Or Molecular

Alright, chemistry adventurers! Ever look at a bunch of chemical formulas and feel like you're staring at ancient hieroglyphics? Well, buckle up, buttercups, because we're about to unlock the secret code! It's all about figuring out if these little guys are like a super-tight, super-strong ionic duo, or more like a chill, laid-back group of buddies, forming a molecular squad.
Think of it like this: some compounds are all about that intense, almost dramatic transfer. One element is like, "Here, take my electron! I don't need it!" and the other is like, "OMG, yes! I've been waiting for this!" It's a love story written in the stars (or, you know, electron shells). These are your ionic compounds, and they are powerful.
Then you've got your molecular compounds. These guys are more about sharing. They're like, "Hey, let's pool our resources! We can totally build something awesome together." It's a cooperative effort, a testament to teamwork and mutual understanding. They’re the ultimate friend group, always sticking together.
Let's Dive into Some Examples, Shall We?
Example 1: NaCl
First up, we have NaCl. This is a classic! You probably know it as good old table salt. Now, imagine sodium (Na) is a bit of a show-off, always ready to give away an electron to be extra stable. Chlorine (Cl), on the other hand, is a bit of a hoarder, desperately wanting just one more electron to feel complete.
So, what happens? Sodium, with a flourish, hands over its electron to chlorine. It's a complete transfer! This creates a charged situation, where the now-positive sodium and the now-negative chlorine are super attracted to each other, like tiny magnets. This is the hallmark of an ionic compound!
NaCl is a rockstar ionic compound. It’s not messing around with sharing; it's all about that electron transfer!
Example 2: H₂O
Next on our chemical catwalk is H₂O. This one is so familiar, you probably drink it every day – it’s water! Now, hydrogen (H) and oxygen (O) are a bit more reserved. They're not really into the whole "give and take" drama.

Instead, they decide to share their electrons. It's like they're holding hands really, really tightly. This sharing of electrons is what makes them a molecular compound. They're a team, working together to achieve stability.
H₂O is the ultimate chill squad. They prefer cooperation and sharing over all that dramatic electron transfer business. Definitely molecular!
Example 3: KBr
Let's look at KBr. This one is potassium bromide, and it’s another one that’s all about that electron exchange! Potassium (K) is super generous, always ready to donate an electron to achieve that perfect electron configuration. Bromine (Br) is right there, eager to accept that electron.
When potassium gives its electron to bromine, they become charged ions. These oppositely charged ions then stick together like superglue due to their strong electrostatic attraction. This is the definition of an ionic compound in action!

KBr is definitely a fan of the electron transfer tango. Get ready for some serious attraction – it’s ionic all the way!
Example 4: CO₂
Now, let's talk about CO₂, also known as carbon dioxide. This is the stuff we breathe out! Carbon (C) and oxygen (O) are not into giving up their electrons. They'd rather play the sharing game.
Carbon and oxygen atoms share electrons to form a stable molecule. They form strong covalent bonds, where the electrons are shared equally (or at least, pretty close to equally). This collaborative effort is the signature move of a molecular compound.
CO₂ is all about that shared vibe. These atoms are best friends who believe in working together, making it a classic molecular compound.
Example 5: MgO
Moving on to MgO, or magnesium oxide. Magnesium (Mg) is a metal that’s practically bursting with electrons it wants to get rid of. Oxygen (O), on the other hand, is a nonmetal that’s always looking to grab some electrons.
Magnesium enthusiastically transfers its electrons to oxygen. This creates positively charged magnesium ions and negatively charged oxide ions. The strong attraction between these opposite charges holds them together in a crystal lattice. Yep, you guessed it – this is ionic behavior!
MgO is all about the grand electron transfer. It’s a powerful bond, and that makes it a definitive ionic compound!
Example 6: SO₂
Let’s analyze SO₂, sulfur dioxide. Sulfur (S) and oxygen (O) are both nonmetals. When nonmetals get together, they usually prefer to share their electrons rather than engage in any dramatic giveaways.
Sulfur and oxygen atoms share electrons to form a stable molecule. They form covalent bonds, where the electrons are part of a shared team. This sharing is the key characteristic that classifies them as a molecular compound.
SO₂ prefers a friendly game of electron sharing. They’re a solid team, and that means they are molecular!
Example 7: KCl
And now, KCl, potassium chloride! Potassium (K) is a metal, and chlorine (Cl) is a nonmetal. When you have a metal meeting a nonmetal in the chemical world, it’s almost always a recipe for electron transfer.
Potassium is super happy to donate an electron to chlorine. This creates potassium ions and chloride ions, which are then strongly attracted to each other. This powerful electrostatic attraction is the defining feature of an ionic compound.
KCl is a prime example of the electron transfer phenomenon. It’s the classic metal-nonmetal combo, making it undeniably ionic.
Example 8: NH₃
Finally, let’s wrap up with NH₃, ammonia! This compound is made of nitrogen (N) and hydrogen (H), both of which are nonmetals. When nonmetals team up, they are masters of electron sharing.
Nitrogen and hydrogen atoms share electrons to form stable bonds. They form covalent bonds, where the electrons are a shared resource. This cooperative electron arrangement is the signature of a molecular compound.
NH₃ is the epitome of teamwork! These atoms are all about sharing their electrons, which makes them a fantastic molecular compound.
See? It’s not so scary, is it? Just remember the drama of electron transfer for those ionic powerhouses, and the friendly sharing for the cooperative molecular buddies. You've totally got this! Keep exploring, and happy chemical classifying!
