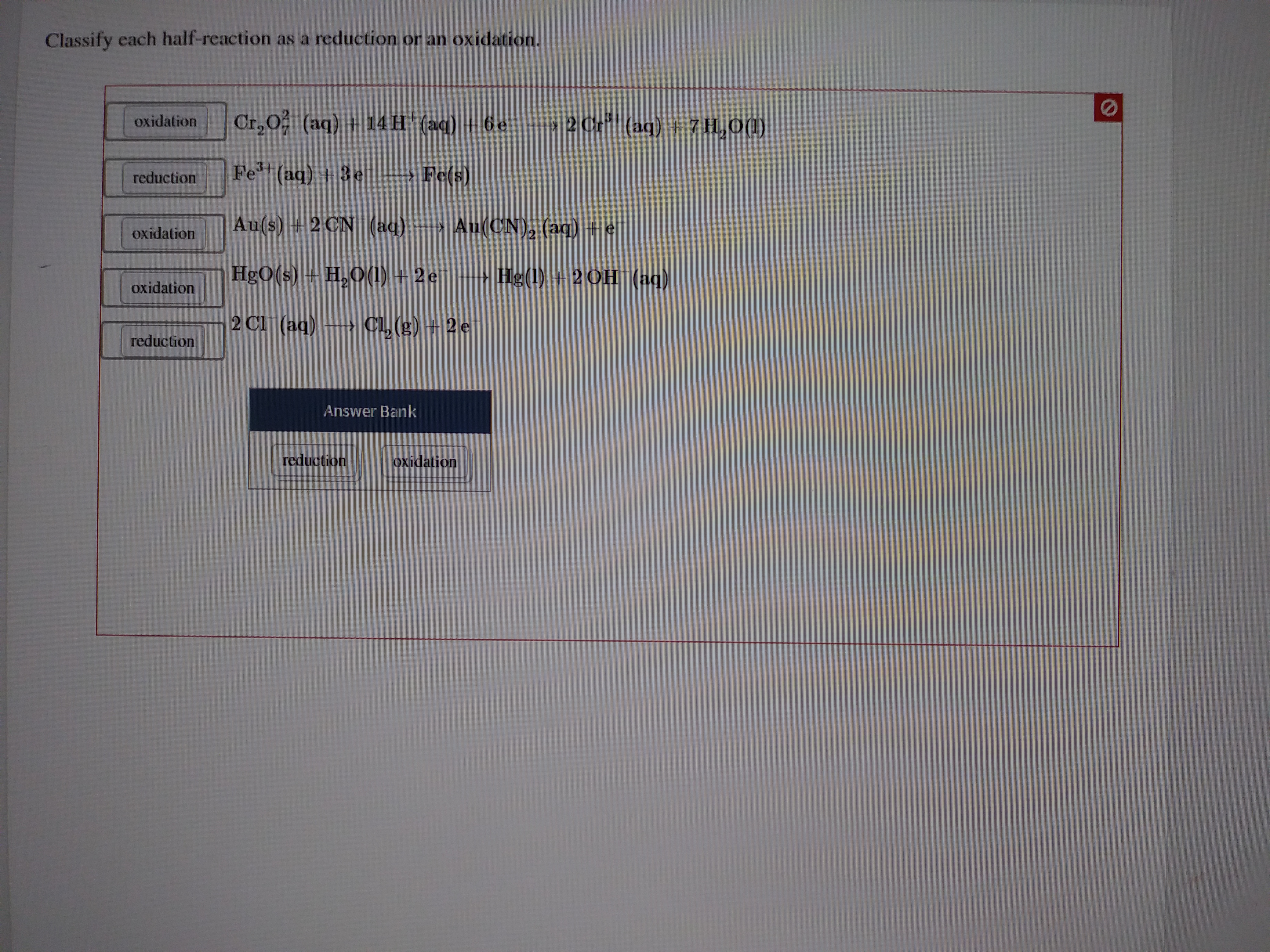
Classify Each Half-reaction As A Reduction Or An Oxidation.
Ever seen those vibrant, intricate patterns that seem to dance across a canvas or a piece of pottery? Or maybe you've admired the way light plays off a beautifully crafted metal sculpture. These aren't just random splashes of color or accidental shaping; they often stem from a fascinating and surprisingly accessible scientific process: classifying half-reactions as reduction or oxidation! It might sound like something reserved for a chemistry lab, but this concept, in its artistic application, is a realm of pure creative magic, offering a unique way to imbue your projects with incredible depth and visual appeal. Think of it as painting with electrons – a truly electrifying art form!
So, what's in it for you, whether you're a seasoned artist, a curious hobbyist, or just someone looking for a new and inspiring way to engage your creativity? For artists, understanding the principles of oxidation and reduction can unlock entirely new palettes and textural possibilities. Imagine creating patinas on metal that shift from deep blues to earthy browns, or developing glazes for ceramics that exhibit stunning color variations based on these chemical reactions. Hobbyists can explore DIY projects like creating their own "verdigris" effects on decorative items or experimenting with conductive inks for electronic art. Even casual learners will find immense satisfaction in demystifying a scientific concept and seeing it manifest in tangible, beautiful creations. It’s a fantastic way to bridge the gap between science and art, making both more engaging and understandable.
The beauty of applying this concept lies in its versatility. In terms of styles, think of the ancient art of bronze patination, where controlled oxidation creates those sought-after aged looks. Or consider the vibrant, often iridescent finishes achieved in enameling, which relies on controlled heating and cooling of metal oxides. Subject-wise, the applications are limitless. You can create abstract pieces that play with color gradients and chemical reactions, or perhaps more representational works where the texture and finish contribute significantly to the subject's mood and character. Variations include experimenting with different metals, chemical agents, and heat treatments to achieve a spectrum of unique effects. Some artists even incorporate these principles into mixed-media pieces, combining painted elements with chemically altered surfaces.
Ready to dip your toes into this captivating world? Trying it at home can be surprisingly straightforward. For a simple introduction, consider working with copper. You can purchase copper sheets and experiment with different household acids like vinegar or lemon juice, perhaps with the addition of salt, to induce oxidation. Remember to work in a well-ventilated area and wear appropriate safety gear, such as gloves and eye protection. For a slightly more advanced project, explore using commercially available patinas. Always follow the product instructions carefully. Even observing how a piece of iron rusts over time is a fundamental example of oxidation in action!
Ultimately, the joy of exploring oxidation and reduction in creative pursuits lies in the element of surprise and discovery. It’s a process where you collaborate with chemistry, allowing the materials to speak and reveal their hidden potentials. There’s a profound satisfaction in witnessing a chemical transformation unfold before your eyes, resulting in something uniquely beautiful and entirely your own. It’s a reminder that even the most complex scientific principles can lead to the most elegant and inspiring artistic outcomes.
