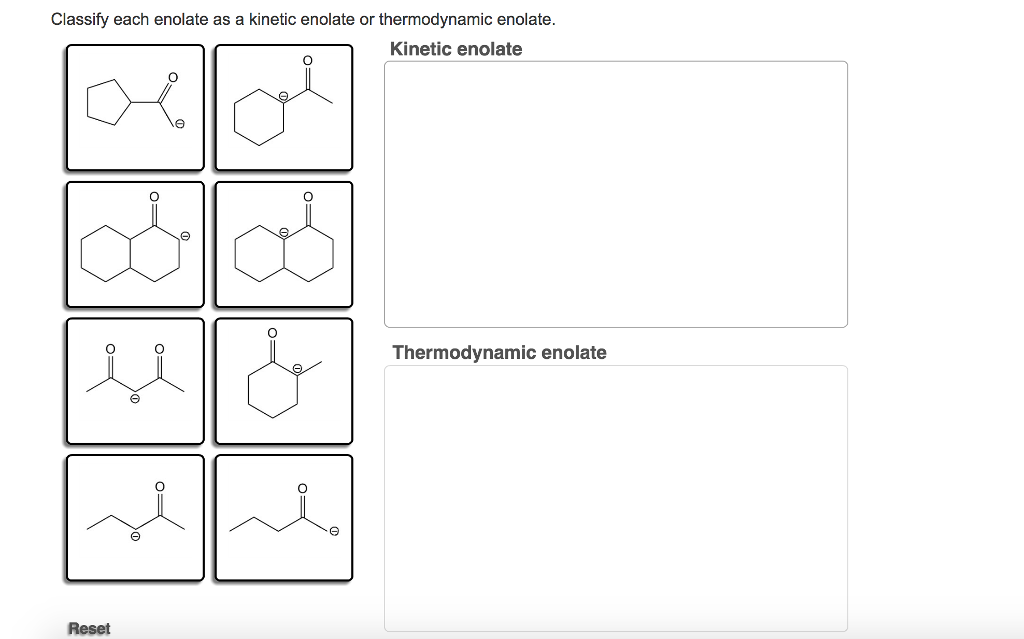
Classify Each Enolate As A Kinetic Enolate Or Thermodynamic Enolate.
You know, I used to have this neighbor, bless her heart. She was obsessed with making the perfect pie crust. Every single time, she’d go through this whole elaborate ritual, meticulously measuring, chilling, and then, at the crucial moment, she’d swear that a slightly warmer, more pliable dough yielded a flakier crust. I, on the other hand, always went for the super-chilled, almost crumbly dough, convinced that that was the secret to crispiness. We’d have these friendly little debates over the fence, and honestly, neither of us was wrong. We were just approaching the same goal from different angles, prioritizing different things.
And that, my friends, is exactly what we’re going to dive into today, but for the world of organic chemistry! We’re talking about enolates, those fascinating, reactive intermediates that pop up when you deprotonate a carbonyl compound. And just like my neighbor and her pie crusts, there are different ways to make an enolate, and which way you choose matters. Today, we’re going to classify enolates as either kinetic or thermodynamic. Don’t worry, it’s not as scary as it sounds, and by the end of this, you’ll be able to spot them like a seasoned baker spotting a perfectly golden-brown crust!
So, what exactly is an enolate? Imagine a carbonyl group – that’s a carbon double-bonded to an oxygen, like in a ketone or an aldehyde. Now, if you have a strong enough base, it can snatch a hydrogen atom from the carbon next door to the carbonyl (we call this the alpha-carbon). When that hydrogen leaves, it takes its electrons with it, and those electrons can then rearrange to form a double bond between the alpha-carbon and the carbonyl carbon. The oxygen then picks up a negative charge. Voila! You’ve got yourself an enolate. It’s like a chemical chameleon, ready to react!
Now, the crucial part is that this alpha-carbon can have two hydrogens attached. And depending on which hydrogen gets snatched and under what conditions, you can end up with two different enolate structures. This is where our kinetic and thermodynamic friends come into play. Think of it as choosing between speed and stability, or perhaps, between a quick fix and a long-term investment.
The Speedy, Slightly Less Stable One: The Kinetic Enolate
Let’s start with the kinetic enolate. The name itself gives us a hint, right? "Kinetic" is all about movement, about speed. When we're aiming for a kinetic enolate, we want the reaction to happen fast, and we don't really care too much if the resulting enolate is the absolute most stable thing in the universe. It’s like grabbing the first ripe strawberry you see – it’s easy, it’s quick, and you get a strawberry!
How do we achieve this speedy enolate formation? We use a strong, sterically hindered base. Think of bases like Lithium Diisopropylamide (LDA). LDA is your ultimate battering ram of a base. It’s bulky, meaning it has a hard time getting into tight spaces. Because it's so bulky, it prefers to grab the easiest hydrogen to remove, which is usually the one on the alpha-carbon that has fewer substituents. This means if you have a ketone with an alpha-carbon that has one hydrogen and another alpha-carbon that has two hydrogens, LDA will go for the one with just one hydrogen. Easy peasy, lemon squeezy!
The other key condition for a kinetic enolate is low temperatures. Why? Because at low temperatures, the reaction has less energy to overcome any activation barriers. The base just goes for the path of least resistance, the one that requires the least energy to get going. It’s like trying to get out of bed on a chilly Monday morning – you want the quickest route to coffee, not a leisurely stroll.
So, when you have a ketone like 2-butanone, which has two different alpha-carbons, one with one hydrogen and one with two: the LDA at low temperature will preferentially remove the hydrogen from the carbon with fewer hydrogens. This results in an enolate where the double bond is formed between the carbonyl carbon and the alpha-carbon that originally had fewer hydrogens. This enolate might be formed faster, but it’s often less substituted at the double bond compared to its thermodynamic counterpart.
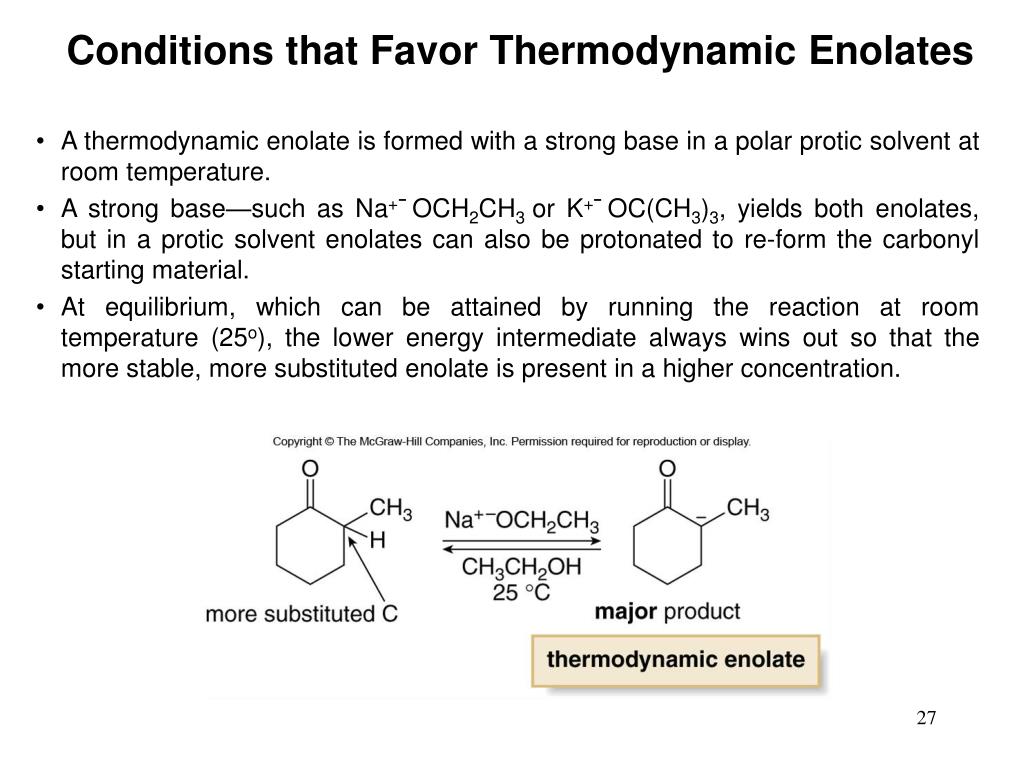
It's all about rate control. We're controlling the rate at which the enolate is formed. The kinetic product is the one that forms fastest. And fast often means taking the most accessible route. Think of it as a race where the runner takes the shortest, most direct path, even if it’s a little less scenic.
The irony here, of course, is that the kinetic enolate is often the less thermodynamically stable one. Less stable? Yes! But formed faster! It’s like having a quick snack that isn’t super satisfying but tides you over in a pinch. You’re getting the enolate you want now, and you can worry about its long-term stability later, or perhaps the next step in the reaction will stabilize it anyway.
So, to recap the kinetic enolate: use a strong, bulky base and low temperatures. This leads to the enolate formed via the fastest reaction pathway, typically removing the less substituted alpha-hydrogen. It’s all about getting that enolate out of the gate as quickly as possible!
The Slow, But Worth It One: The Thermodynamic Enolate
Now, let’s switch gears to the thermodynamic enolate. If kinetic is about speed, thermodynamic is about stability. We want the most stable enolate possible, even if it takes a little longer to get there. This is the pie crust that’s been chilled perfectly, the one that’s been lovingly and patiently prepared. It might take more effort, but you know it’s going to be good.
How do we coax out this super-stable enolate? We use a less hindered base, and often, we use protic solvents or higher temperatures. Think of a base like sodium hydroxide or potassium hydroxide. These are strong bases, but they’re not as bulky as LDA. They’re more willing to hang around and wait for the most energetically favorable outcome.
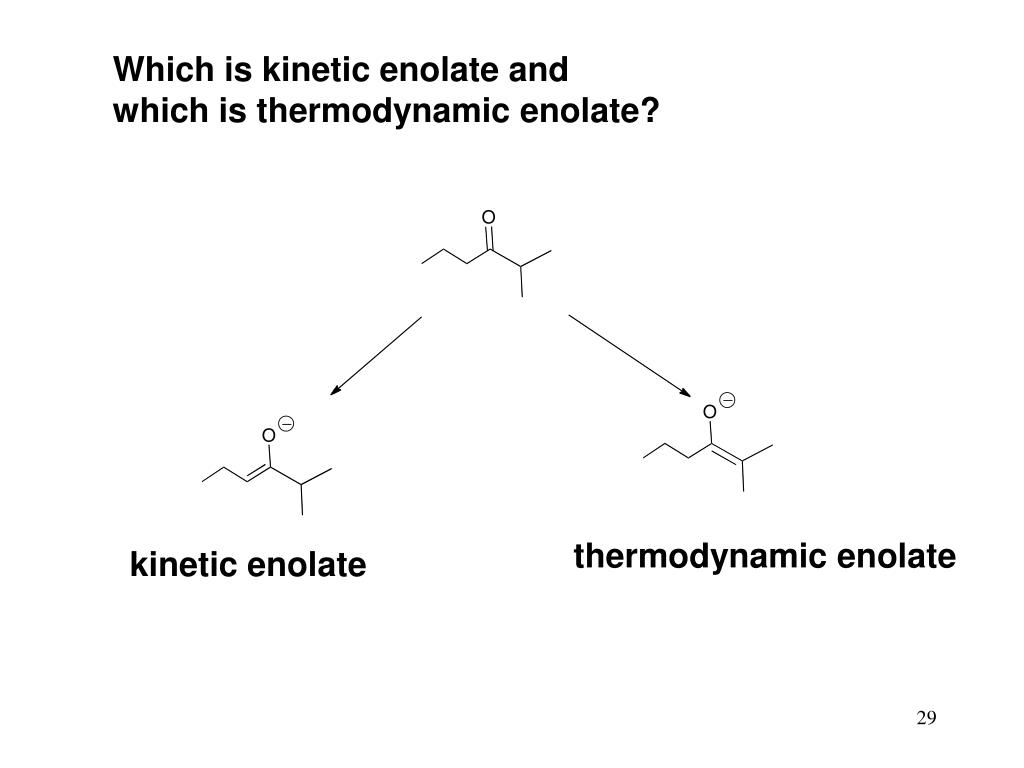
The key here is equilibrium. We want the enolate formation to reach equilibrium, where both the starting material and the enolate can interconvert. At equilibrium, the most stable species will be favored. And which enolate is usually the most stable? The one where the double bond is formed to the alpha-carbon that is more substituted. This is because, generally, more substituted double bonds are more stable due to hyperconjugation.
So, going back to our 2-butanone example: if we use a less hindered base and allow the reaction to reach equilibrium, the base will eventually abstract a hydrogen from either alpha-carbon. However, the resulting enolate where the double bond is formed to the more substituted alpha-carbon (the one that originally had two hydrogens) will be the more stable one. And at equilibrium, this more stable enolate will be present in higher concentrations.
This is why higher temperatures can favor the thermodynamic enolate. At higher temperatures, molecules have more energy, and they can overcome the energy barriers to rearrange and reach the most stable state. It’s like letting a complex puzzle sit for a while – eventually, the pieces will settle into their most stable configuration.
The thermodynamic enolate is the product that is thermodynamically favored. It's the one that has the lowest energy, the most stable structure. Think of it as the perfectly aged cheese – it took time, but the flavor is rich and complex!
The beauty of the thermodynamic enolate is that it's usually the more substituted enolate. More substitution at the double bond often means greater stability. It's like having a well-decorated room versus a sparsely furnished one – the well-decorated one feels more complete and stable.
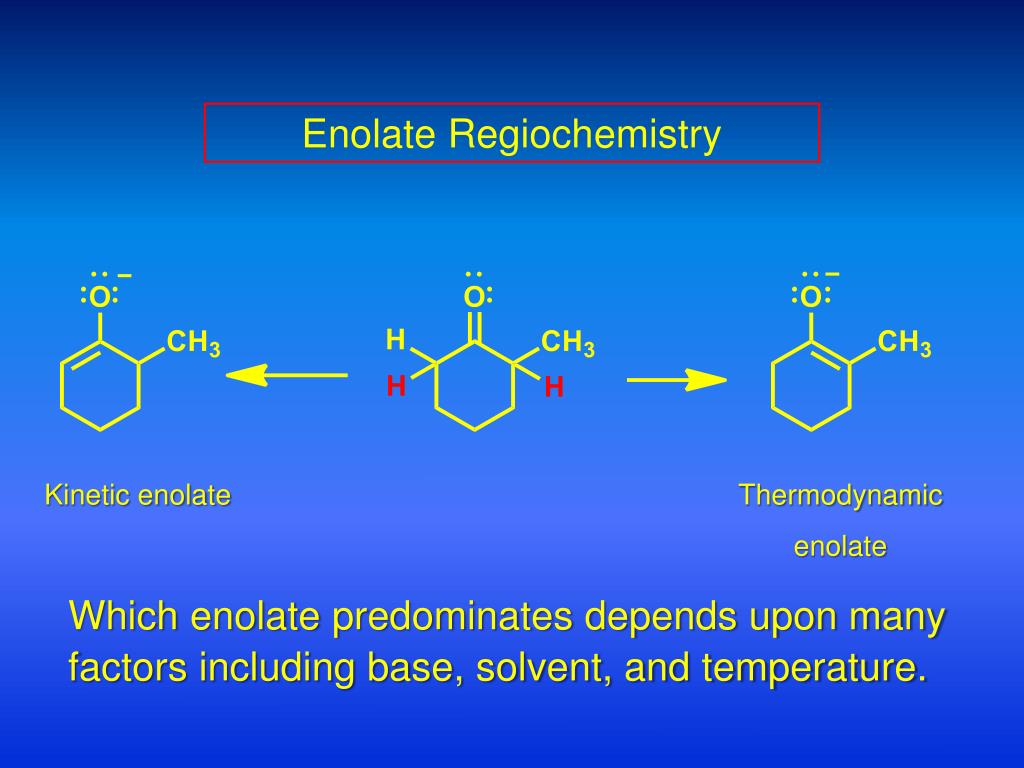
So, to sum up the thermodynamic enolate: use a less hindered base, and allow the reaction to reach equilibrium, often by using higher temperatures or protic solvents. This leads to the enolate that is thermodynamically most stable, typically the more substituted enolate. It’s all about getting the best, most stable outcome, even if it’s a slower process.
Putting It All Together: Spotting the Difference
Now, how do you actually classify a given enolate? It’s like looking at a painting and deciding if it’s abstract expressionism or photorealism. You look for the clues!
First, consider the base. Is it a strong, bulky base like LDA? If yes, lean towards kinetic. Is it a strong but less hindered base like NaOH or KOH, or perhaps an alkoxide? If yes, lean towards thermodynamic.
Next, consider the temperature. Is the reaction being run at very low temperatures (like -78°C)? If so, kinetic is the likely player. Is it being run at room temperature or higher, or is there mention of allowing the reaction to reach equilibrium? Then, thermodynamic is probably in charge.
Finally, look at the structure of the potential enolates. If you have a choice between forming an enolate with a double bond to a monosubstituted alpha-carbon versus a disubstituted alpha-carbon, which one is likely to form faster under kinetic conditions? The one with fewer substituents on the alpha-carbon. Which one is likely to be more stable under thermodynamic conditions? The one with more substituents on the alpha-carbon.
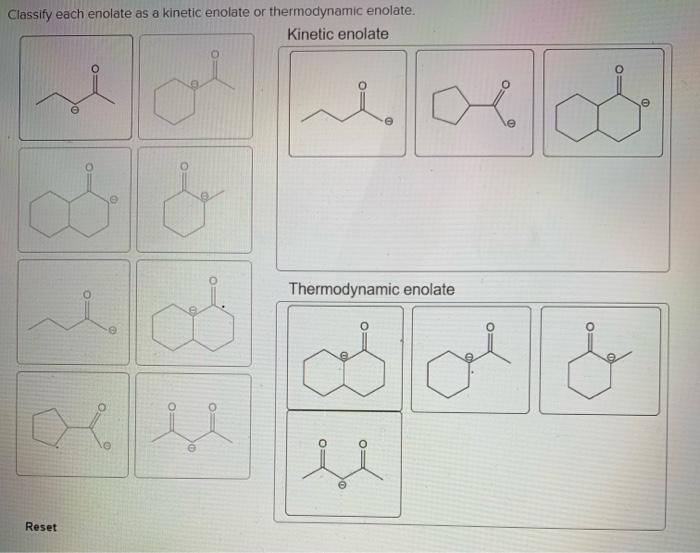
Let's try a quick mental exercise. Imagine a ketone with an alpha-carbon bearing one hydrogen and another alpha-carbon bearing two hydrogens. If you're told the base used is LDA and the temperature is -78°C, what kind of enolate are you most likely forming? That’s right, a kinetic enolate! The LDA, being bulky and the low temperature, forces the abstraction of the less hindered hydrogen.
Now, imagine the same ketone, but this time the base is sodium ethoxide, and the reaction is allowed to proceed at room temperature. What are you thinking now? Probably a thermodynamic enolate! The less hindered base and room temperature favor the formation of the more substituted, more stable enolate.
It's not always black and white, of course. Sometimes, the difference in stability between the two potential enolates isn't huge, and the reaction conditions can be a bit ambiguous. But understanding these fundamental principles – the role of the base, the temperature, and the inherent stability of the resulting enolate structure – will give you a very strong basis for classification.
Think of it this way: my neighbor’s slightly warmer dough might still result in a decent pie crust, and my super-chilled dough might sometimes be a bit too crumbly. There's a spectrum! But in chemistry, we like to categorize. And when it comes to enolates, the kinetic versus thermodynamic distinction is a really powerful way to predict the outcome of reactions and understand the underlying mechanisms. It helps us chemists make informed decisions, just like my neighbor made informed decisions about her pie crust!
So, the next time you encounter an enolate, don't just see it as a negative charge on an oxygen and a double bond. Ask yourself: was this enolate born of haste or of patience? Was it a quick grab for the easiest hydrogen, or a slow, deliberate pursuit of ultimate stability? The answer will tell you a lot about its character and what it’s likely to do next!
Keep practicing, keep observing, and you’ll become a pro at spotting these enolate personalities. And who knows, maybe you’ll even start a friendly debate over the fence about which enolate is truly superior!
