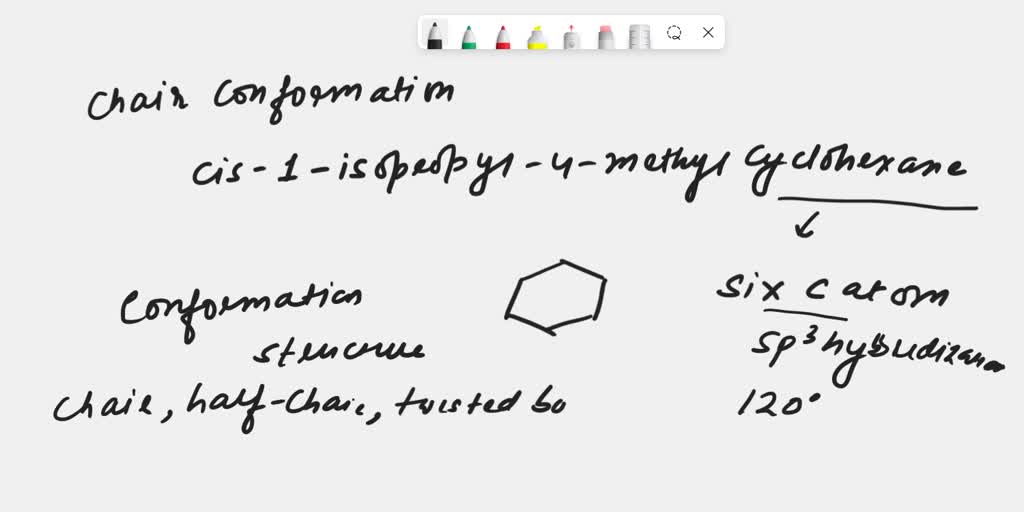
Cis 1 4 Dimethylcyclohexane Chair Conformation
Hey there, fellow curious minds! Ever feel like life's a little too… flat sometimes? Well, buckle up, because we're about to take a delightful detour into the wonderfully wiggly world of molecules, specifically a rather charming chap named cis 1,4-dimethylcyclohexane. Now, I know what you're thinking: "Molecules? Sounds a bit… nerdy." But trust me, by the end of this little adventure, you'll see how even the tiniest building blocks of our universe can add a splash of fun and a whole lot of aha! moments to your day.
So, what's the big deal with this "cis 1,4-dimethylcyclohexane"? Let's break it down, shall we? Imagine a ring of six carbon atoms, like a perfectly round hula hoop. That's our cyclohexane. Now, imagine we put two little methyl groups (that's just a fancy term for a carbon with three hydrogens sticking to it – think of them as tiny little arms waving hello) onto this hoop. And the "1,4" part? That simply means these two methyl groups are sitting on opposite sides of the ring, like they're doing a little dance across the circle from each other.
But here's where the magic truly happens, my friends! Molecules, especially those with rings, aren't stiff and unyielding. Oh no, they're more like little acrobats, constantly shifting and bending into different shapes. This, my dear reader, is what we call conformation. And for our special friend, cis 1,4-dimethylcyclohexane, the most stable and favorite shape is the oh-so-elegant chair conformation.
Picture this: you're sitting in a comfy armchair. That's the vibe! The cyclohexane ring, instead of being flat like a pancake, buckles and bends to form this armchair shape. Two carbon atoms are pointing upwards, like the armrests, and two are pointing downwards, like the legs. It's a sturdy, balanced pose, much like when you're feeling perfectly content and ready to tackle anything. This chair conformation is all about finding the lowest energy, the most relaxed state for our molecule. Think of it as the molecular equivalent of a really satisfying stretch after a long day!
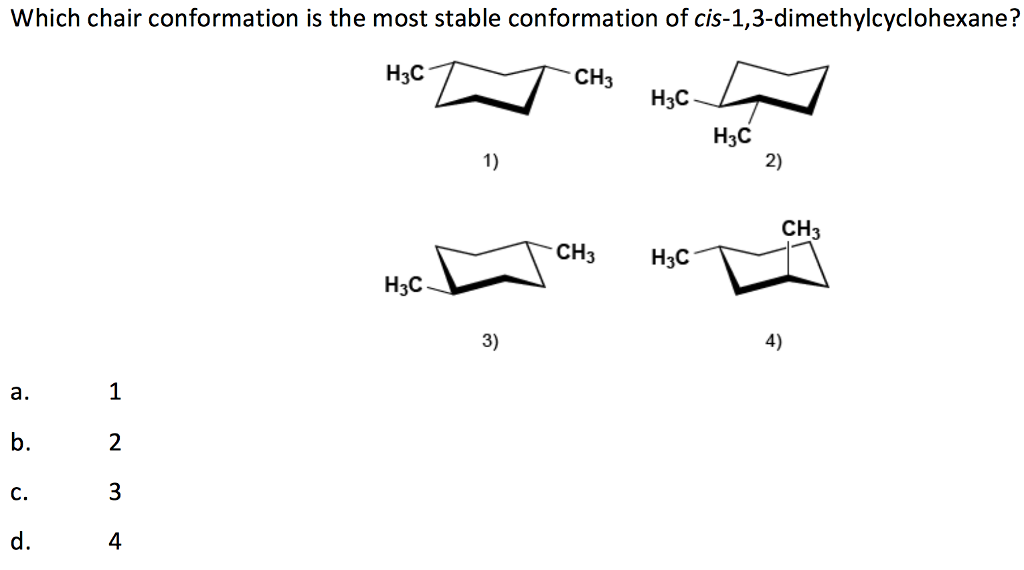
Now, why is this cis configuration so important in the chair? Remember our two methyl groups? In the cis arrangement, they're on the same side of the ring's general plane. When our cyclohexane ring adopts its comfy chair form, these two methyl groups can take up positions that are a bit more… well, polite. One will be pointing up (we call this 'axial') and the other will also be pointing up, but in a slightly different direction relative to its neighbor. Or, one could be 'axial' up and the other 'equatorial' (sticking out sideways, like it's lounging on the armrest). The key is that in the cis isomer, even though they're on the same side, they manage to avoid too much bumping and jostling.
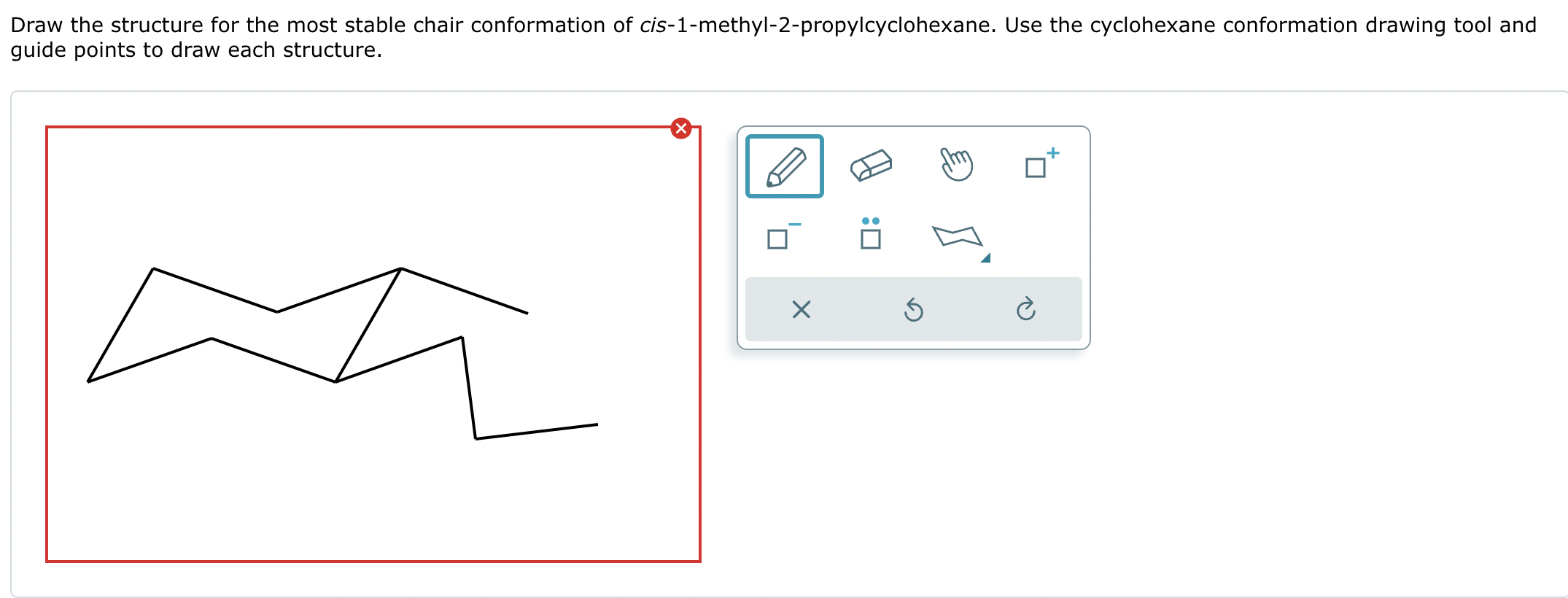
Let's talk about these 'axial' and 'equatorial' positions. Imagine the chair again. The axial positions are like the straight-up-and-down poles. They're directly above or below the ring. The equatorial positions are more like the arms of the chair, sticking out to the sides. Now, these methyl groups, being a little bit bulky, prefer to be in the equatorial positions because they have more elbow room. They don't want to be crammed up against other parts of the molecule, right? It's like having too many people in a small elevator – nobody's happy!
For cis 1,4-dimethylcyclohexane, the most stable chair conformation is when both methyl groups are in these more spacious equatorial positions. It’s like two friends finding the most comfortable spots on the sofa, each with plenty of legroom. This arrangement minimizes any awkward clashes, making the molecule feel super content and stable. It’s this preference for specific positions that helps us understand how molecules behave and interact. Pretty neat, huh?
And this understanding, my friends, is not just for the lab coats! It's a little peek into the intricate dance of the universe. It’s about appreciating how even the smallest pieces have their own unique ways of fitting together, their own favorite ways of existing. Think about it: understanding these molecular shapes can help scientists design new medicines, create stronger materials, or even develop more efficient fuels. It's the science behind the everyday magic we often take for granted.
Plus, isn't there something inherently satisfying about understanding how things work at their most fundamental level? It’s like solving a tiny, three-dimensional puzzle that’s been around forever. It gives you a sense of order and a deeper appreciation for the complexity and beauty of the natural world. So, the next time you're sipping your morning coffee or enjoying a walk in the park, remember that even those simple things are made possible by these fascinating molecular structures, doing their chair-yoga and finding their most comfortable conformations.
So, cis 1,4-dimethylcyclohexane and its chair conformation might sound like a mouthful, but it's a beautiful illustration of how shape matters, how stability is achieved, and how even within the realm of tiny molecules, there's a whole lot of elegance and efficiency going on. It’s a reminder that there’s a hidden world of wonder all around us, just waiting to be explored. Don't you just feel a little bit smarter and a whole lot more inspired to dive deeper into the amazing science that shapes our reality? Keep that curiosity alive, and who knows what other fascinating molecular adventures await you!
