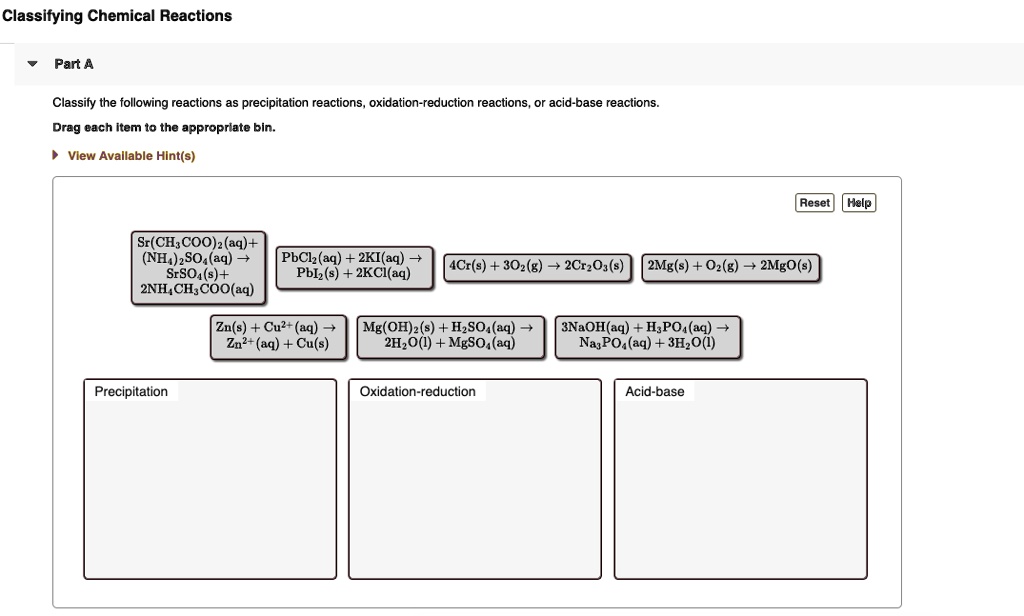
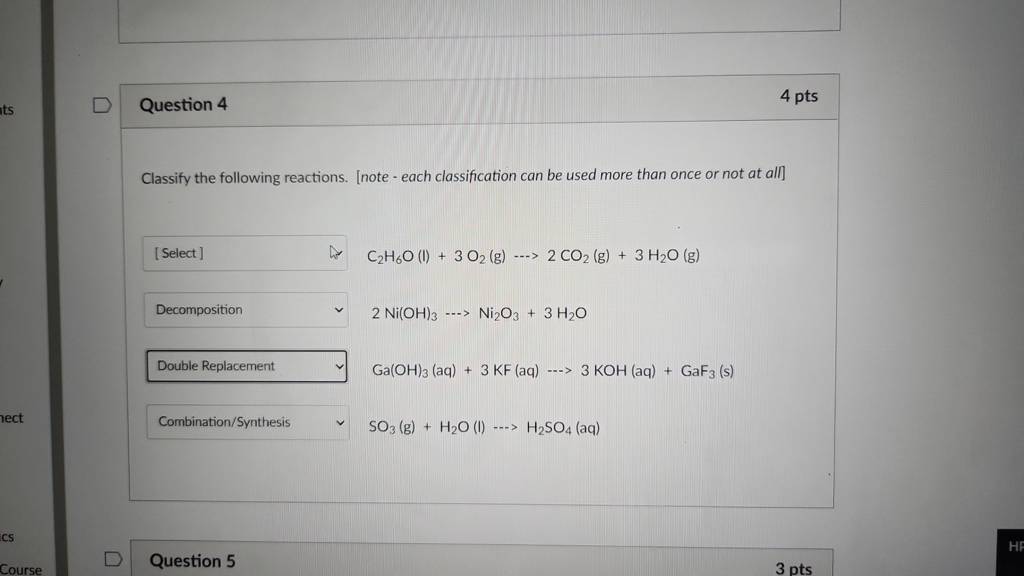
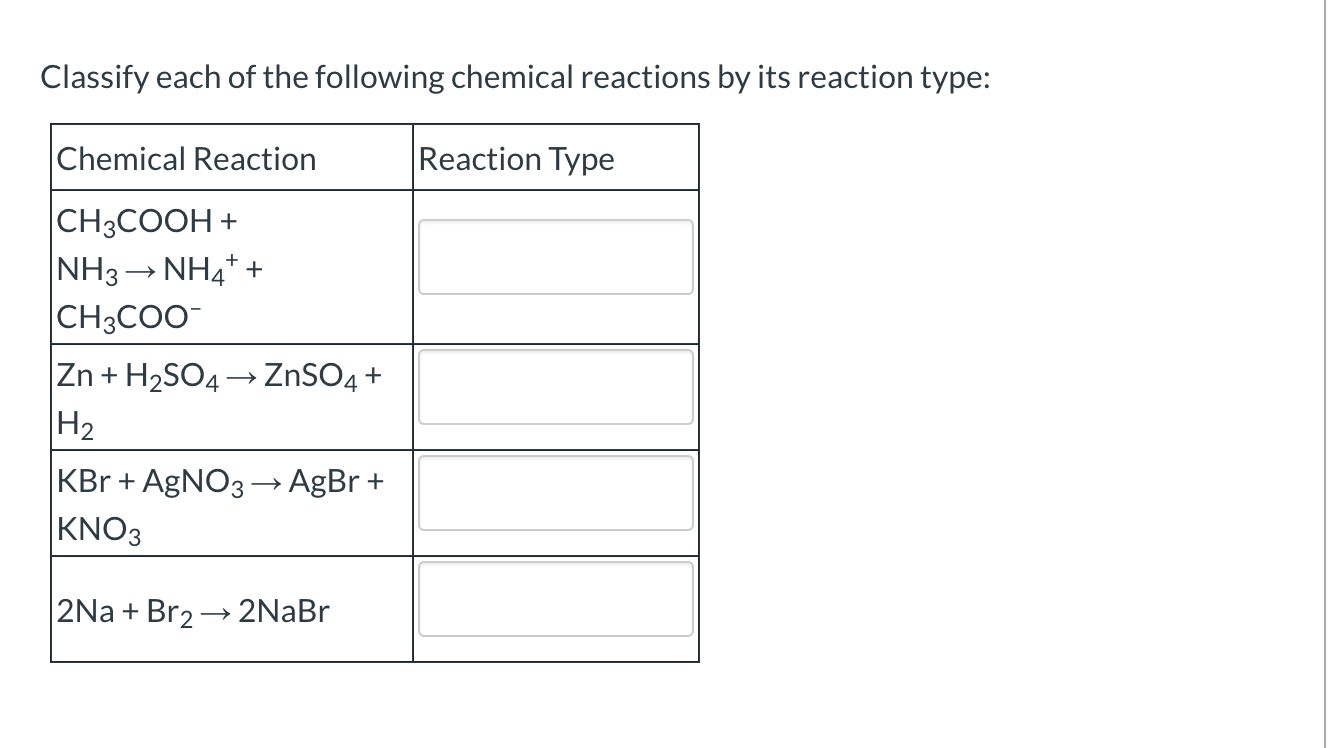
Classify Each Of The Following Chemical Reactions.
Hey there, science enthusiasts and curious minds! Ever feel like chemistry class was a bit of a snoozefest? Well, buckle up, buttercup, because we're about to dive into the wonderfully weird world of classifying chemical reactions. Think of it like sorting your sock drawer, but way more explosive and with fewer lost socks. (Hopefully.)
Why bother classifying? It's like giving things names. It helps us understand them. It's like saying, "Oh, that's a cat!" instead of just pointing at a furry creature. Knowing the "type" of reaction helps scientists predict what's gonna happen. Pretty neat, right?
Let's get this party started with the O.G. of reaction types: Synthesis. Imagine two shy molecules, feeling a little lonely. They meet, they click, and BAM! They become one bigger, happier molecule. It's basically chemical "getting together." Like two peas in a pod, but with more electron sharing.
Think of it this way: A + B --> AB. Simple, right? Like baking a cake. You take flour and eggs and sugar (your A and B) and you combine them to make a delicious cake (your AB). See? Not so scary.
A super common example? When hydrogen gas and oxygen gas get frisky and make water. H₂ + O₂ --> H₂O. That’s the stuff we drink and swim in! Pretty essential synthesis, wouldn’t you say?
Next up, the opposite of synthesis: Decomposition. This is when a big, established molecule is just like, "You know what? I'm done." It breaks apart into smaller, independent molecules. It's the chemical equivalent of a breakup, but without the awkward shared Netflix password situation.
The general vibe here is AB --> A + B. One thing becomes two or more. Like when you take a big chocolate bar and break it into smaller pieces to share. (Or, you know, not share.)
A classic example? When hydrogen peroxide (that stuff your mom used to clean cuts) breaks down into water and oxygen gas. 2H₂O₂ --> 2H₂O + O₂. Poof! Gone.
Now, things get a little more dramatic with Single Displacement (or Single Replacement, if you’re fancy). This is like a social butterfly in the molecular world. One element decides it wants to ditch its current partner and hook up with another element. It's a chemical love triangle!

The pattern is A + BC --> AC + B. Element A swoops in and kicks B out of its bond with C, and then A bonds with C. It’s a real chemical power play.
Imagine zinc metal dropped into a solution of copper chloride. The zinc is like, "Hey copper, this chlorine is mine now!" and the zinc bumps the copper out. Zn + CuCl₂ --> ZnCl₂ + Cu. Suddenly, you have zinc chloride and free-wheeling copper. Awkward for copper!
Then we have Double Displacement (or Double Replacement, you know the drill). This is where things get really interesting. It's like a square dance for molecules. Two compounds swap partners, and usually, something new and exciting (or sometimes, a solid precipitate) pops out.
The blueprint here is AB + CD --> AD + CB. Both the "A" and "C" atoms (or ions, to be precise) switch places and find new partners. It’s a complete molecular reshuffle.
Picture mixing silver nitrate and sodium chloride (that's just salt!). The silver and sodium get confused and switch partners with the nitrate and chloride. AgNO₃ + NaCl --> AgCl + NaNO₃. The cool part? Silver chloride (AgCl) is a solid that clumps up and falls out of the solution. It’s called a precipitate, and watching it form is seriously mesmerizing. It's like chemistry magic happening right before your eyes!
Don't forget the spicy one: Combustion! This is where things get fiery. Combustion reactions are all about reacting with oxygen, usually to produce heat and light. Think fire. Think explosions. Think your birthday candles!
The most common combustors are hydrocarbons (molecules made of hydrogen and carbon). When they burn completely, they make carbon dioxide and water. CH₄ + 2O₂ --> CO₂ + 2H₂O. That’s methane, the main component of natural gas, doing its fiery thing.

The amazing thing about combustion is the energy released. It’s what powers our cars, heats our homes, and makes fireworks so darn pretty. It’s the ultimate chemical showstopper.
And sometimes, reactions don't fit neatly into one box. That's okay! Science is messy, and so is chemistry. Some reactions might have elements of multiple types. It's like a chameleon reaction, changing its stripes depending on how you look at it.
But understanding these basic categories gives you a fantastic starting point. It's like learning the basic chords on a guitar before you start shredding solos. You can start to predict what will happen, and that's where the real fun begins.
So next time you see something bubbling, fizzing, or maybe even catching fire (please don't try that at home!), you'll have a better idea of what kind of chemical dance it's performing. It’s not just random chaos; it’s organized, predictable, and sometimes, utterly spectacular. Pretty cool, huh? Keep exploring, keep questioning, and keep that sense of wonder alive!
