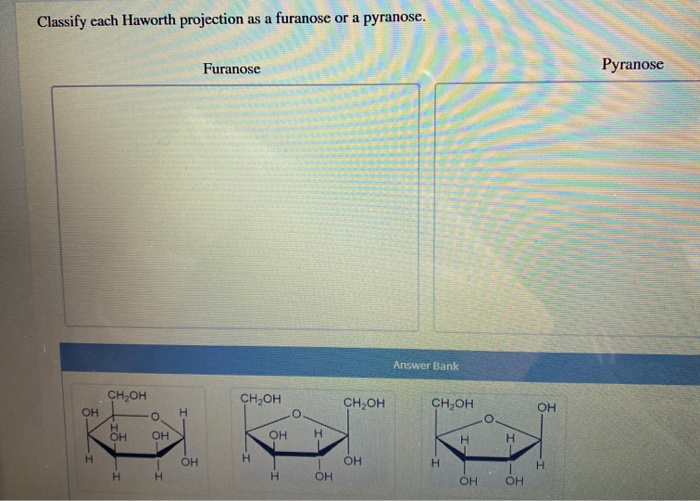
Classify Each Haworth Projection As A Furanose Or A Pyranose.
Ever stared at those twisty, hexagonal diagrams of sugar molecules and wondered what all the fuss is about? Well, get ready to unlock a secret world of carbohydrate coolness! Understanding how sugar molecules arrange themselves in their ring forms isn't just for super-scientists; it's like learning a secret handshake for a whole universe of delicious and vital compounds. From the energy in your morning toast to the building blocks of life itself, sugars are everywhere, and their ring shapes play a huge role in how they function. So, let's dive in and discover how to tell apart these two fundamental sugar rings – it’s easier than you think and surprisingly satisfying!
The Wonderful World of Sugar Rings
Carbohydrates, or sugars, are the fuel that powers our bodies and the foundation of so many natural substances. When sugars are floating around in water, like in your bloodstream or a cell, they don't just stay as open chains. Oh no, they get fancy and curl up into rings! These rings are the most common and biologically relevant forms of sugars. Now, there are two main types of rings that sugars love to form, and knowing the difference is key to understanding their chemistry and how they interact with everything around them.
Think of it like this: imagine you have a flexible noodle. You can bend it into different shapes. Sugars are a bit like that noodle. In their ring form, they usually end up as either a five-membered ring or a six-membered ring. These aren't just arbitrary shapes; they have specific names and properties that tell us a lot about the sugar itself. The practice of classifying these ring structures, especially using a particular type of drawing called a Haworth projection, is incredibly useful. It’s a standardized way to represent these complex 3D structures in a clear, 2D format.
Why Does This Matter? The Purpose and Benefits
So, why bother classifying sugar rings? Well, it's all about clarity and communication in the world of chemistry and biology. When scientists talk about sugars, using precise terminology helps everyone understand exactly which form of the sugar is being discussed. This is crucial for:
- Understanding Biological Processes: The shape of a sugar ring influences how it interacts with enzymes, how it's transported across cell membranes, and how it stores energy. For example, glucose, our primary energy source, exists predominantly as a six-membered ring.
- Drug Design: Many medicines are based on or interact with sugar molecules. Knowing the precise structure of these sugars is vital for designing effective and safe drugs.
- Food Science: The sweetness, texture, and digestibility of foods are all influenced by the types of sugars present and their ring structures. Think about how different sugars behave in baking!
- Genetics and Molecular Biology: Sugars are fundamental components of DNA and RNA (as ribose and deoxyribose, which are furanoses) and are involved in cell signaling.
The Haworth projection is our superhero tool here. It's a simplified, flat representation that highlights the key features of the ring structure. It helps us visualize which atoms are above or below the plane of the ring, giving us essential clues about the sugar's identity and behavior.

Furanose vs. Pyranose: The Big Two
Now, let's get to the main event! The two main types of sugar rings we encounter in Haworth projections are called furanoses and pyranoses. The names themselves give us a hint. They are derived from two common cyclic organic compounds:
Furan: A five-membered ring containing four carbon atoms and one oxygen atom.
Pyran: A six-membered ring containing five carbon atoms and one oxygen atom.
CHAPTER 7 Carbohydrates and Glycobiology - ppt download
When a sugar molecule curls up, it often forms a ring that resembles one of these structures. The trick to classifying them in a Haworth projection is simply to count the atoms that make up the ring. Remember, the oxygen atom within the ring is part of the ring structure itself. Typically, the oxygen atom is shown at the "back" of the ring in a Haworth projection.
How to Classify Them: A Quick Guide
Let’s break down how to identify a furanose versus a pyranose from a Haworth projection:
.png?revision=1&size=bestfit&width=412&height=201)
Furanose Rings:
- Look at the ring structure in the Haworth projection.
- Count the number of atoms that are part of the ring itself. This includes the oxygen atom shown within the ring.
- A furanose ring will have a total of five atoms in the ring structure. This consists of four carbon atoms and one oxygen atom.
- Visually, furanose rings tend to look more "pentagonal" or five-sided in their representation.
- Common examples of furanoses are the rings formed by ribose and deoxyribose, which are crucial components of RNA and DNA respectively.
Pyranose Rings:
- Again, examine the ring in the Haworth projection.
- Count the atoms forming the ring, including the ring oxygen.
- A pyranose ring will have a total of six atoms in the ring structure. This consists of five carbon atoms and one oxygen atom.
- These rings typically appear more "hexagonal" or six-sided in the Haworth projection.
- The most famous example of a pyranose is the ring form of glucose, the sugar your body uses for immediate energy. Other common monosaccharides like fructose and galactose also commonly form pyranose rings.
It's truly as simple as a quick count! When you see a Haworth projection, just find the ring, count the atoms that make up that ring (remembering to include the oxygen atom that's part of the ring), and voilà! Five atoms in the ring means it's a furanose. Six atoms in the ring means it's a pyranose. This fundamental skill opens up a much deeper understanding of the fascinating world of carbohydrates and their indispensable roles in life.

