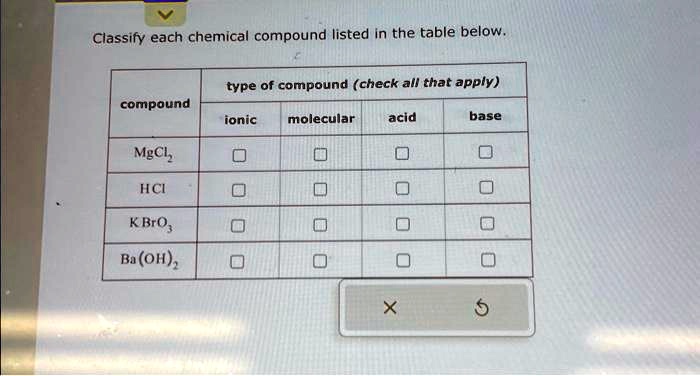
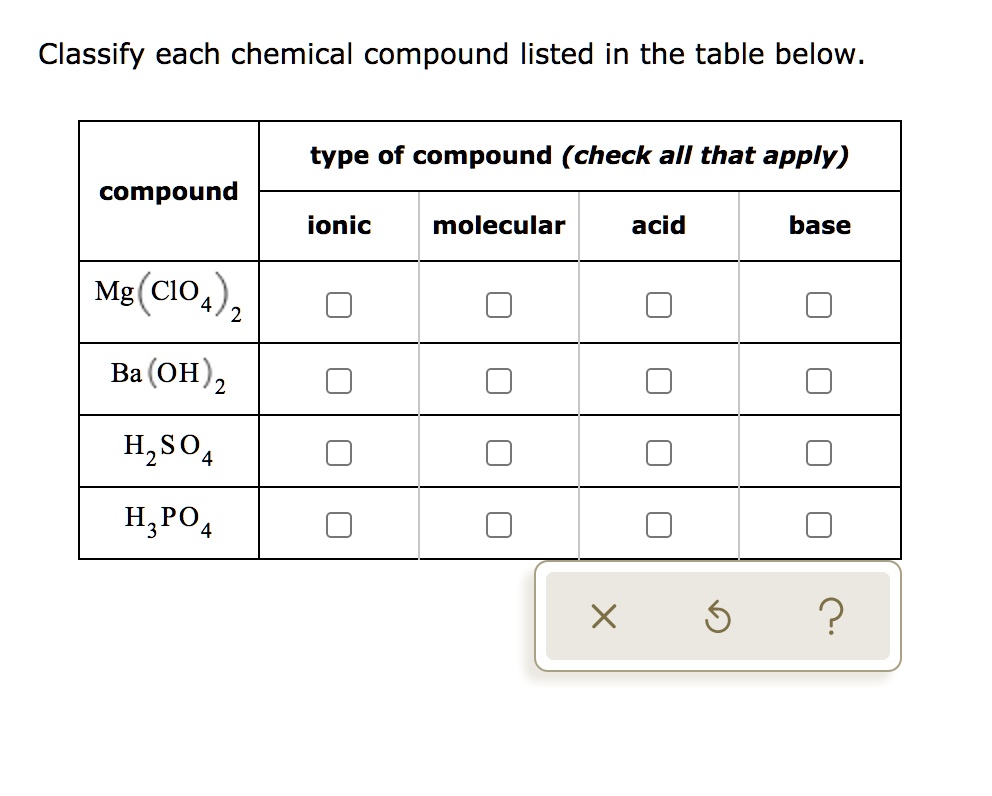
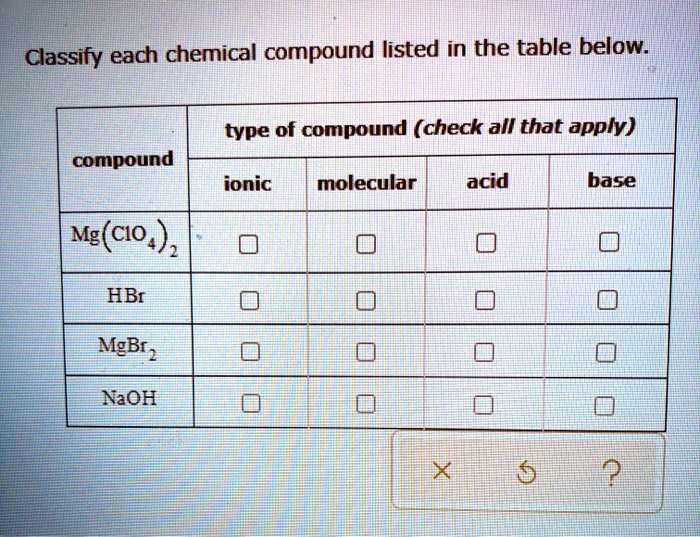
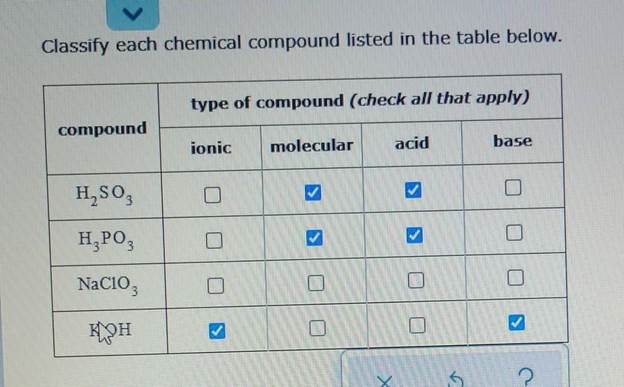
Classify Each Chemical Compound Listed In The Table Below
Hey there, lovely people! Ever look around your kitchen or the beauty products on your bathroom shelf and wonder what all those fancy-sounding ingredients actually are? It can feel like a secret code sometimes, right? Well, today, we're going to crack a little bit of that code together in a way that's super chill and won't make your brain hurt. Think of it like getting to know your everyday ingredients a bit better – like recognizing your favorite neighbor on the street!
We're going to be chatting about classifying chemical compounds. Now, "chemical compound" sounds a bit lab-coat-y, but really, it's just a fancy term for two or more different kinds of atoms stuck together. Like LEGO bricks! You can have different colors and shapes of LEGOs, and when you snap them together, you get something new. Chemical compounds are just like that, but with teeny-tiny atoms.
Why should you care? Because understanding these basic groupings helps us understand so much of the world around us! From the food we eat to the medicines that help us feel better, knowing these classifications is like having a little peek behind the curtain. It makes things less mysterious and, dare I say, a bit more fascinating.
Let's Meet the Main Players: The Big Families of Compounds
Scientists love to sort things into groups, and chemical compounds are no exception. It makes it easier to study them and predict how they'll behave. Think about sorting your socks – you probably have your everyday socks, your cozy fuzzy ones, and maybe even those fancy dress socks. Each type has its own job, right? Chemical compounds have their own "families" too, and each family has its own special characteristics.
The Salty Bunch: Salts
When you hear "salt," you probably think of the stuff you sprinkle on your fries. And you're not wrong! Table salt, or sodium chloride (NaCl), is a super common salt. But in chemistry, a salt is a much bigger group. Generally, salts are formed when an acid and a base have a little chat and decide to team up. It's like when your sweet and sour candy has that perfect balance – salts often have a slightly salty taste, but they can also be bitter or even tasteless. They're often made up of a metal atom and a non-metal atom joined together.
Think about baking soda, which is sodium bicarbonate (NaHCO₃). That's a salt too! It helps your cookies rise and can even help clean your sink. Or how about Epsom salts, magnesium sulfate (MgSO₄), that you might soak your tired muscles in? Those are all part of the salt family, each with its own special talents. They're the workhorses of the chemical world, popping up in everything from food preservation to fertilizers.
The Zesty Crew: Acids
Now, let's talk about acids. When you think of an acid, you might imagine something that fizzes or has a sour taste. Lemons? Definitely acidic! That's because they contain citric acid. Vinegar? That sour sting is from acetic acid. Acids are known for their ability to donate a hydrogen atom, which gives them their characteristic tartness and their ability to react with things.
Acids are like the energizer bunnies of the chemical world. They love to react! They can dissolve metals (carefully, of course – don't go pouring acid on your car keys!), and they are essential for many biological processes. Your stomach acid, mostly hydrochloric acid (HCl), is what helps break down your food. Without it, digestion would be a whole lot harder. They often have a sharp, sometimes even biting, taste, and can feel slippery to the touch in dilute solutions.

The Smooth Operators: Bases
Opposite to acids, we have bases. If acids are the zesty ones, bases are more like the smooth, calming operators. They tend to feel a bit slippery, like soap. Think about dish soap – it's a base, and it helps cut through grease, right? That slippery feel is a big clue you're dealing with a base.
Bases, like sodium hydroxide (NaOH) – also known as lye, which is used in soap making – often have a bitter taste and can feel quite different from acids. They are great at neutralizing acids. Remember how we talked about acids and bases teaming up to form salts? That's the neutralization party happening! Bases are all around us, from cleaning products to the things that help regulate the pH of our blood, keeping us healthy.
The Watery Wonders: Oxides
Okay, let's not get too bogged down in the technicalities, but another important group are oxides. These are compounds that contain oxygen and at least one other element. Think of rust on your bike – that's iron oxide! It's basically iron that has reacted with oxygen. It's a natural process, but it shows how readily oxygen likes to combine with other things.
Water itself (H₂O) is technically an oxide, but it's such a special and vital compound that it often gets its own category. But many other oxides are important too. For example, carbon dioxide (CO₂) is an oxide of carbon. It’s what we breathe out, and plants use it to grow. So, even though "oxide" sounds a bit plain, these compounds play huge roles in our environment and in our bodies.
Why Does This Matter to You?
You might be thinking, "This is interesting, but why should I, a person who just wants to make a nice cup of tea and not have a science lecture, care?" Well, here's the thing: these classifications aren't just for scientists in white coats. They help us understand the world we live in.
When you pick up a bottle of antacid to settle your tummy after a spicy meal, you're using a base to neutralize excess stomach acid. When you choose a cleaning product, knowing whether it's acidic or basic can help you understand how it works and if it's the right tool for the job. Even the vitamins you take are often in the form of salts or other compounds that are easily absorbed by your body.
It's about making informed choices. It's about appreciating the chemistry that makes life possible. Think about how different foods taste – the sourness of a grapefruit (acid!), the saltiness of cheese (often containing salts!), the way baking soda helps your cake fluff up (a base reacting with acids in the batter). It's all chemistry!
So, the next time you see a chemical name on a label, don't let it intimidate you. Try to remember these basic families. Is it likely an acid, a base, or a salt? This little bit of knowledge can be like a superpower, helping you understand why things happen the way they do, from your cooking to your cleaning to how your own body functions. It’s a fun way to connect with the everyday science that surrounds us, and honestly, it makes the world a little more predictable and a lot more interesting!
