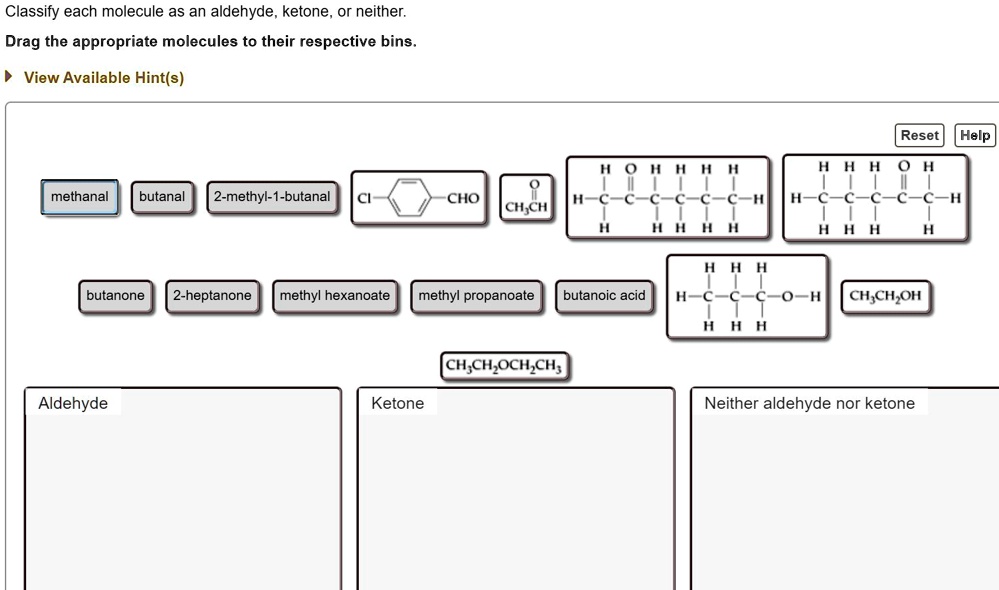
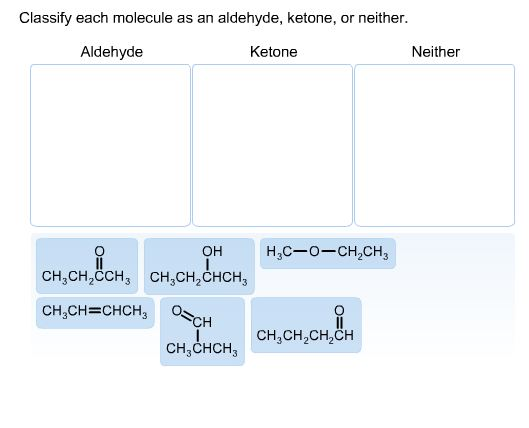
Classify Each Molecule As An Aldehyde Ketone Or Neither
Alright, chemistry nerds and anyone who’s ever vaguely remembered a lab class from, like, forever ago, gather ‘round. We’re about to embark on a grand adventure. No, no, it’s not a quest for the Holy Grail, or even finding matching socks. This is far more thrilling. We’re classifying molecules! Cue the dramatic music!
Now, you might be thinking, “Molecules? Isn’t that… work?” And to that, I say, “Maybe a little. But we’re making it fun. Think of it as a molecular guessing game. And the stakes? Just the satisfaction of knowing. Which, let’s be honest, is a pretty big deal.”
So, we’ve got three categories we’re playing with today. It’s like a tiny, chemical popularity contest. We’ve got our rockstars: the Aldehydes. Then we have their slightly more… grounded cousins, the Ketones. And finally, the wild cards, the rebels without a carbonyl group, the “Neither” category. They’re just… there. Doing their own thing. And that’s okay.
Let’s talk about our first contenders, the Aldehydes. These guys are pretty distinctive. They’ve got this special little feature, a part of them that’s like a tiny, eager hand reaching out. It’s a carbon double-bonded to an oxygen, and then, critically, it has at least one hydrogen attached. This makes them quite reactive, you see. They’re the ones who are always ready for a party, always willing to get into a reaction. Think of them as the social butterflies of the molecular world. Always mingling, always involved.
On the other hand, we have the Ketones. They’re also pretty neat. They have that same carbon double-bonded to an oxygen thing going on, that lovely carbonyl group. But here’s the twist, the plot thickening: instead of having a hydrogen attached to that carbonyl carbon, it has two other carbon atoms. So, the carbonyl carbon is sandwiched between two other pieces of the molecule. It’s like they’re a bit more reserved, a bit more self-contained. They still have the sparkle, but they’re not quite as quick to jump into everything.
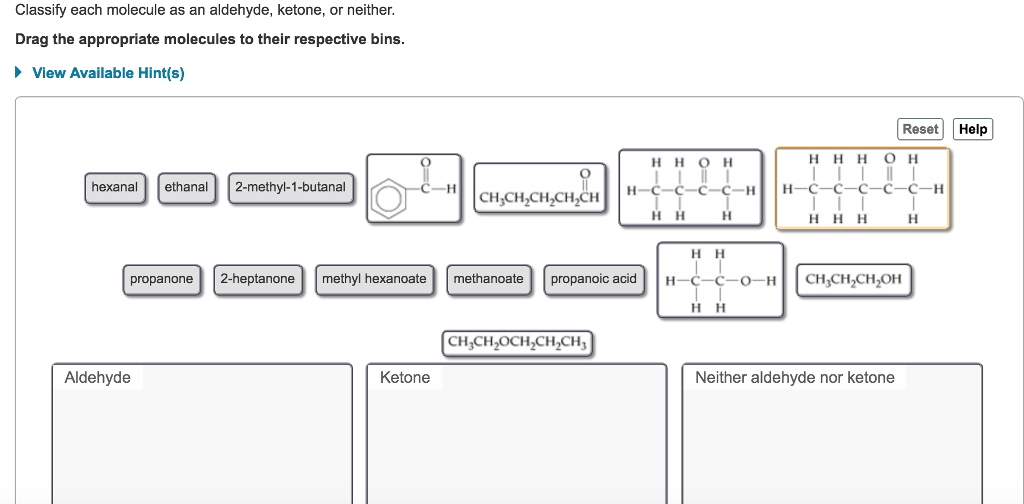
And then there’s the “Neither” club. These are our independent thinkers. They might have oxygen, they might have carbons, but they just don’t tick the boxes for aldehyde or ketone. They might have an oxygen in a different spot, or maybe that carbonyl group is attached to something that disqualifies it from being an aldehyde or ketone. They are the ones who are just… vibing. They exist, they contribute to the grand tapestry of chemistry, but they’re not going to win any “most reactive group” awards. And you know what? That’s totally valid. Sometimes, just being yourself is enough.
It’s sort of like looking at a group of friends. You have the one who’s always organizing the outings, the life of the party (that’s our Aldehyde). Then you have the one who’s happy to go along with the plan, maybe suggesting the snacks (our Ketone). And then you have the friend who’s happy to just chill, read a book, or scroll through TikTok. They’re not doing anything wrong, they’re just not the one spearheading the next adventure. They’re the “Neither.”

So, how do we spot these guys? It’s all about that specific arrangement of atoms. That little group: a carbon double-bonded to an oxygen. If that carbon also has a hydrogen, hello Aldehyde! If that carbon has two other carbons attached to it, say hello to Ketone! If it doesn’t fit either of those descriptions, well, you know where they belong. It’s not rocket science, but it is molecule science. And that’s practically the same thing, right?
Sometimes, you’ll see molecules that look almost like they belong. They’re like that friend who says, "Oh, I almost bought tickets to that concert!" Close, but no cigar. These are our “Neither” folks. They might have that carbonyl, but it’s hanging out somewhere it shouldn’t be for our strict aldehyde or ketone rules. Maybe the carbon in question is also bonded to another oxygen, or it's part of a ring structure in a way that disqualifies it. They’re the molecular equivalent of a “participation trophy.” And again, no shade!
Let’s take a peek at some examples, shall we? Imagine something like formaldehyde. That’s our classic aldehyde. Simple, effective, gets the job done. Then you have acetone, the stuff in nail polish remover. That’s a perfectly good ketone. Easy peasy. Now, what about something like ethanol, the alcohol? It has an oxygen, but it’s in the wrong place to be an aldehyde or ketone. So, ethanol is firmly in the “Neither” camp. See? You’re basically a chemist now.
There's a whole universe of molecules out there, each with its own personality and purpose. And understanding these basic classifications is like learning the alphabet. Once you know your A's, B's, and C's, you can start reading the whole book. So, next time you’re wondering about a molecule’s identity, just remember: does it have that special handshake with a hydrogen (aldehyde)? Is it cozy with two other carbons (ketone)? Or is it just doing its own molecular thing (neither)? You’ve got this. Now go forth and classify!
