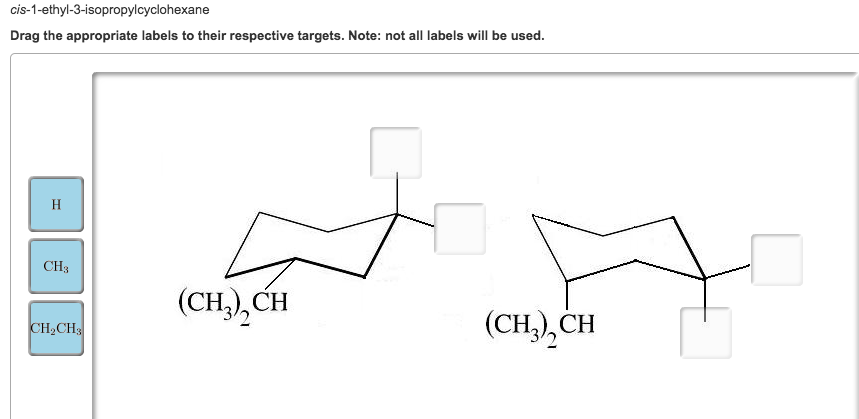
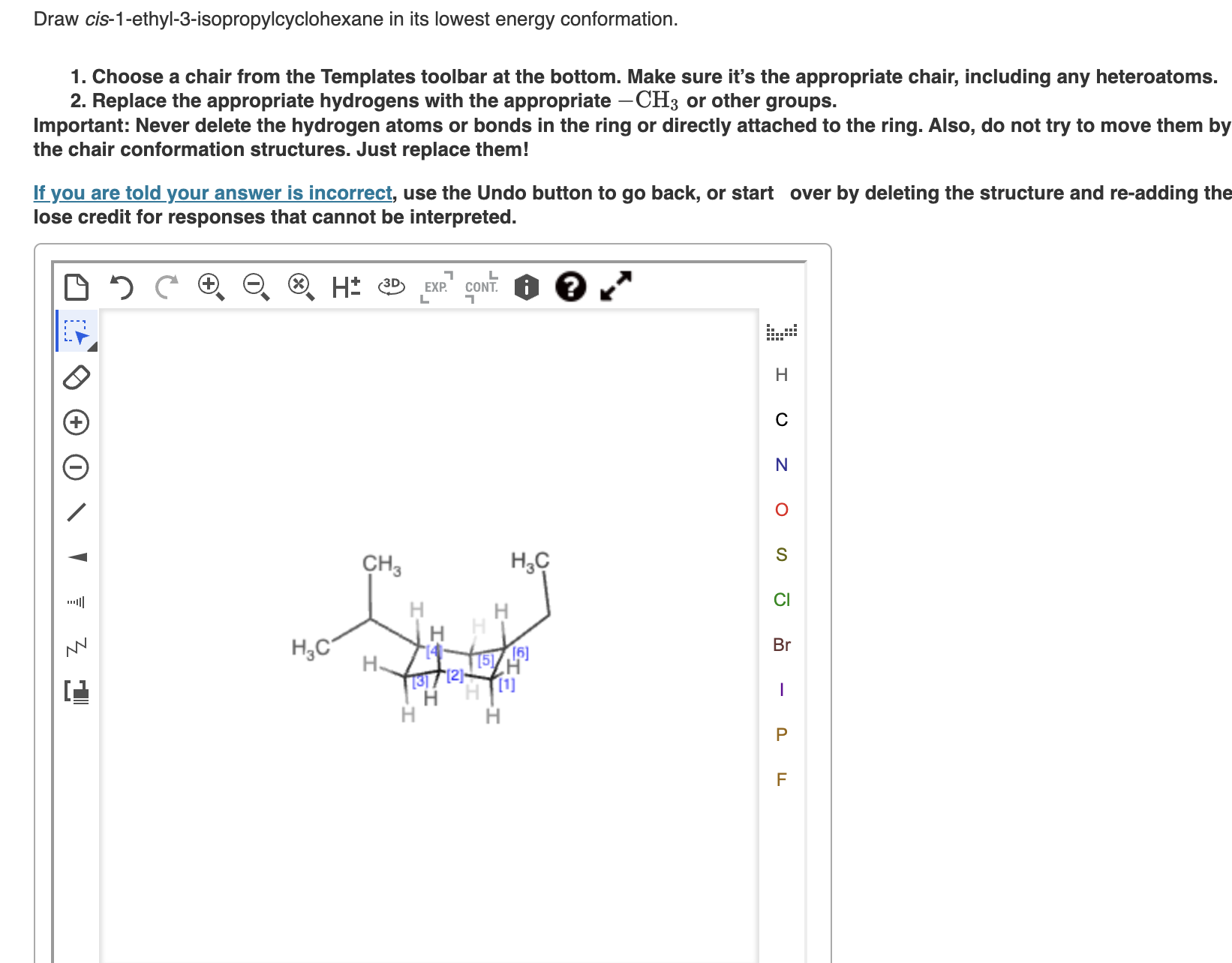
Cis 1 Ethyl 3 Isopropylcyclohexane In Its Lowest Energy Conformation

Ever looked at a molecule and thought, "Wow, that's… complicated"? Yeah, me too. Especially when you start throwing in fancy names like "cis 1-ethyl-3-isopropylcyclohexane." Sounds like something a mad scientist brewed up in a forgotten lab, right? But stick with me, because even these super-scientific-sounding things have a surprisingly down-to-earth side. Think of it like this: we all have our favorite comfy couch, our go-to Netflix binge position, or that one outfit that just feels right. Molecules are kind of the same way. They like to get comfy too, and when they do, they settle into what's called their "lowest energy conformation."
So, what's a "conformation," anyway? Imagine you've got a bunch of pipe cleaners. You can bend them, twist them, and generally make them into all sorts of shapes. A molecule is a bit like that, but with atoms and bonds. A conformation is just one of those possible shapes. And "lowest energy" means it's the shape where the molecule is feeling the most relaxed, the least stressed. It’s like when you finally collapse onto your bed after a long day – that's your lowest energy conformation for that moment.
Our star today is cis 1-ethyl-3-isopropylcyclohexane. Let’s break that down, shall we? "Cyclohexane" is our main stage. Think of it as a six-membered ring, like a perfectly round hula hoop. Now, imagine that hula hoop is made of little connected balls (atoms) and sticks (bonds). It’s not perfectly flat, though. It actually likes to wobble a bit, forming a shape that looks remarkably like a chair. Seriously, it’s called the "chair conformation," and it’s the most stable shape for a simple cyclohexane ring. It’s like how you instinctively know to sit on a chair rather than trying to balance on a tightrope.
Now, we’ve got some decorations on our chair. We've got an ethyl group and an isopropyl group. These are just little side chains of carbon and hydrogen atoms. The "1-ethyl-3-isopropyl" tells us where they're attached to our cyclohexane ring. They're on carbons 1 and 3. And the "cis" part? Ah, that's the kicker! "Cis" is like saying they're on the same side of the ring. Imagine two people sitting next to each other on a park bench. If they're on the same side, they're "cis." If one's on one end and the other's on the other end, they're "trans." For our molecule, the ethyl and isopropyl groups are chilling on the same side of that cyclohexane chair.
So, we have our cyclohexane chair, and on two specific "legs" of the chair, we have these ethyl and isopropyl groups. Now, the big question is: how do they prefer to sit to be the most comfortable? You see, these groups, especially the isopropyl with its extra bulk, can get in each other's way. It’s like trying to cram two big suitcases into the back of a tiny car. You have to arrange them just right to make them fit without everything popping open.
In the chair conformation, there are two important types of positions for these groups: axial and equatorial. Think of the chair again. The axial positions are like the upright posts on the back and legs of the chair. They stick straight up or straight down. The equatorial positions are more like the arms or the seat of the chair – they’re more out to the sides and a bit flatter.
Now, molecules, bless their little atom hearts, are a bit like us. They don’t like things bumping into each other unnecessarily. These axial positions? They can be a bit crowded. If you have bulky groups sitting in axial positions, they tend to get a bit of a steric clash. It's like trying to wear a giant, puffy winter coat indoors. You're just bumping into furniture all the time. You’re not relaxed; you’re annoyed. The molecule feels that annoyance too, in the form of increased energy.
So, to reach that coveted lowest energy conformation, our cis 1-ethyl-3-isopropylcyclohexane is going to try its best to put its bigger, bulkier groups in the equatorial positions. Why? Because the equatorial positions are, well, more spacious. They’re like the wide-open spaces where you can stretch your legs without kicking the person next to you. Less bumping, less fuss, less energy.
Let's visualize this. Imagine our cyclohexane chair. We have an ethyl group and an isopropyl group. The isopropyl group is bigger and has more atoms sticking out. It’s like the bigger, more boisterous friend. The ethyl group is a bit more slender, like the quieter acquaintance.
Because they are "cis," they are on the same side of the ring. This means they'll likely be positioned such that one is "up" and the other is "down" relative to the plane of the ring (even though the ring isn't flat, we can imagine a conceptual plane). In the chair conformation, this means one will tend towards an axial position and the other towards an equatorial, or they can both be equatorial, or both axial. The molecule’s goal is to minimize those awkward bumps.
The isopropyl group, being the bigger of the two, really wants to be in an equatorial position. It’s the biggest guy at the party, and he wants to spread out and not be crammed into a corner. If the isopropyl group is in an axial position, it’s going to have some serious "sternal clashes" – it’s going to be bumping up against other parts of the molecule. Imagine trying to do yoga with a giant beach ball stuck to your back; it’s just not going to happen comfortably.
The ethyl group, while not as hefty as the isopropyl, also prefers to be equatorial if it can. It’s like the slightly less boisterous friend who also appreciates a bit of personal space. When both groups can occupy equatorial positions, that’s generally the sweetest spot for the molecule.
Now, for our cis isomer, things get interesting. Because they are on the same side, one of them must be in an axial position if the other is in an equatorial position. Or, in a less favorable arrangement, both could be in axial positions. Let's think about the "flipping" of the chair. The cyclohexane ring can interconvert between two chair conformations, like a hammock swaying back and forth. When it flips, the axial positions become equatorial and vice versa. So, what might be axial in one chair conformation will be equatorial in the flipped one, and vice versa.
The lowest energy conformation for cis 1-ethyl-3-isopropylcyclohexane is the one where the largest group (isopropyl) is in the equatorial position. This minimizes those dreaded steric clashes. Even if it means the smaller ethyl group has to take on an axial position in that same conformation, it’s still a better situation than having the big isopropyl group crammed axially.
Think about arranging furniture in a small room. You wouldn’t put the biggest, bulkiest armchair right in the doorway, would you? No, you'd push it to the side, maybe near a window, to maximize your walking space. That’s exactly what our molecule is doing. The isopropyl group, like the bulky armchair, is going to find the most open spot – the equatorial position.
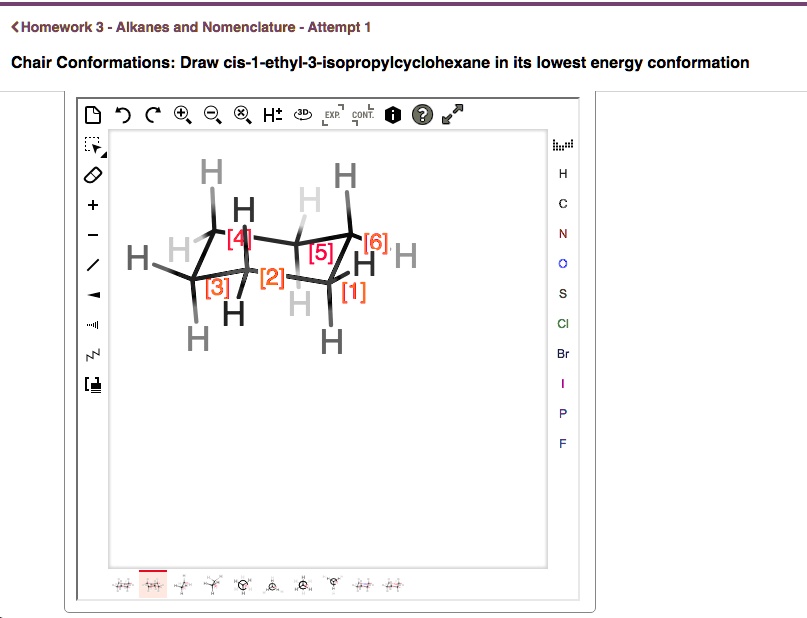
So, in its most relaxed, chill-out mode, you’ll find cis 1-ethyl-3-isopropylcyclohexane with the isopropyl group comfortably nestled in an equatorial position. The ethyl group might be in either an axial or equatorial position depending on the specific flip of the chair, but the overriding principle is that the biggest offender (the isopropyl) is going to be in the least offensive spot. It's all about avoiding those uncomfortable, energy-sapping bumps.
This isn't just some abstract chemistry concept. Understanding these shapes is super important. It helps scientists predict how molecules will behave, how they'll interact with each other, and even how drugs will work in our bodies. A drug molecule needs to fit into a specific spot, like a key in a lock. If it's all twisted up in a weird, high-energy shape, it might not fit properly. The lowest energy conformation is often the shape that's most effective for its job.
Imagine trying to use a remote control with a bent battery cover. It’s awkward, it doesn’t work right, and it’s just generally frustrating. The molecule in its lowest energy conformation is the one with the perfectly aligned battery cover – it just works.
So, the next time you hear a name like cis 1-ethyl-3-isopropylcyclohexane, don't let it intimidate you. Just think of it as a little molecular character trying to find its most comfortable pose. It’s a story of minimizing awkward bumps and maximizing chill. And who among us can't relate to that?
It's a constant dance of atoms and bonds, all striving for that sweet spot of stability. They’re not consciously thinking about it, of course, but the fundamental laws of physics dictate that lower energy states are more favored. It’s like a ball rolling downhill; it naturally seeks the lowest point. Our molecule is doing the same, just in three dimensions and with more carbon atoms.
The “cis” nature is a key constraint. If it were “trans,” the groups would be on opposite sides of the ring. In that scenario, it might be possible for both to occupy equatorial positions, which would be even more stable. But with “cis,” the universe of possible comfortable arrangements is a little more restricted, and the molecule has to make the best of it. It’s like being at a crowded concert; you might not have your ideal personal space, but you find a spot where you can still enjoy the music without getting too jostled.
The conformational analysis, the fancy term for figuring out these preferred shapes, is a huge part of organic chemistry. It’s how we understand reactivity, build complex molecules, and design new materials. And it all boils down to this idea of minimizing energy. It’s the molecular equivalent of tidying up your room so you can actually find things and move around without tripping. A tidy, low-energy molecule is a happy, functional molecule.
So, next time you see a cyclohexane derivative with a bunch of substituents, just remember: they’re all vying for the best seat in the house, the most spacious equatorial position, to avoid those pesky axial clashes. It’s a silent, constant negotiation happening at the molecular level, all in the pursuit of ultimate comfort and stability. And that, my friends, is a concept we can all get behind.
