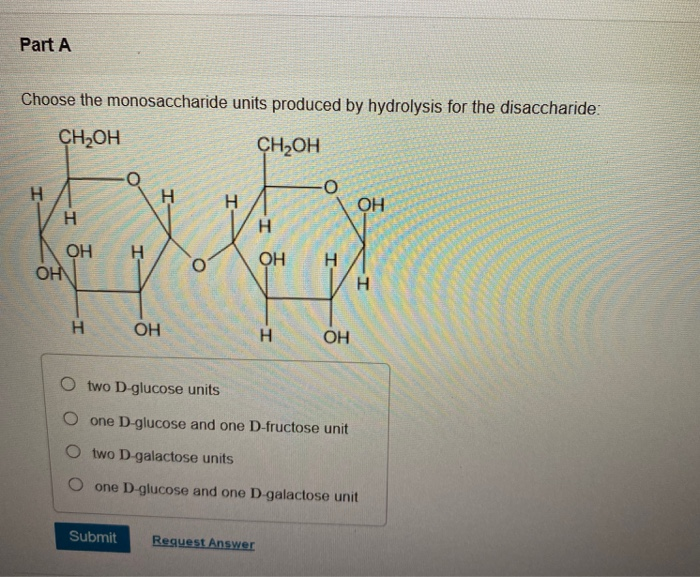
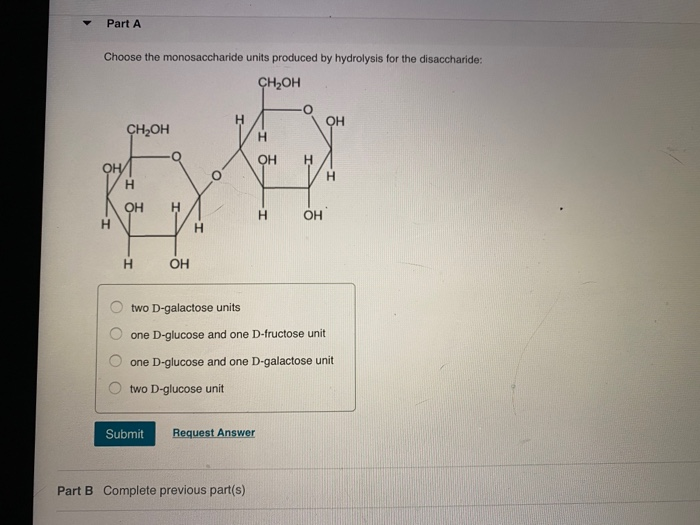
Choose The Monosaccharide Units Produced By Hydrolysis For The Disaccharide
Ever wondered what makes that sweet treat taste so sweet? Or how our bodies get the energy to run, jump, and even just think? It's all thanks to the fascinating world of carbohydrates, and at the heart of it all are these tiny building blocks called monosaccharides. Think of them as the LEGO bricks of the sugar world. Now, sometimes these single bricks like to join together to form bigger structures, creating what we call disaccharides. And here's where the real fun begins: when we break these disaccharides apart, we get back those original monosaccharide units. Choosing which monosaccharide units are produced by hydrolysis for a disaccharide is like being a master chef, deciding which ingredients will create your perfect dish!
So, why is this whole process of breaking down disaccharides into monosaccharides so cool and, dare we say, useful? Well, for starters, it's how our bodies digest the sugars we eat. When you enjoy a spoonful of honey (which is mostly fructose and glucose), or sip on milk (which contains lactose), your digestive system gets to work. It uses special enzymes, like tiny molecular scissors, to snip apart the disaccharides, releasing the individual monosaccharides. These simple sugars, like glucose, are then absorbed into your bloodstream, giving you that immediate energy boost. Without this breakdown, those complex sugars would just pass through us, unutilized – imagine eating a delicious cookie and getting no energy from it! That’s a carb catastrophe!
Beyond just our own bodies, understanding how disaccharides break down into specific monosaccharides is crucial in many industries. Think about the food industry. They use this knowledge to control sweetness levels, textures, and even the shelf life of products. For example, in making baby formula, understanding the hydrolysis of lactose is vital to ensure it's digestible for infants. Or consider the brewing industry, where the breakdown of complex sugars into simpler ones by yeast is what ultimately produces alcohol. It’s all about controlling those molecular reactions to achieve a desired outcome.
Let's dive into some of the common disaccharides and the monosaccharide units they yield upon hydrolysis. One of the most familiar is sucrose, the table sugar we sprinkle on our cereal. When sucrose is hydrolyzed, it breaks down into one unit of glucose and one unit of fructose. These are both incredibly important monosaccharides. Glucose is our body's primary fuel source, powering everything from our brain cells to our muscles. Fructose, often called fruit sugar, is also a monosaccharide and contributes to the sweetness of fruits and honey. It’s metabolized differently than glucose and can be a good source of energy, though too much can lead to certain health concerns.
Another common disaccharide is lactose, found naturally in milk and dairy products. Hydrolysis of lactose yields one unit of glucose and one unit of galactose. Galactose is another sugar that plays a role in our bodies, although it's less common as a standalone sugar in our diet. It's important for building certain complex molecules. For people who are lactose intolerant, their bodies lack sufficient amounts of the enzyme (lactase) needed to break down lactose into glucose and galactose, leading to digestive discomfort. This inability to perform that specific hydrolysis highlights the importance of these individual monosaccharide units for proper digestion and energy absorption.
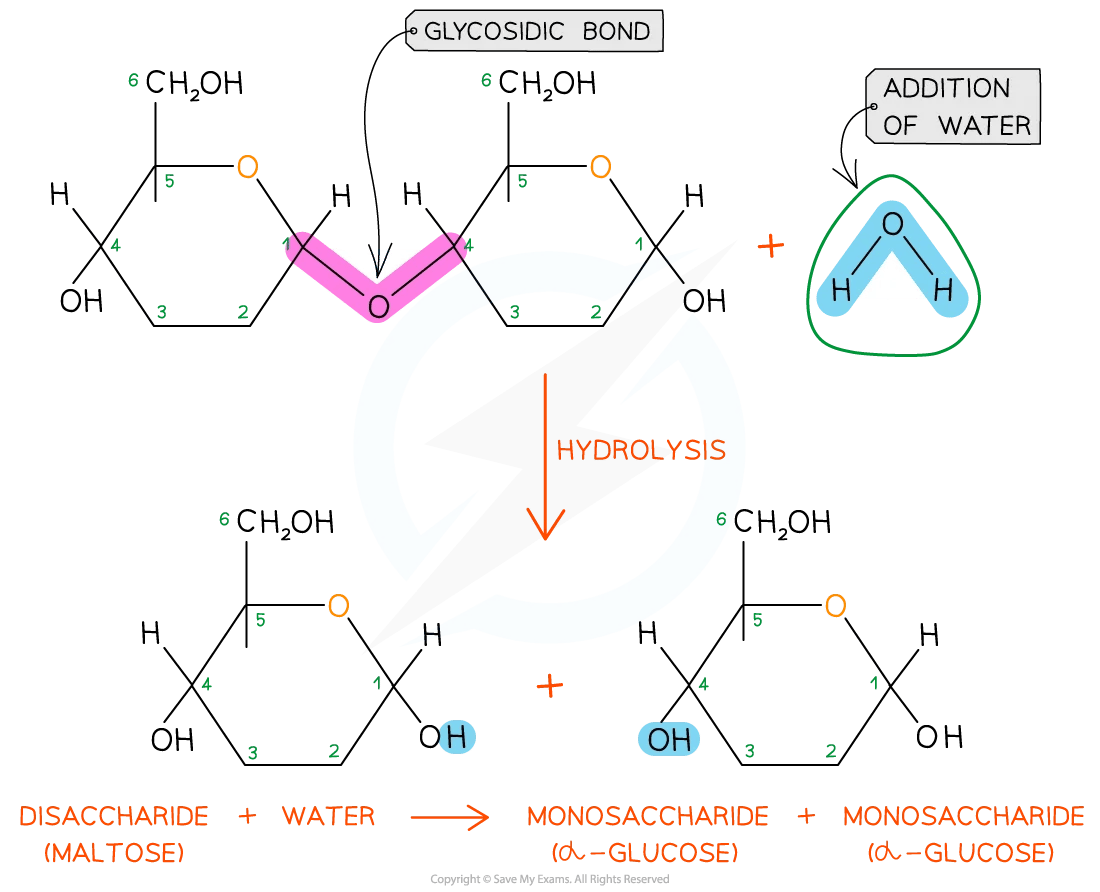
Then there’s maltose, often called malt sugar. It’s commonly found in germinating grains, like barley, and is a key component in the brewing process. Hydrolysis of maltose produces two units of glucose. This simple breakdown is fascinating because it shows how a disaccharide can be made of identical monosaccharide units. The energy stored within these two linked glucose molecules is efficiently released when they are separated, providing fuel for the yeast during fermentation.
Choosing the monosaccharide units produced by hydrolysis for a disaccharide is like unlocking a treasure chest of energy and building blocks!
Solved Part A Choose the monosaccharide units produced by | Chegg.com
The ability to choose, or rather, to understand and predict, which monosaccharide units will be produced from a specific disaccharide is fundamental to biochemistry and nutrition. It allows scientists to design diets, develop medical treatments, and engineer food products. For instance, in clinical nutrition, understanding the enzymatic hydrolysis of disaccharides helps in formulating specialized diets for individuals with metabolic disorders or digestive issues. We can tailor food intake based on how effectively specific disaccharides are broken down into usable monosaccharides.
Think about the sweet science of making ice cream. The type of sugar used, and how it breaks down, affects not just the sweetness but also the freezing point and texture. Using sucrose versus a blend that might yield more fructose can lead to different results. It's a delicate balance, and understanding the hydrolysis of these disaccharides is key to mastering that balance.
Ultimately, exploring the monosaccharide units produced by hydrolysis for a disaccharide opens up a world of understanding about the fundamental processes that sustain life and power our modern world. From the food we eat to the energy that fuels our bodies, these simple sugars and their larger counterparts are at the very core of it all. It’s a sweet subject, indeed, and one that’s surprisingly central to so many aspects of our daily lives!