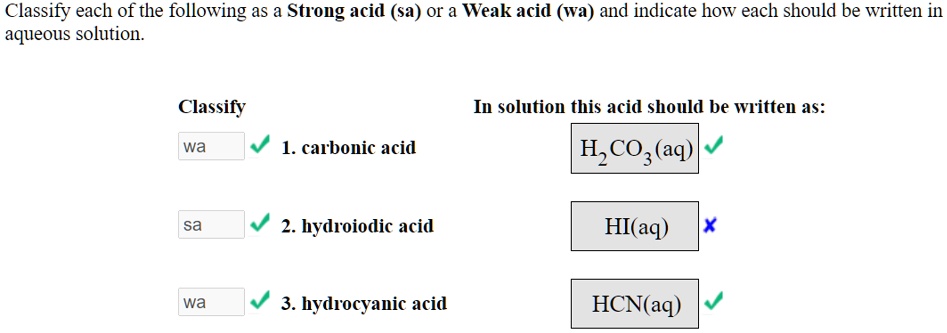
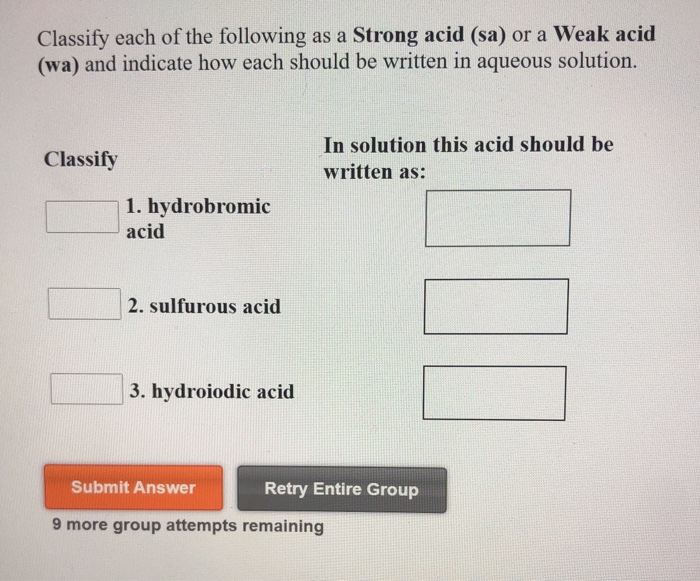
Classify Each Of The Following Acids As Strong Or Weak

Hey there, fellow humans! Let's chat about something that sounds a bit science-y but is actually kinda cool and pops up in our lives more than you might think: acids. Now, don't let the word "acid" scare you. We're not talking about the kind that eats through metal like in a mad scientist movie (though some of those exist!). We're talking about acids that are part of our everyday world, from the tang in your orange juice to the stuff that helps clean your bathroom.
The really interesting thing about acids is that they come in different "strengths." Think of it like people: some are super outgoing and talkative, while others are a bit more reserved. Acids are kind of the same! They can be strong or weak, and understanding that difference can actually be pretty neat. Why should you care? Well, it helps us understand why some things taste sour and others don't, why certain cleaners are tougher than others, and even why our bodies work the way they do!
The "Let's Go All Out!" Acids: Strong Acids
So, let's start with the big guns, the strong acids. Imagine a tiny party animal who loves to mingle. When a strong acid gets into water (which is like its social scene), it completely dives in and breaks apart into its smallest bits, called ions. It's like that friend who, at every party, immediately finds the dance floor and starts a conga line, dragging everyone with them. There's no holding back!
A classic example you might have heard of is hydrochloric acid (HCl). Now, you won't find this casually in your kitchen cabinet, but guess what? Your own stomach lining produces a diluted version of it to help digest your food! Yep, your belly is a little chemistry lab, and hydrochloric acid is its hardworking manager, breaking down your lunch so you can get all the good stuff. Pretty wild, right?
Another strong acid is sulfuric acid (H₂SO₄). This one is a real powerhouse and is used in tons of industrial processes, like making fertilizers and even in car batteries. Think of it as the ultimate go-getter. When it hits water, it’s like a tidal wave of activity. It’s so good at donating its acidic bits that it's practically unstoppable in that regard.
The key thing about strong acids is that they are completely willing to give up their acidic parts. There's no hesitation, no "maybe later." They just go for it. This makes them very reactive and very effective at what they do, whether that's dissolving food in your stomach or helping to make the products we use every day.
The "Chill and Take My Time" Acids: Weak Acids
Now, let's meet the other side of the coin: the weak acids. These are more like the thoughtful conversationalist at the party. When a weak acid enters water, it's a bit more hesitant. It might break apart a little, but it also holds onto a lot of itself. It's not as gung-ho about diving in and dissociating.

Think about acetic acid (CH₃COOH). You know this one! It's the main ingredient that gives vinegar its distinctive tangy bite. When you drizzle vinegar on your salad, you're experiencing a weak acid. It's definitely acidic – it makes your taste buds tingle! – but it's not going to cause a dramatic chemical explosion. It's just doing its thing, adding a little zing.
Another common weak acid is citric acid. This is found in lemons, oranges, and all those other delicious citrus fruits. That zesty, sour punch in your lemonade? That's citric acid at work! It’s strong enough to give that sour taste, but it’s also perfectly safe and even beneficial for us to consume.
The "weakness" of these acids comes from their reluctance to fully break apart. They exist in a bit of a happy medium, with some of their acidic parts free and some still attached. It’s like having a conversation with someone who occasionally pauses to think before continuing. It’s still a valid conversation, just a bit more measured.
Why Does This "Strength" Thing Even Matter?
Okay, so we've got the all-out party animals (strong acids) and the thoughtful conversationalists (weak acids). Why should we, as everyday people, give a hoot about this distinction?
Well, for starters, it explains a lot about taste. That sharp, biting sourness you get from, say, lemon juice (citric acid, a weak acid) is different from the intensely corrosive feel of something truly strong. Strong acids, if not handled with extreme care, can cause serious damage because they are so readily reactive. Weak acids, on the other hand, are often what give our food its appealing flavors.
/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)
It also impacts how we use them for cleaning. A strong acid might be excellent for dissolving tough mineral deposits or rust. However, for everyday cleaning around the house, you’ll often find yourself using products with weak acids. Think about bathroom cleaners that use citric acid or products that leverage the cleaning power of vinegar. They get the job done without being overly harsh or dangerous.
And then there’s our own bodies! As we mentioned, your stomach uses a strong acid (hydrochloric acid) to break down food. But the rest of your body needs to maintain a very delicate balance, and strong acids circulating freely would be a disaster. So, your body uses sophisticated buffer systems, often involving weak acids and their counterparts, to keep everything in check. It's like having tiny chemical referees making sure the acidity levels stay just right.
Understanding the difference between strong and weak acids helps us appreciate the chemistry happening all around us and even within us. It's not just abstract science; it's the reason your orange juice is refreshing, why your digestive system works, and why certain cleaning products are effective yet safe. So next time you're enjoying a tangy fruit or using a household cleaner, you can give a little nod to the fascinating world of strong and weak acids!
