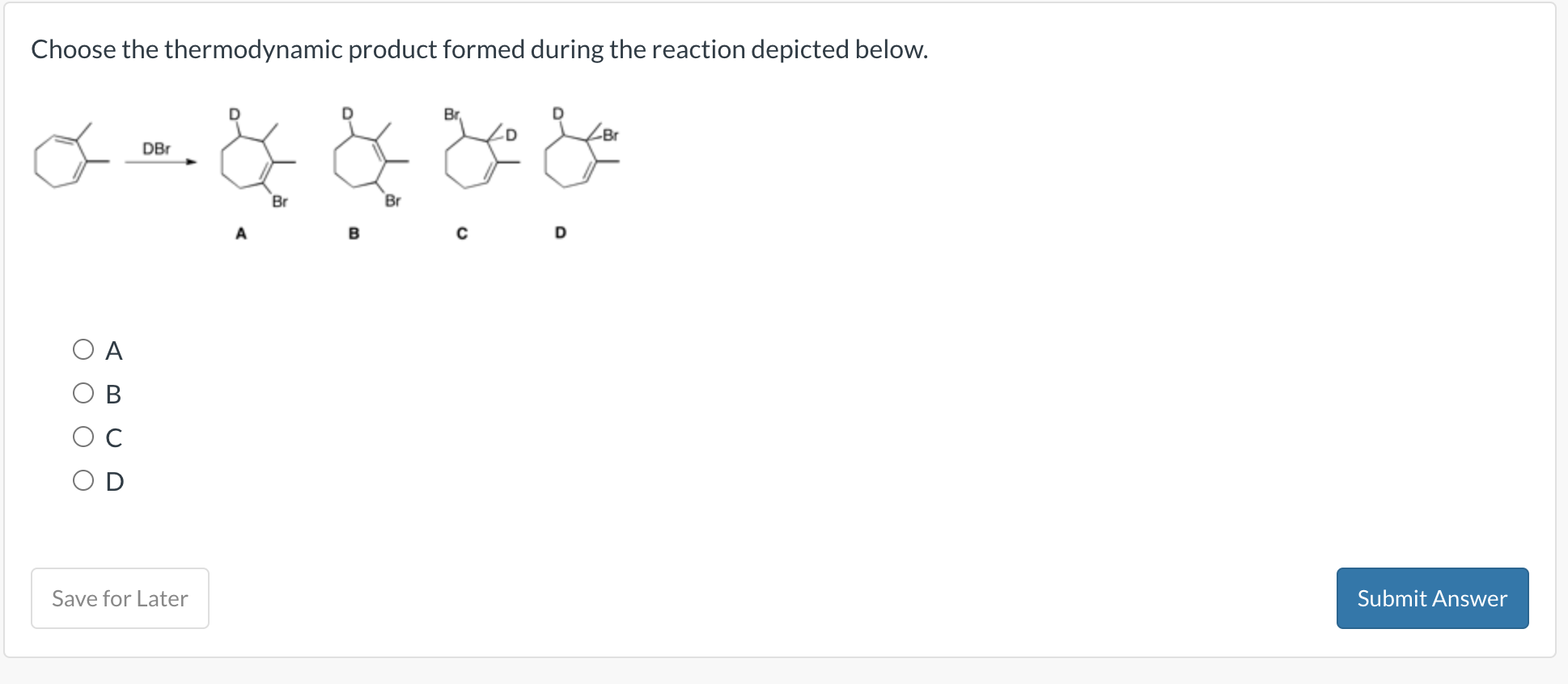
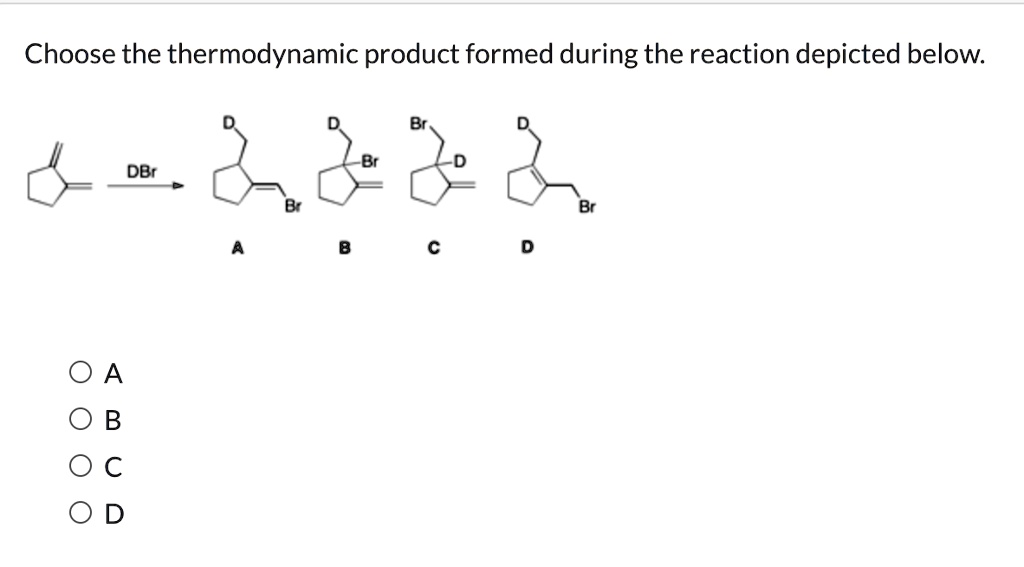
Choose The Thermodynamic Product Formed During The Reaction Depicted Below.
Get ready for a little bit of chemical magic, folks! We're about to dive into a world where atoms do the tango and molecules throw a party. It's all about what wonderful new stuff gets made when things react, and let me tell you, it's more exciting than a surprise pizza delivery.
Think of a chemical reaction like baking a cake. You start with a bunch of ingredients – flour, sugar, eggs – and then you mix them up and apply some heat. What do you get at the end? A delicious cake, totally different from the individual ingredients! In the same way, when chemicals get together, they can form something brand new, and today, we're going to figure out what that "something new" is.
We're going to be looking at a specific reaction, a little chemical drama playing out right before our eyes. It’s like watching a nature documentary, but instead of lions hunting gazelles, we have atoms bonding and breaking apart. And the star of our show today is the thermodynamic product. Ooh, sounds fancy, right? But don't worry, it's not as complicated as it sounds.
Imagine you have a bunch of LEGO bricks. You can build a simple little house, or you can build a magnificent castle. The castle takes more effort, more pieces, and it's definitely more impressive. In chemistry, the thermodynamic product is a lot like that castle. It's the most stable, the most comfortable, and the most "happy" arrangement the atoms can get into after the reaction is all done.
Think about it this way: if you leave a pile of LEGOs scattered on the floor, they're not exactly thrilled, are they? They're kind of chaotic. But if you build them into a sturdy castle, they're in their happy place, all locked together in a solid structure. The thermodynamic product is the chemical equivalent of that castle – the ultimate chill zone for the atoms.
Now, sometimes, reactions can take a shortcut. It's like if you're hungry and you just grab a handful of chips instead of making a gourmet meal. That's more like the kinetic product. It forms faster, it’s easier to get to, but it might not be as perfectly arranged or as stable in the long run.
Our mission today, should we choose to accept it (and trust me, you totally want to!), is to identify the thermodynamic product in the reaction we're about to witness. It’s like being a detective, sniffing out the clues and figuring out which outcome is the ultimate winner. The winner in this case is the one that’s the most energetically favorable.
So, we'll be presented with a chemical reaction, a little tableau of atomic interaction. Your job is to channel your inner chemical guru and pick out the product that represents the most stable, most relaxed, and ultimately, the most thermodynamic outcome. It's the one that nature sighs with contentment over.
Let's think about energy. Chemical reactions either give off energy (like a firecracker – boom!) or they soak up energy (like an ice pack – brrr!). The thermodynamic product is the one that results in the lowest overall energy state. It’s like the universe saying, "Ah, yes, this arrangement is the most peaceful."
Imagine you're at a playground. You can either run around wildly (that's like a fast, less stable product) or you can sit on the swings and enjoy a gentle, steady motion (that's like a slow, stable, thermodynamic product). The swings offer a more sustained, relaxed state.
So, when you look at the reaction provided, don't just be wowed by the initial burst of activity. Look deeper, like you're peering into the soul of the molecules. Which arrangement seems like it would be the most content, the least stressed out, the most… settled? That's your thermodynamic product calling out to you.
It’s important to remember that reactions don’t always take the easiest path. Sometimes they can overshoot, or get stuck in a less-than-ideal situation. But given enough time and the right conditions, they'll eventually settle into their most comfortable, most thermodynamic configuration. It's like letting a jiggly Jell-O set – it might wobble a bit at first, but eventually, it becomes firm and stable.
Think about the names of the products that are formed. Sometimes, the name itself can give you a hint, like a secret code. We're looking for that ultimate, low-energy, super-stable structure. It’s the one that's going to stick around, the one that’s truly found its chemical happy place.
Sometimes, you might see a reaction where two different products could form. One might form really quickly, like a flash in the pan. The other might take a little longer to get going, but it ends up being much more robust and enduring. That enduring one is our beloved thermodynamic product.
It’s like choosing between a quick hug and a long, warm embrace. The quick hug is fast and easy, but the long embrace leaves you feeling truly comforted and secure. The thermodynamic product is the chemical equivalent of that long, warm embrace.
So, when you're presented with the reaction, take a good look at the potential outcomes. Ask yourself, "Which one of these looks like it's settled down the most?" Which one seems like it’s got all its atoms perfectly aligned and in their most relaxed state? That’s your target!
Sometimes, there are subtle differences between the possible products. It’s like looking at two very similar shades of blue paint. One might be a bit brighter and more exciting, but the other has a depth and richness that just feels right. The thermodynamic product has that "just right" quality.
Let's get excited about this! We're not just looking at letters and arrows; we're witnessing the birth of new chemical entities. And our goal is to identify the one that's the most energetically content, the one that has achieved ultimate chemical bliss.
The thermodynamic product is the one that has the lowest Gibbs free energy. But don't let that scare you! Just think of it as the product that has the most "chill" factor. It's the one that has reached its lowest energy state, its happy equilibrium.
So, gaze upon the reaction. Let your eyes wander over the reactants and the potential products. Which one whispers promises of stability and lasting contentment? That, my friends, is the thermodynamic product you’re looking for! It’s the chemical champion of chill.
Think of it like a race. Some products are sprinters, zooming to the finish line in a blur. Others are marathon runners, pacing themselves for the long haul and crossing the finish line with steady grace. The thermodynamic product is the marathon runner – it might not be the fastest off the blocks, but it's the one that's built to last and in the most balanced state.
It's all about finding that sweet spot, that perfect arrangement where the atoms are as comfortable as they can possibly be. And that, my dear reader, is the essence of the thermodynamic product. So go forth, and choose wisely! Your chemical detective skills are about to shine.
The thermodynamic product is the most stable one, like the ultimate cozy blanket for atoms!
So, when you see that reaction, don't just guess. Look for the signs of ultimate chemical contentment. The one that looks like it's taken a deep, satisfied breath and settled in for the long haul. That's your winner!
It's a beautiful dance of atoms, and we get to play the role of discerning judge. We're looking for the most elegantly arranged, the most energetically sound outcome. The thermodynamic product is the masterpiece of chemical stability.
So, embrace the challenge! Embrace the chemistry! And most importantly, embrace the joy of finding that perfectly stable, wonderfully thermodynamic product. It's a little win for your brain and a lot of fun for your curiosity!
