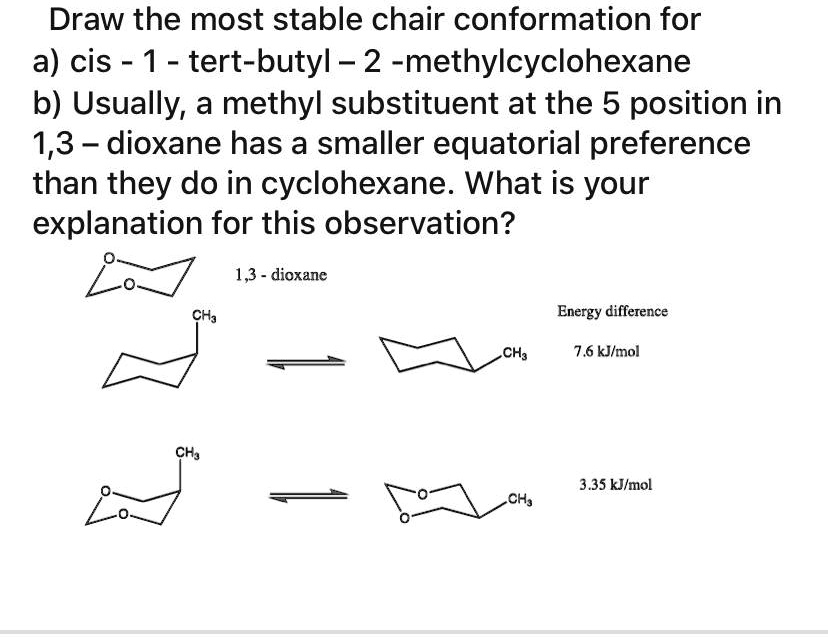
Cis 1 Tert Butyl 2 Methylcyclohexane Chair Conformation
Hey there, fellow humans and molecule enthusiasts (even if you didn't know you were one)! Ever found yourself staring at a perfectly organized pile of laundry and feeling a strange sense of satisfaction? Or maybe you’ve wrestled with getting that awkwardly shaped pillow into a standard pillowcase? Well, get ready, because we’re about to dive into something that, believe it or not, shares a surprisingly similar vibe: cis 1 tert-butyl 2 methylcyclohexane chair conformation.
Now, I know what you’re thinking. "Chair conformation? Is this some kind of extreme armchair travel for chemists?" Not quite! But stick with me, because understanding this little molecular dance can actually be pretty fun and, dare I say, useful.
What's in a Name? (Besides a Mouthful!)
Let's break down this mouthful of a name. Think of it like a secret code for a specific molecule.
First, we have cyclohexane. Imagine a tiny, perfect hexagon made of carbon atoms, all linked together. That's our basic ring. Now, this ring isn't flat and boring like a frisbee. Nope, it’s a bit more… flexible. Think of a folding lawn chair – it has bits that stick up and bits that point down. That's kind of what cyclohexane does, in what we call its chair conformation. It’s the most stable and comfy way for that six-carbon ring to arrange itself in space. It’s like the molecule’s favorite way to kick back and relax.
Then we have the “1 tert-butyl 2 methyl” part. This tells us what's attached to our cyclohexane ring.
Imagine your cyclohexane chair. Now, at the "1" position, we're sticking on a tert-butyl group. This is like a really bulky, three-dimensional blob. Think of a big, round cushion you’d put on your favorite armchair. It takes up a lot of space!
At the "2" position, we're attaching a methyl group. This is a much smaller, simpler group. Think of a small decorative tassel on the edge of that same armchair.
The "Cis" Conundrum: Together or Apart?
Now, here’s where things get interesting, and where the "cis" comes into play. "Cis" is a fancy way of saying that our two attachments – the bulky tert-butyl and the smaller methyl – are on the same side of the cyclohexane ring.
Imagine you’re looking at your armchair from the side. If both the big cushion (tert-butyl) and the small tassel (methyl) are sticking up, or both are pointing down, that’s cis. They’re on the same team, so to speak. If one was up and the other was down, that would be "trans" – they’d be on opposite sides.

The Chair's Comfy Positions: Axial and Equatorial
Back to our chair! Remember how the cyclohexane ring is like a folding chair? It has two main kinds of positions for things to stick out from:
- Axial: These are the positions that stick straight up or straight down, like the legs of the chair. They’re pretty much parallel to the ring’s imaginary axis.
- Equatorial: These are the positions that stick out more to the side, sort of in a flat plane around the ring. Think of where you’d put your drink on a side table attached to your armchair.
Now, here's the key to our story. Bulky things, like our big tert-butyl cushion, really don’t like being in the axial positions. Why? Because they get cramped! Imagine trying to cram that big cushion into the tight space between the back and the arm of the chair. It's just not comfortable. They much prefer the spacious equatorial positions.
The "Cis" Dilemma: Where Does Everyone Sit?
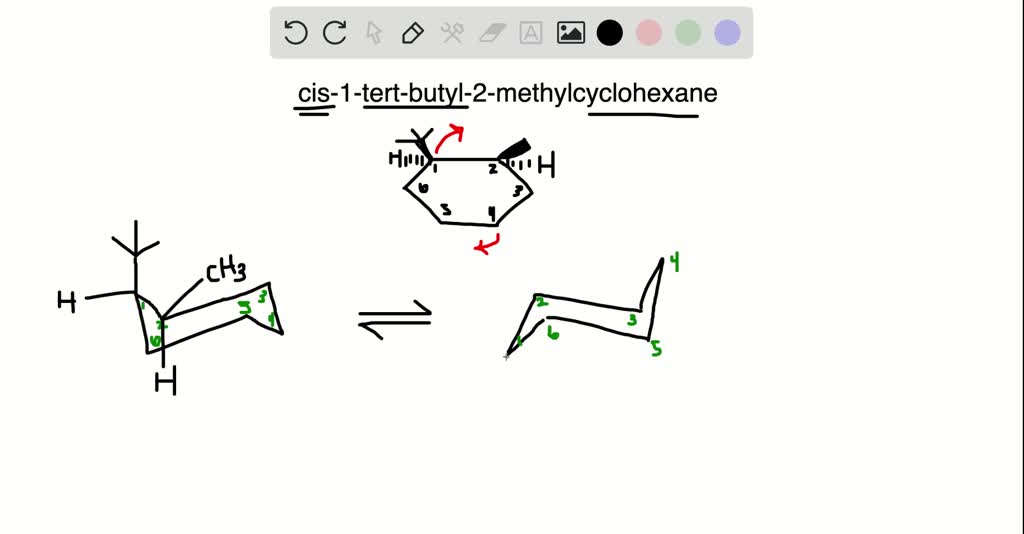
So, we have our cis 1 tert-butyl 2 methylcyclohexane. The tert-butyl is bulky, and the methyl is smaller. They’re on the same side of the ring. And our cyclohexane ring is doing its chair thing, with axial and equatorial spots.
Because the tert-butyl group is so massive, it’s going to want to be in the equatorial position. It’s just too big and awkward to be axial. Now, the methyl group is smaller, so it has a bit more flexibility.

In our cis arrangement, if the tert-butyl takes an equatorial spot, the methyl group also tends to end up in an equatorial spot. This is the molecule's happiest, most comfortable arrangement. Think of it like arranging guests at a dinner party. You wouldn't put your most boisterous guest right next to someone who wants a quiet meal, right? You'd try to give everyone comfortable seating.
If, by chance, the tert-butyl was forced into an axial position (which is less likely for such a big group), and the methyl was also axial (since they’re cis), they’d be bumping into each other quite a bit. It would be like trying to have a conversation in a crowded elevator – a bit awkward and cramped.
Why Should You Care About This Molecular Squabble?
Okay, so why should you, the everyday reader, bother with this seemingly obscure bit of molecular origami? Well, it turns out that how molecules arrange themselves – their conformation – can have a huge impact on how they behave.
Think about how the shape of a key determines if it fits into a lock. Or how the specific fit of a glove on your hand makes it useful. Molecules are no different! The precise three-dimensional shape, influenced by these chair conformations and the position of substituents, dictates:
- How drugs interact with our bodies: The active ingredient in a medication needs to fit perfectly into a specific receptor in your cells. If its shape is off, it won’t work, or worse, it could cause harmful side effects. This is like trying to use the wrong key to open a door – it just won't do the job.
- How flavors and scents work: That delicious strawberry flavor? Or the calming scent of lavender? These sensations are triggered when specific molecules fit into the receptors in your nose and mouth. Their shape, influenced by their conformation, is everything!
- How materials are made: The strength of plastics, the way dyes color fabrics, even the properties of fuels – all of these are influenced by the shapes and interactions of the molecules they're made of.
So, when chemists are designing new medicines, creating new materials, or trying to understand how biological processes happen, they need to know the preferred shape, or conformation, of the molecules they're working with. Our cis 1 tert-butyl 2 methylcyclohexane, with its preference for the equatorial arrangement to avoid steric clashes (that’s the fancy word for bumping into each other!), is a classic example of this principle.
A Little Bit of Molecular Manners
In a nutshell, this molecule teaches us about molecular preferences. Just like you have a favorite chair or a preferred way to arrange your books, molecules have preferred shapes that minimize energy and maximize stability. The bulky tert-butyl group, with its inherent desire for space, forces the molecule into a conformation where it can spread out comfortably, especially in the case of the cis isomer where it aims for that dual equatorial setup.
So, the next time you’re tidying up, arranging cushions, or even just admiring a well-made piece of furniture, take a moment to appreciate the hidden world of molecular shapes. It’s a world where things are always trying to find their most comfortable, stable, and therefore, most functional, arrangement. And that, my friends, is pretty neat, even if it does come with a name that’s a bit of a tongue twister!
