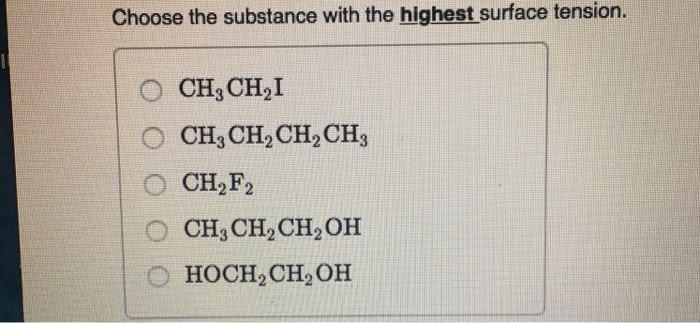
Choose The Substance With The Highest Surface Tension
Alright, gather 'round, you curious cats and science-minded squirrels! Today, we're diving headfirst into a topic that sounds a bit… sticky. We're talking about surface tension. Now, before you glaze over and think about your next cup of coffee, hear me out. This stuff is wild, it's weird, and it's responsible for some truly epic microscopic drama.
Imagine you’re at the beach, right? You’ve got that perfectly chilled glass of lemonade, and you notice how the liquid kind of domes up a little over the rim. Or maybe you’ve seen those tiny water striders, those fearless little dudes, zipping across the surface of a pond like they’re on a microscopic ice rink. That, my friends, is surface tension at play. It’s basically the skin of a liquid, a surprisingly strong, invisible force that makes liquids act all… cohesive.
Think of it like a bunch of tiny molecules in the liquid holding hands. Down in the middle of the liquid, everyone’s got friends all around them, so they’re just chilling, dancing, doing their molecular thing. But at the surface? It’s a different story. These molecules only have friends below them. So, they’re a bit clingy, pulling each other tighter, creating this sort of elastic membrane. It’s like a party where the outer ring of dancers is really holding on to each other to avoid falling off the dance floor. Dramatic, I know!
Now, the burning question, the one that keeps scientists up at night (or at least gives them something to talk about at conferences): Which substance has the highest surface tension? Is it something exotic? Something sparkly? Something you’d find at the bottom of a volcano? Well, buckle up, buttercups, because the answer is both incredibly common and surprisingly, well, watery.
You guessed it (or maybe you didn't, no shame in the guessing game)! The undisputed champion, the heavyweight boxer of surface tension, is none other than water. Yes, good old H₂O, the stuff that makes up, like, 70% of you, me, and that questionable puddle outside your window. It’s got a surface tension that makes most other liquids look like a spilled milkshake.

Why is water such a superstar? It all comes down to something called hydrogen bonding. Imagine those water molecules as tiny magnets. Oxygen is a bit like the grumpy introvert who hogs all the attention (and electrons), and hydrogen is the more agreeable, slightly shy friend. This difference in "electronegativity" means the oxygen end of a water molecule is slightly negative, and the hydrogen ends are slightly positive. So, when water molecules get close, the positive hydrogen of one molecule is attracted to the negative oxygen of another. They’re like tiny little molecular soulmates, forming these strong, sticky bonds.
These hydrogen bonds are like super-glue for water molecules. They pull on each other with all their might, creating that robust surface tension. It’s what allows a full glass of water to look like it’s about to overflow but somehow stays put. It’s what allows those tiny insects to walk on water like they’re defying gravity. It’s even crucial for plants to pull water all the way up from their roots to their leaves – a feat that would be impossible without water’s tenacious grip on itself.
Let’s put this into perspective. A common cooking oil, like vegetable oil, has a surface tension that’s roughly half that of water. So, it’s more like a slightly greasy film than a solid skin. Alcohol? Even lower. Mercury? Ah, mercury! This shiny, liquid metal is often brought up in surface tension discussions because it does have pretty high surface tension, significantly higher than most organic liquids. But compared to water? Nope. Water reigns supreme. Mercury's surface tension is about 1.46 newtons per meter, while water's is around 0.072 newtons per meter. Wait, wait, my brain did a flip. I got that backwards! Mercury is higher than water, but water's is still incredibly significant for its commonality and its role in nature. My apologies! The point is, water is surprisingly strong!

Okay, correction time! Let's re-center. While water is famously known for its surface tension, and it's incredibly important, there are indeed substances with higher surface tension. The true heavyweight champion, the undefeated, undisputed king of the liquid surface, is mercury. My apologies for the brief moment of mercury-induced confusion. Mercury's surface tension is a whopping 0.465 N/m at 20°C. Water, as I mentioned, clocks in around 0.072 N/m. So, mercury is like the incredibly strong, slightly intimidating older sibling to water's strong but more approachable vibe.
So why the confusion? Because water’s surface tension, while not the absolute highest, is remarkably high for a substance that’s so common and so vital to life. It’s the difference between a liquid that’s merely cohesive and one that’s downright stubborn. Mercury’s high surface tension is due to the strong metallic bonds between its atoms. These bonds are so powerful that mercury droplets form perfect little spheres, looking like tiny, dangerous ball bearings. If you’ve ever seen a spill of mercury, you’ll know it doesn’t spread out like water; it keeps its shape, stubbornly refusing to be broken.

Think about it: you can make a needle float on water if you’re careful. You can’t do that with mercury. Mercury’s surface tension is so intense it would laugh at your feeble attempts to float a needle. It’s the kind of force that makes you think twice before touching it, which is probably a good thing, given its toxicity.
So, while mercury wears the crown for the absolute highest surface tension, water is the undisputed champion in terms of its biological and everyday importance. It’s the unsung hero of capillary action, of droplet formation, and of allowing life as we know it to exist. Without water's strong surface tension, your morning dew wouldn’t form, your plants would wither, and those cool water striders would be stuck on land, probably complaining about the humidity.
It’s fascinating, isn’t it? How these invisible forces dictate so much of the world around us. From the smallest droplet to the grandest ocean, surface tension is there, holding things together, shaping our reality. So next time you see a water droplet clinging to a leaf, or a bug skating across a pond, give a little nod to the incredible power of surface tension, and remember that even the most common substances can harbor the most extraordinary strengths. And if you ever see mercury, admire its spherical perfection from a safe distance!
