Classify Each Element As A Metal Nonmetal Or Semimetal
Hey there, curious minds! Ever looked at something shiny and wondered what it's made of? Or maybe you've seen those cool gadgets and thought, "How do they even work?" Well, buckle up, because we're about to dive into the absolutely fascinating, and dare I say, fun, world of classifying the elements. Yep, those building blocks of everything around us – from the sparkle in your jewelry to the very air you breathe!
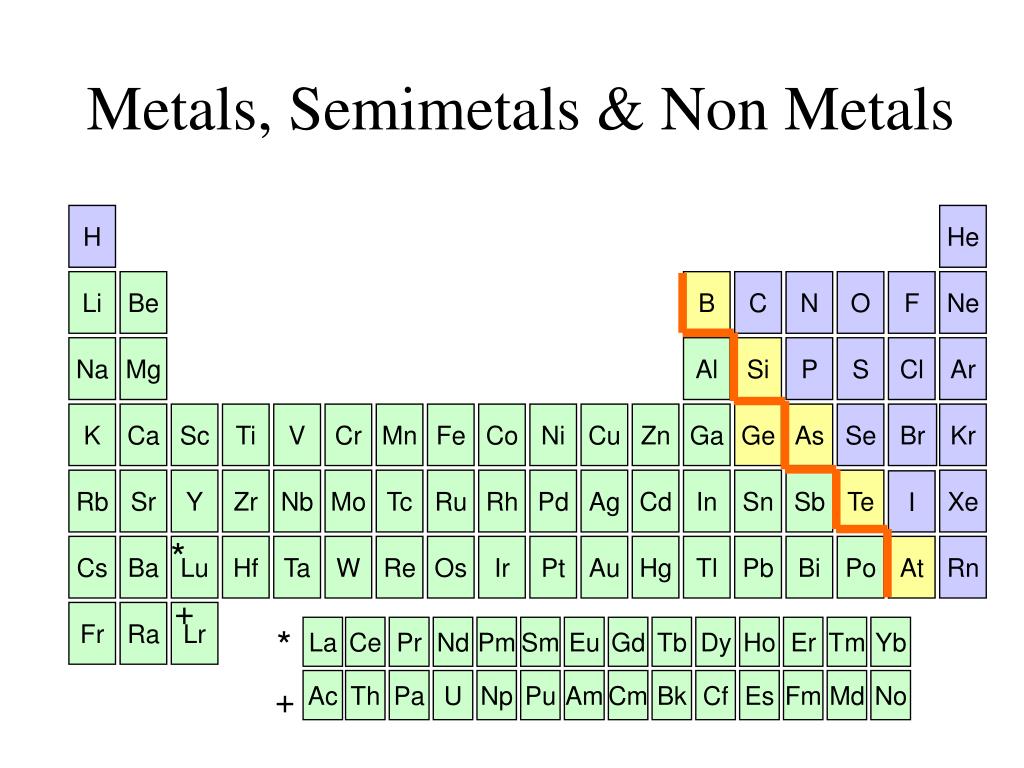
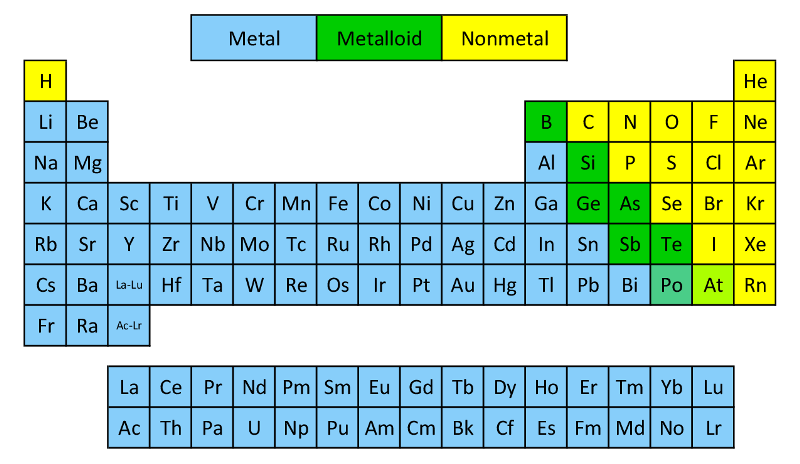
Now, the periodic table can look a bit intimidating at first, right? All those letters and numbers. But imagine it as a super-organized party guest list. And today, we're playing a fun game of "Who's Who?" We're going to sort these elements into three awesome categories:
Metals: The Shiny Stars of the Show!
Let's start with the crowd-pleasers, the absolute superstars: the
And they're super
Think about your everyday life. Your car? Lots of metal. Your coins? Definitely metal. That super-strong bridge you drive over? Yep, metal. They're the backbone of so much of our technology and infrastructure. They're the reliable, sturdy friends who are always there, doing their essential jobs without a fuss. And honestly, who doesn't love a bit of sparkle? That's the metals for you!

Nonmetals: The Unsung Heroes!
Now, let's talk about the
Others are brittle solids, like
Nonmetals might not be as flashy as metals, but they are absolutely crucial. They make up a huge part of our atmosphere, our water, and our bodies. They're the quirky, creative types of the element world, bringing a different kind of essential magic to the mix. They're the artists, the thinkers, the ones who add variety and depth to the whole party.

Semimetals: The Versatile Villains (or Heroes?!)
And then, we have the fascinating
For example, semimetals can sometimes conduct electricity, but not as well as metals. This makes them perfect for electronics! Think about your computer, your phone, your gaming console – they all rely on semimetals like
They can be shiny or dull, hard or brittle. It all depends on how you treat them! This adaptability is their superpower. They’re the ultimate team players, able to switch up their game to fit the situation. They are the engineers and the negotiators of the element party, finding ways to make things work beautifully.
Why Does This Matter? It's Actually Super Cool!
So, why all this talk about classifying elements? Because understanding these categories helps us understand the world around us in a whole new light! When you know that something is a metal, you expect it to be conductive. When you know it's a nonmetal, you might expect it to be less dense or to form a gas. And when you encounter a semimetal, you know you're dealing with something with unique potential.
This knowledge unlocks a deeper appreciation for the materials we use every day. It’s like learning a secret code that explains how things work. It's the difference between just seeing a rock and knowing it's a specific type of mineral with unique properties. And that, my friends, is incredibly empowering and, yes,

The periodic table isn't just a chart in a science textbook; it's a roadmap to the universe's ingredients. Each element has its own personality, its own role to play. And by understanding their classifications, we can start to see the incredible ingenuity and diversity that nature has to offer.
So, the next time you pick up something, take a moment to consider what it might be made of. Is it shiny and conductive like a metal? Is it essential for breathing like a nonmetal? Or is it a versatile connector like a semimetal? The possibilities are endless, and the more you learn, the more the world will open up to you.
Don't be shy about exploring further! The world of elements is a vast and exciting playground, full of wonders waiting to be discovered. Every question you ask, every little bit you learn, is a step towards a more fascinating and inspired understanding of everything. So go forth, be curious, and let the elements surprise you!
