Classify Each Characteristic As Either A Metal Or A Nonmetal.

Alright, settle in, grab your latte, and let's dive into the wacky world of elements. You know, those building blocks of everything from your fancy smartphone to that slightly questionable cheese you found in the back of the fridge. Today, we're playing a game called "Metal or Not Metal?" It's like elemental bingo, but way cooler and with fewer itchy bingo dabbers.
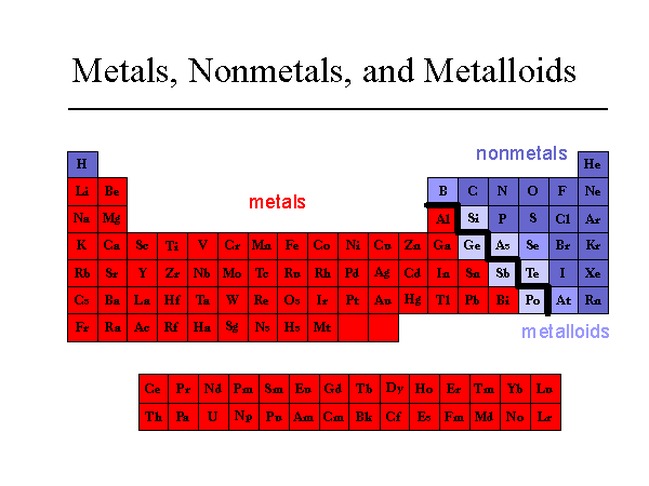
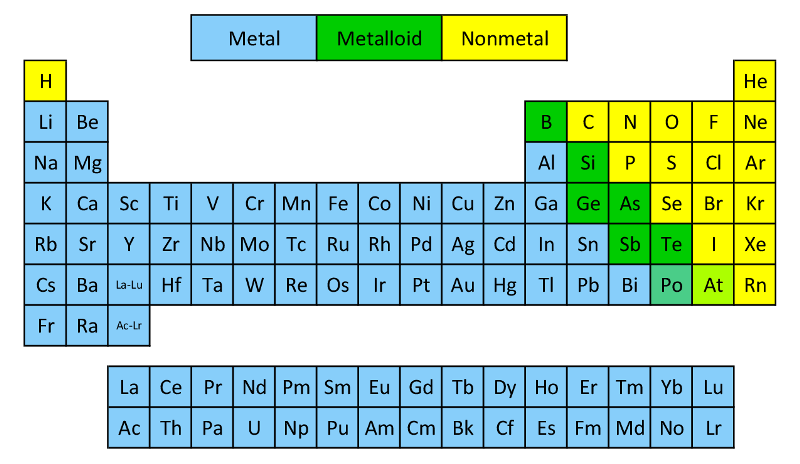
So, we've got two main camps in the periodic table, and it’s a bit like a high school cafeteria. On one side, you have the Metals. They’re the jocks, the popular kids, the ones who always seem to be shining and… well, metal-y. On the other side, you have the Nonmetals. These guys are a bit more diverse. Some are gases, some are solids, some are even… liquids. They’re the artists, the introverts, the ones who might make your breath smell like rotten eggs, but hey, they’re essential!
Let's start with our metallic friends. What’s the first thing you think of when you hear "metal"? Probably something shiny, right? That’s because metals are typically lustrous. Think of a knight in his gleaming armor, or a freshly polished spoon. They’re like nature’s disco balls. They love to reflect light, and frankly, they know it. They strut their stuff, beaming with pride.
Now, these metallic titans are also incredibly malleable and ductile. This is where things get fun. Malleable means you can hammer them into thin sheets. Think of gold leaf on a fancy cake – that’s some seriously hammered gold! Ductile means you can stretch them into wires. That’s how we get all those wires in your electronics, zipping information around like tiny electrical superheroes. Imagine trying to hammer oxygen into a sheet or stretch it into a wire. You’d just end up with… well, nothing but air. Bless its non-metallic heart.
Another biggie for metals is that they’re generally good conductors of heat and electricity. This is why your pan gets hot when you put it on the stove, and why you can charge your phone without electrocuting yourself (usually). They’re like little energy highways, letting heat and electricity zoom through them. Nonmetals, on the other hand? Not so much. They’re more like energy speed bumps, or sometimes, complete energy dead ends.

And then there’s the whole state of matter thing. At room temperature, most metals are solids. Big, strong, solid blocks of metallic awesomeness. There’s one notable exception that likes to break the rules – mercury. Yeah, that silvery liquid stuff you used to see in old thermometers. It's basically a metal that decided it was too cool for solids and opted for a more fluid lifestyle. Talk about a rebel!
Now, let’s swing over to the nonmetal side of the cafeteria. These guys are a bit of a mixed bag. For starters, they’re usually dull in appearance. No flashy disco balls here, folks. They’re more likely to be powdery, crumbly, or just… invisible (if they’re gases). Imagine your nail file – that’s probably made of something nonmetallic. It’s not exactly a beacon of light, is it?
When it comes to shaping them, forget it. Nonmetals are brittle. If you try to hammer them, they’ll shatter like your grandma’s finest china when you accidentally drop it. And stretching them into wires? You’d have better luck teaching a cat to tap dance. They just don’t have that flexible spirit.
And the conductivity? Big fat NO. Nonmetals are generally poor conductors of heat and electricity. They’re the insulators, the blockers, the ones who say, "Nope, no energy allowed through here!" This is why your oven mitts are made of nonmetal materials – to stop that heat from reaching your delicate, non-heat-resistant hands. Thank you, nonmetals, for saving us from third-degree burns.
The state of matter for nonmetals is a party of three. You’ve got your gases, like oxygen (thank goodness for that!) and nitrogen, which make up most of the air we breathe. Then you have your solids, like carbon (hello, diamonds and pencils!) and sulfur. And don’t forget the one liquid nonmetal, bromine. Bromine is a bit of a spooky one, a reddish-brown liquid that fumes nastily. Definitely not a swimming pool option.

Let’s throw in some more characteristics to solidify our understanding, pun intended. Metals tend to have high melting and boiling points. They’re tough cookies, requiring a lot of energy to change their state. Think of melting down steel – it takes some serious heat! Nonmetals, on the other hand, often have low melting and boiling points. That’s why oxygen is a gas at room temperature. Easy peasy transformation for them.
Here’s a fun one: metals tend to lose electrons when they react. They’re like generous donors, happily handing out electrons to other atoms. Nonmetals, conversely, tend to gain or share electrons. They’re the takers and the sharers, the ones who are a bit more clingy with their electron buddies. This electron-trading business is what makes them bond and form all sorts of cool compounds.
And what about density? Metals are usually quite dense. They’re heavy for their size. Think of a lead weight versus a balloon. The lead is dense, the balloon is… not. Nonmetals can be dense, but many are quite light, especially the gases.
Let’s do a quick recap with some fun examples. Is that shiny doorknob you’re about to turn? Definitely a metal – lustrous, probably solid, and a good conductor if you’ve left the electricity on (don’t do that). Is that puff of air you just exhaled? Nonmetal – a gas, dull, and definitely not conducting electricity. Is that diamond ring on your finger? Nonmetal – a solid form of carbon, incredibly hard, but a terrible conductor of electricity (which is why diamond-studded circuit boards are a thing of the future, not the past!). Is that copper wire in your phone charger? You guessed it – metal! Ductile, lustrous, and a fantastic conductor.
So, next time you’re looking around, play this game! Is it shiny? Malleable? A good conductor? Chances are, it’s a metal. Is it dull? Brittle? A bad conductor? Probably a nonmetal. Remember mercury and bromine, our liquid outliers, and don’t forget that metals are generally solids while nonmetals can be solids, liquids, or gases. It's like a secret code to the universe, and now you've got the decoder ring. Go forth and classify, my friends!
