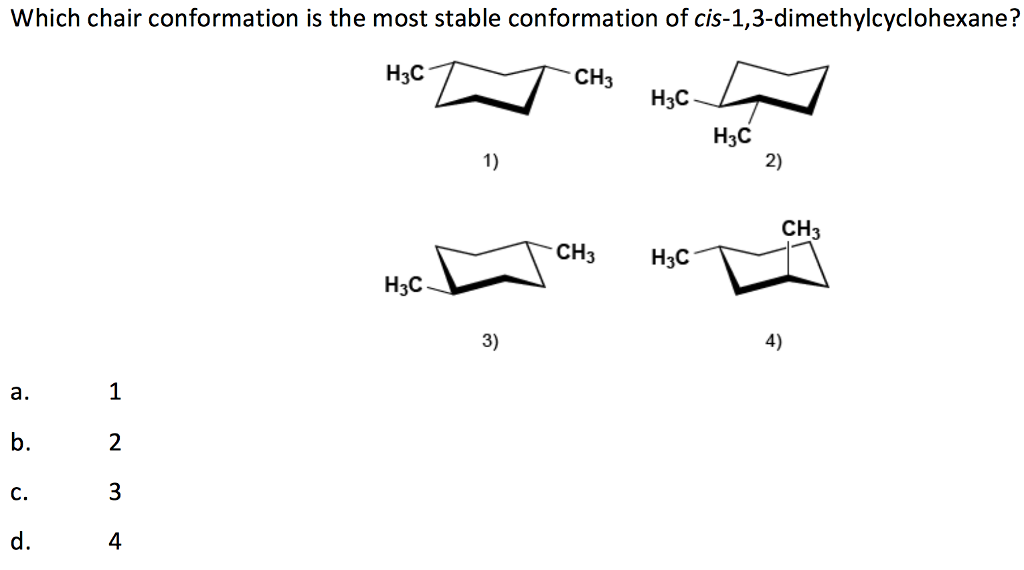
Cis 1 3 Dimethylcyclohexane Chair Conformation
Hey there! Ever find yourself staring at a molecule and wondering, "What's the big deal?" Well, today we're going to dive into a little something called cis 1,3-dimethylcyclohexane and its rather fascinating "chair conformation." Don't worry, it's not as complicated as it sounds, and actually, it's a bit like understanding how your favorite comfy armchair is put together. Stick around, and we'll make sense of it together!
So, imagine you've got a ring of six carbon atoms. That's our basic cyclohexane, the foundation for a lot of things in chemistry. Now, let's say we've attached two tiny "arms" – methyl groups, chemists call them – onto this ring. These are like adding little decorative cushions to our basic chair frame. We've got one methyl group at position 1 and another at position 3. Easy enough, right? The "cis" part is where it gets a tiny bit more interesting. Think of "cis" as meaning "on the same side." So, if you imagine our cyclohexane ring lying flat for a second (which it doesn't really do, but bear with me!), both of our methyl "cushions" would be sticking up, or both sticking down. They're pals, hanging out together on the same side of the ring.
Now, here's the fun part: the chair conformation. Molecules, especially rings like cyclohexane, aren't stiff, flat plates. They're more like wobbly, bendy structures that try to find the most comfortable, lowest-energy shape. And for cyclohexane, that shape is the chair. Think about a real armchair. It has a seat, a back, and arms. It's not a flat board; it's designed for comfort, right? Our cyclohexane molecule does something similar. It twists and bends into a shape that looks remarkably like a little three-dimensional chair.
Why is this "chair" thing so important? Well, it's all about how these molecules interact with their surroundings and with each other. Just like how you can sit more comfortably in a well-designed chair than a wobbly stool, molecules have preferred shapes that make them more stable. This stability affects everything from how drugs work in your body to how plastics are formed.
Let's get back to our cis 1,3-dimethylcyclohexane. We have our two methyl groups, and they're on the same side. In the chair conformation, these groups can either be in an "axial" position or an "equatorial" position. Imagine the chair again. Axial positions are like the legs of the chair – they point straight up or straight down, parallel to the imaginary central axis of the molecule. Equatorial positions are more like the arms and the seat – they stick out more to the side, roughly in a plane around the middle of the molecule.

Now, here's where our little methyl cushions come into play. When a methyl group is in an axial position, it's like having a big, bulky cushion sticking straight up and potentially bumping into other things. It's a bit crowded. Think of trying to squeeze into a seat with someone's armrest digging into your ribs. It's not ideal, and it takes more energy to maintain that position.
On the other hand, when a methyl group is in an equatorial position, it's like that cushion is placed nicely on the armrest or the seat. It's out of the way, less crowded, and much more comfortable for the molecule. It's like finding the perfect spot on the sofa where nothing is poking you.
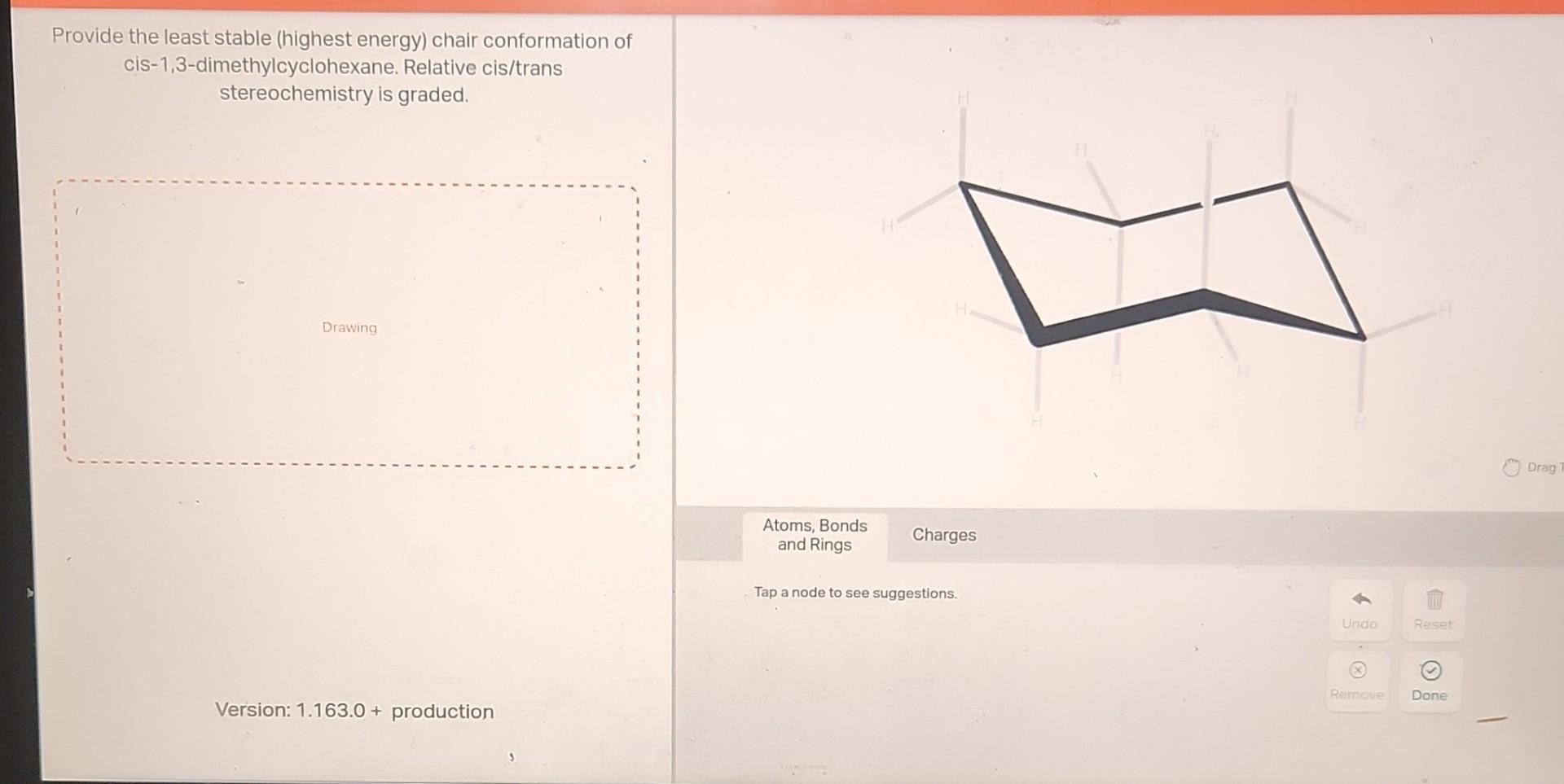
So, for cis 1,3-dimethylcyclohexane, we have two methyl groups on the same side. One way to arrange them is to have both in axial positions. This is like having two big cushions sticking straight up, right next to each other. It's pretty cramped and energetically unfavorable. Imagine trying to fit two bulky suitcases in the overhead bin, both trying to stick straight up – a struggle!

The other way to arrange them, while still keeping them "cis" (on the same side), is to have one methyl group in an axial position and the other in an equatorial position. Or, and this is key, the molecule can "flip" its chair shape. Think of it like the chair flipping upside down and righting itself, but the groups switch roles. If one methyl was axial, it might become equatorial, and vice versa. In the case of cis 1,3-dimethylcyclohexane, the most comfortable arrangement – the one that takes the least energy – is when both methyl groups are in equatorial positions. This is because they are "cis," meaning they are on the same side of the ring. If they are both equatorial, they can both stick out away from the ring, like two nicely placed armrests, making the whole molecule much happier and more stable.
Why should you, a regular human being who probably doesn't spend their days drawing molecular structures, care about this? Well, it's all about the subtle ways the world around us works. Understanding these molecular shapes helps scientists design better medicines. If a drug molecule has to fit into a specific slot in your body, like a key in a lock, its shape – its conformation – is absolutely critical. A slightly different arrangement of those methyl groups could mean the difference between a drug that works and one that doesn't, or even one that causes unwanted side effects.
Think about it like trying to fit a oddly shaped Lego brick into a specific spot. If it's twisted the wrong way, it just won't go. Molecules are a bit like that. The way they "sit" and the positions of their "arms" (like our methyl groups) determine how they interact with other molecules, like those in our bodies or in the environment.
It also helps us understand the properties of materials. The strength of plastics, the way paints dry, even the smell of a perfume – all these things are influenced by the shapes of the molecules involved. By understanding the preferred chair conformation of cis 1,3-dimethylcyclohexane, chemists can predict how it will behave and use that knowledge to create new and improved products.
So, the next time you're sitting in your favorite comfy chair, take a moment to appreciate its shape. It's not just about aesthetics; it's about stability and comfort. And in the microscopic world of molecules, that same principle of finding the most stable, comfortable shape – like our cis 1,3-dimethylcyclohexane settling into its all-equatorial chair – is what drives so many of the chemical reactions and interactions that make up our world. It’s a little bit of everyday magic, hidden in the three-dimensional dance of atoms!
