Choose The Correct Orbital Diagram For Titanium.

Imagine you're a little kid, and your absolute favorite toy is a super cool, shiny set of building blocks. These aren't just any blocks; they're special, and each one has a very specific place it likes to live. Some blocks are super comfy and like to be alone, while others are perfectly happy crammed together with their best buddies. It's all about finding the right spot for each and every block!
Now, let's talk about Titanium. This isn't a toy, of course, but in the world of science, it's a bit like a bunch of energetic little particles that have their own favorite "rooms" to hang out in. We call these rooms orbitals. Think of them as cozy little apartments where electrons, the teeny-tiny energetic bits of an atom, love to chill.
Just like you wouldn't shove all your LEGOs into a single tiny box, these electrons have specific rules about where they can go. Some orbital "rooms" are like single studios, perfect for one electron. Others are more like bunk beds, where two electrons can share, but they have to be a bit polite and spin in opposite directions. It’s like a cosmic dance of electrons finding their perfect fit.
When scientists want to understand an element like Titanium, they draw these "orbital diagrams." It’s basically a picture of where all the electrons are likely to be hanging out. It’s like a blueprint of the atom’s social scene!
Now, here’s where it gets a little like a game of musical chairs. Electrons are a bit picky about their seating arrangements. They prefer to spread out into their own individual orbitals first, kind of like how you'd grab your own chair at the dinner table before you'd squeeze in next to someone. It’s a matter of personal space, even for subatomic particles!
So, when we look at Titanium, we're trying to figure out which orbital diagram shows this electron-spreading-out behavior correctly. It’s like trying to find the picture that perfectly captures the way the electrons are arranged. The wrong diagram would be like showing everyone crammed into the smallest room when there are plenty of bigger ones available. That just wouldn’t be very efficient or comfortable for the electrons!
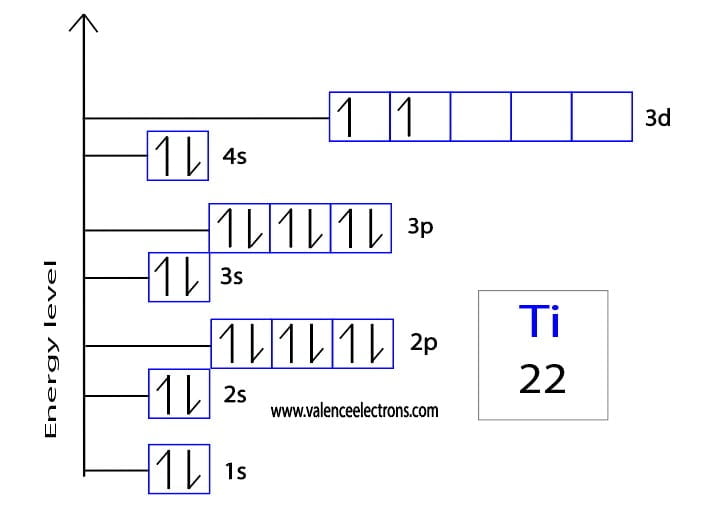
The whole point is to represent the electron configuration, which is just a fancy way of saying how the electrons are arranged. For Titanium, which has 22 electrons (that’s a lot of little roommates!), this arrangement follows some pretty predictable patterns. It’s like following a recipe for arranging your toys, making sure each piece goes in its designated spot.
There are different types of orbital "rooms," and they have funny names like 's', 'p', and 'd'. The 's' rooms are like little round studios, and there’s only one of each. The 'p' rooms are a bit more elongated, like little hallways, and there are three of them. Then you have the 'd' rooms, which are a bit more complex and come in groups of five. Imagine a whole apartment building with different kinds of rooms!
The electrons for Titanium fill up these rooms in a specific order. They start with the smallest, lowest-energy rooms first. It’s like filling your water bottle from the bottom up – you wouldn’t start pouring water into the very top, right? The same principle applies here, but with electrons and orbital rooms.
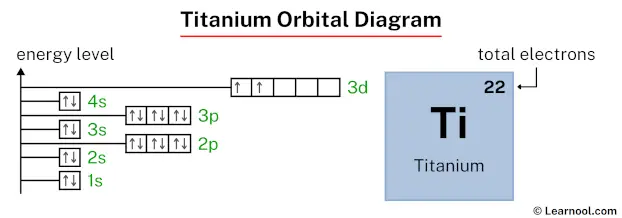
The fun part is seeing how the electrons arrange themselves in those 'd' orbitals for Titanium. This is where the diagrams get a little more interesting. They show how the electrons are distributed among those five 'd' rooms. It’s like seeing how a group of friends decides to spread out on a couch with multiple cushions.
One of the key rules is called Hund's Rule. This rule is like the electrons saying, "Let me get my own space before I have to share!" So, if there are multiple 'd' rooms that are the same size and energy, an electron will try to go into its own room first before it pairs up with another electron. It’s a bit like a shy person at a party – they’d rather have their own corner than be forced into a conversation.
So, when you look at an orbital diagram for Titanium, you're looking for the picture that shows these electrons filling up their rooms in the most sensible and polite way. It’s the diagram that respects their need for personal space and follows the order of energy levels. It’s a little peek into the secret life of an atom, showing how its tiny components organize themselves.
The correct diagram is the one that accurately reflects this electron behavior. It’s not just a random drawing; it’s a scientific representation of how the electrons in Titanium are arranged. It tells a story about how these fundamental particles interact and find their optimal resting places within the atom. It’s like a perfect snapshot of the atom’s inner world.
Imagine you have a bunch of little LEGO figures, and you have to put them into different shaped boxes. Some boxes are round, some are square, and some are long and thin. Each LEGO figure has a favorite type of box and a way they like to sit in it. The orbital diagram is like a picture showing all the LEGO figures happily in their correct boxes.
For Titanium, it's a bit like having 22 LEGO figures to place. They fill up the smaller, simpler boxes first, then move on to the more complex ones. And they always try to spread out into their own individual boxes before they decide to share. It’s all about making sure everyone has their own comfortable spot.
The 'd' orbitals are particularly interesting. Think of them as five medium-sized boxes. The electrons in Titanium will put one electron in each of these five boxes before they start pairing up in any of them. It’s like, "Okay, everyone gets their own chair at the table first!" This rule is fundamental to understanding how elements behave.
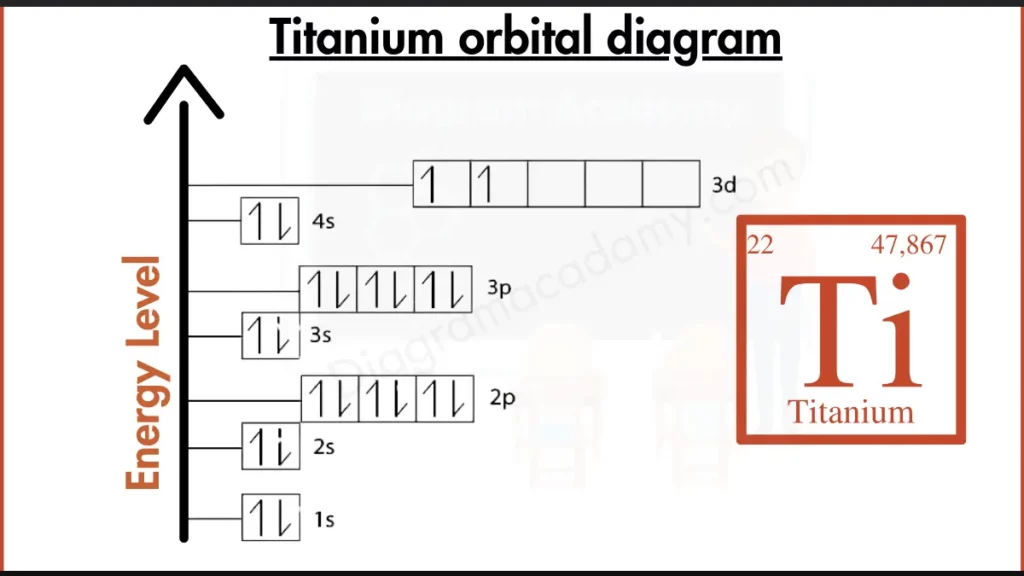
The incorrect diagrams might show electrons pairing up too early, or filling rooms that are higher in energy before the lower ones. That would be like putting your LEGO figures into the wrong shaped boxes or forcing them to sit on top of each other when there’s plenty of space. It just wouldn't look right, and it wouldn't represent how Titanium truly is.
So, when you’re presented with different orbital diagrams for Titanium, you’re essentially being asked to be a detective. You’re looking for the one that tells the truest story of the electrons’ arrangements. It's about finding the picture that makes the most sense according to the rules of electron behavior. It’s a visual puzzle for science enthusiasts!
It’s a surprisingly intricate dance happening at the atomic level. This arrangement of electrons in their orbitals is what gives Titanium its unique properties. It’s why it’s so strong, why it doesn’t rust easily, and why it’s used in everything from airplanes to medical implants. The humble orbital diagram holds the key to understanding this amazing element.
So, next time you see an orbital diagram, don’t just see a bunch of squiggly lines and arrows. See it as a charming little story about electrons finding their perfect homes, following rules of politeness and efficiency. It's a tiny universe of order and organization, all to explain the wonder that is Titanium. It’s a beautiful glimpse into the fundamental building blocks of our world.
