Choose The Correct Mechanism For The Following Sn1 Reaction.
Hey there, science adventurers! Ever stare at a chemical reaction and think, "Whoa, what's actually happening in there?" Today, we're diving headfirst into the wild world of SN1 reactions. It sounds fancy, right? But trust me, it's way more like a super-cool detective story than a boring textbook chapter.
So, what's the deal with SN1? It's all about substitution. Imagine a molecule with a guest it doesn't really like. It's going to kick that guest out and invite a new, friendlier one. That's substitution in a nutshell. And the "1" in SN1? It's a hint about the speed. We'll get to that juicy detail in a sec.
Now, SN1 reactions have a mechanism. Think of a mechanism as the step-by-step game plan. It’s the secret handshake, the secret recipe, the whole shebang that makes the reaction happen. And when it comes to SN1, there’s a specific, super-interesting way it goes down.
The SN1 Grand Entrance: It's a Solo Act!
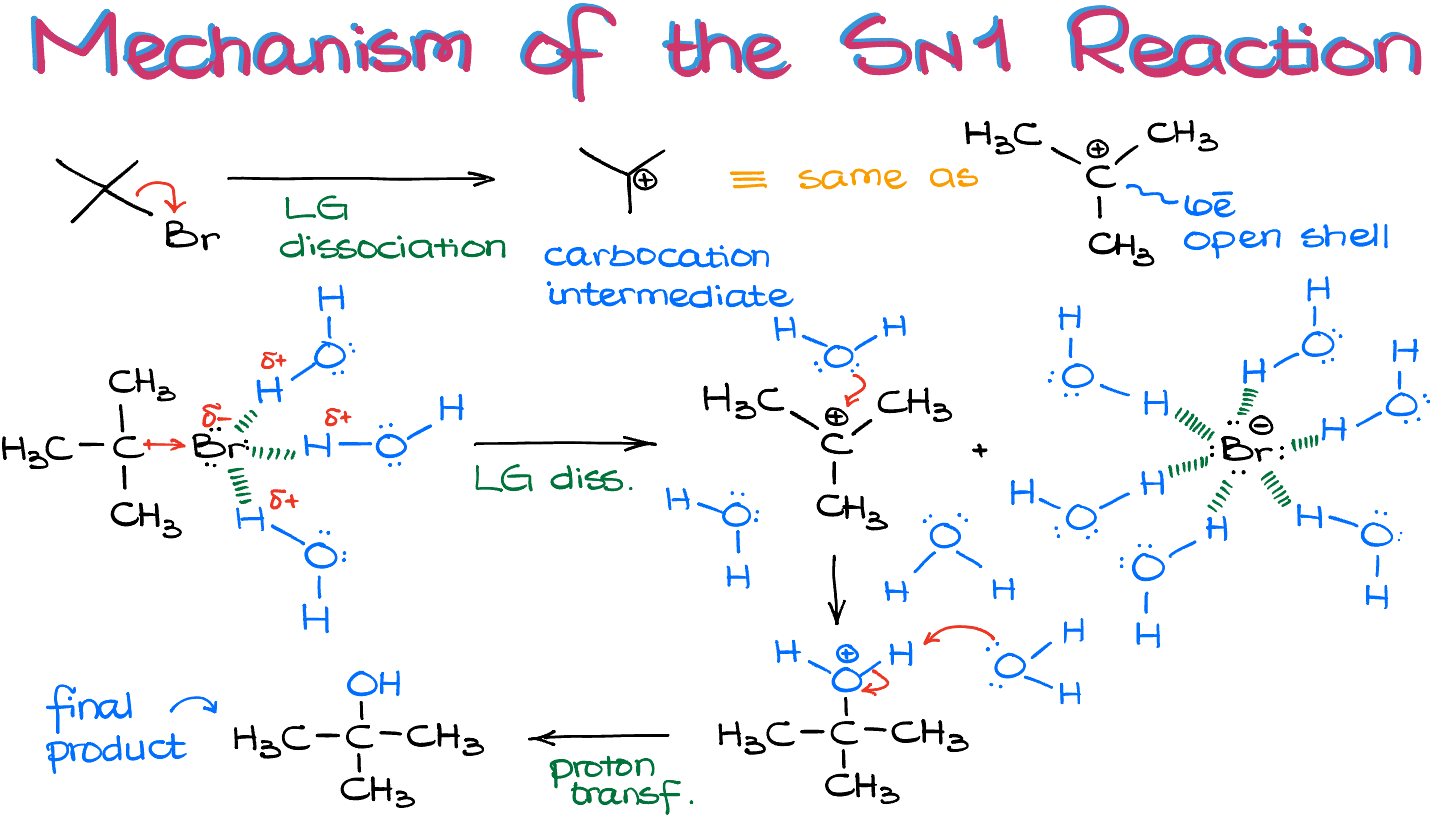
Here’s the really fun part. SN1 reactions are like a shy celebrity. The first step? It happens alone. Yep, the molecule decides to ditch its first "guest" all by its lonesome. No backup needed, no dramatic fanfare. Just… poof! The bond breaks.
This is where our molecule gets a little bit vulnerable. When that bond breaks, the atom that was attached is like, "Whoa, where did my electrons go?!" It’s left with a positive charge. We call this a carbocation. Imagine it like a lonely little dude with a big empty spot where his friend used to be.
This carbocation is the star of the show. It's so important, in fact, that its formation is the slowest part of the whole reaction. This is why we call it SN1. The "1" refers to this unimolecular step. Unimolecular just means it involves only one molecule doing the initial deed. It's the bottleneck, the main event that dictates how fast the entire party gets going.

Why is This Carbocation So Special?
Carbocations are like the supermodels of the chemical world. They're a bit unstable, but they're also incredibly reactive. And they can rearrange themselves! Sometimes, if there's a better spot to be, the positive charge will hop over. It’s like finding a comfier chair at the party. This rearrangement is a quirky fact about carbocations!
So, step one is the departure. The leaving group peaces out, leaving behind our positively charged carbocation. It’s a bit sad, but also… exciting!
The New Arrival: A Dramatic Entrance!
Now that our carbocation is chilling, feeling all positive and a little bit lonely, it’s ready for a new friend. Enter our nucleophile! Think of a nucleophile as a super-friendly, electron-rich party crasher. It’s got extra electrons to share, and it's looking for a positive buddy.
The nucleophile sees that juicy positive charge on the carbocation and is like, "Oh, hello there! You look like you could use some company!" And BAM! It attacks. It swoops in and forms a brand new bond with the carbocation.

This second step is way faster than the first. Why? Because opposites attract! The positive carbocation and the electron-rich nucleophile are drawn to each other like magnets. There’s no hesitation, no second-guessing. It’s a swift, decisive move.
The SN1 Reaction: A Two-Act Play
So, to recap, the SN1 mechanism is basically a two-act play:
Act I: The Lone Departure
- The leaving group bails.
- It leaves behind a positively charged carbocation.
- This is the slow, rate-determining step.
- It's unimolecular.
Act II: The Nucleophile Swoops In

- Our friendly nucleophile arrives.
- It attacks the carbocation.
- A new bond is formed.
- This step is fast.
Why is This So Fun to Talk About?
Because it’s all about how molecules behave! It’s like watching tiny chemical drama unfold. The carbocation is the misunderstood protagonist, the nucleophile is the charming hero (or villain, depending on your perspective!).
And the fact that the first step is the slowest? That’s a classic chemistry quirk! It’s like the nervous moment before a big presentation. Once that’s over, everything else flows much more smoothly. It’s a great way to understand how reactions are controlled.
Think about it: if you want to speed up an SN1 reaction, what do you need to focus on? That first step! Making the leaving group better at leaving, or stabilizing that carbocation. It’s all about understanding the mechanism.
Choosing the Correct Mechanism
Now, you might be asking, "How do we know this is the right mechanism?" Great question! Scientists are super smart and have ways of figuring these things out. They look at things like:
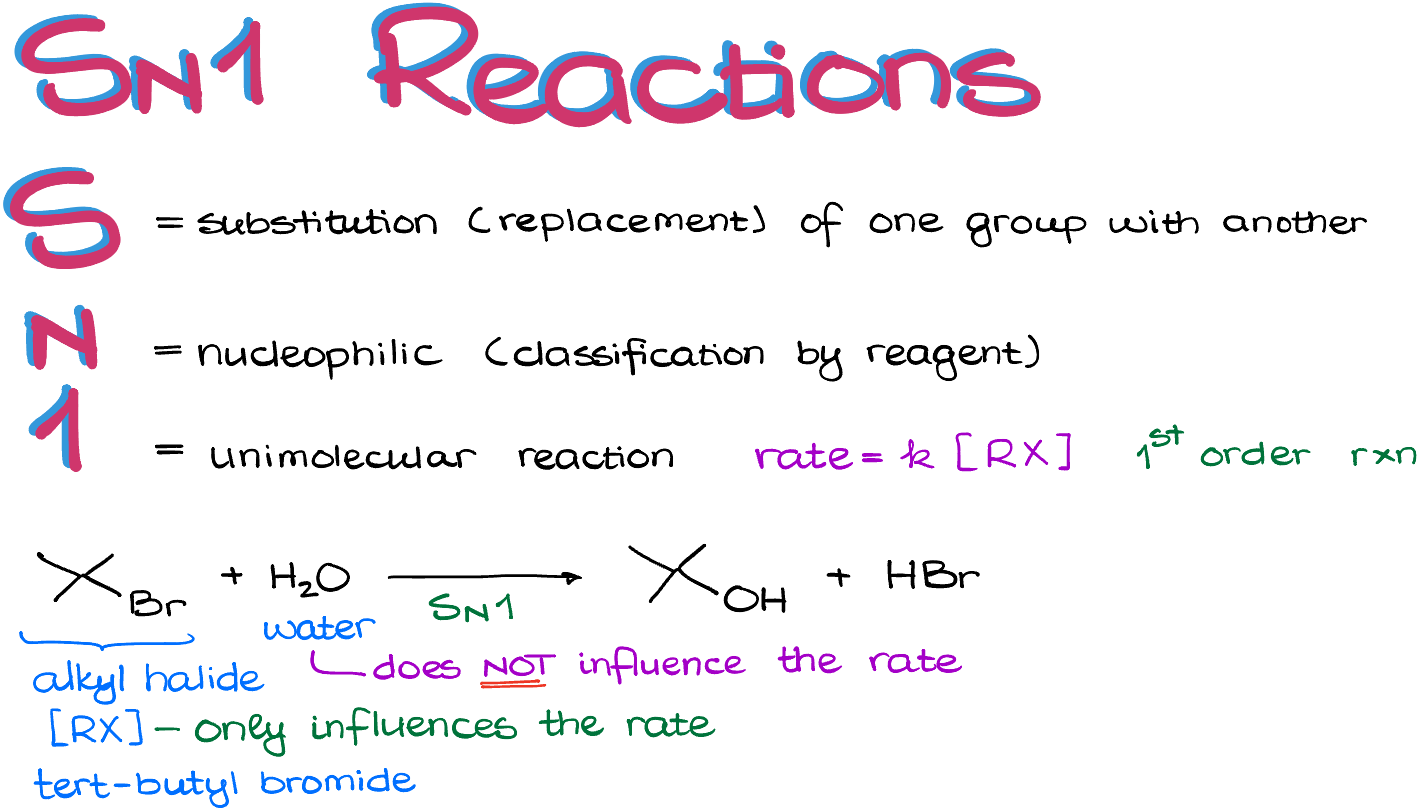
- Reaction Rates: Does the speed of the reaction change if we mess with the concentration of the starting materials? For SN1, changing the nucleophile concentration doesn't usually mess with the speed, because it only shows up in the second, fast step. But changing the concentration of the molecule that loses its leaving group? That definitely speeds things up!
- Stereochemistry: This is the fancy word for the 3D arrangement of atoms. SN1 reactions can sometimes lead to a mix of products, which is another fascinating detail! It's like a fun little spin-off from the main plot.
- Isotope Effects: Sometimes, scientists swap out atoms for slightly heavier versions (isotopes). If that swap affects the reaction rate, it gives clues about which bonds are breaking and when.
So, when you see an SN1 reaction, you're looking for that characteristic two-step process. The lone departure of the leaving group, creating that crucial carbocation, followed by the swift attack of the nucleophile. It’s a dance, a sequence, a story.
It's All About the Steps!
When you're asked to choose the correct mechanism for an SN1 reaction, you're essentially looking for a diagram or description that shows:
- The breaking of the bond between the carbon and the leaving group, forming a carbocation (this is the slow step).
- The attack of the nucleophile on the carbocation, forming the new bond (this is the fast step).
Anything that doesn't show this two-step, carbocation-in-the-middle process is probably not an SN1 mechanism. Maybe it's SN2, or something completely different! But for SN1, remember the solo act, the lonely carbocation, and the speedy reunion.
Isn't chemistry just the coolest? It’s all about understanding the hidden steps, the little dramas that make things happen. So next time you see an SN1 reaction, picture that shy celebrity making its grand, albeit lonely, entrance. It’s a fundamental part of organic chemistry, and honestly, it’s pretty fun to unpack!
