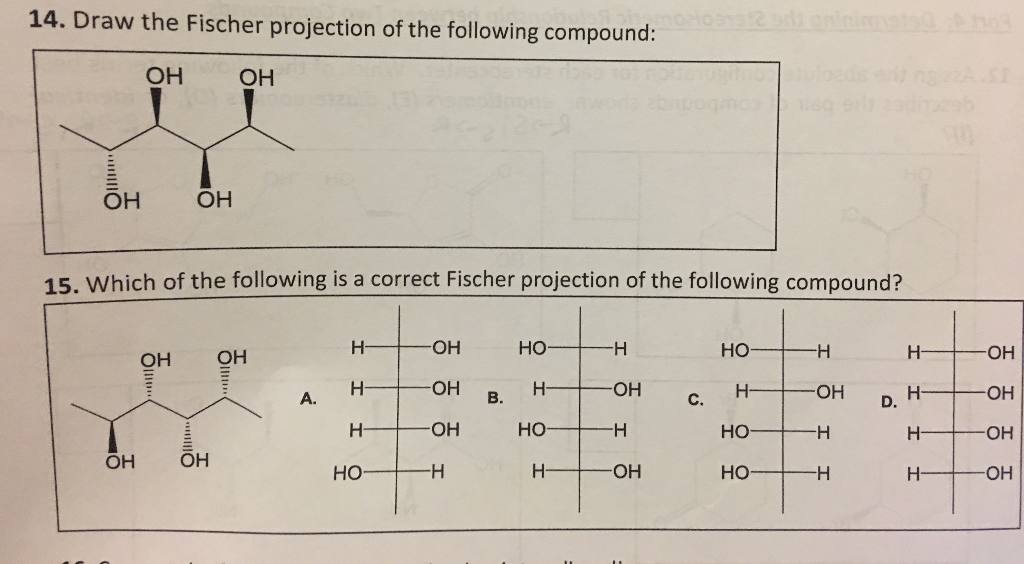
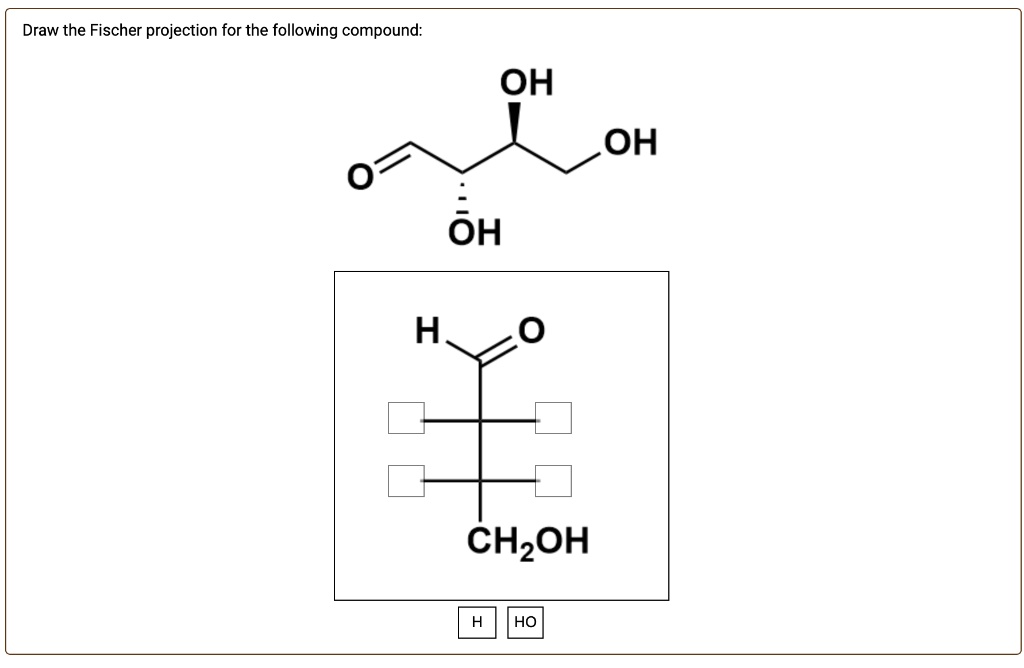
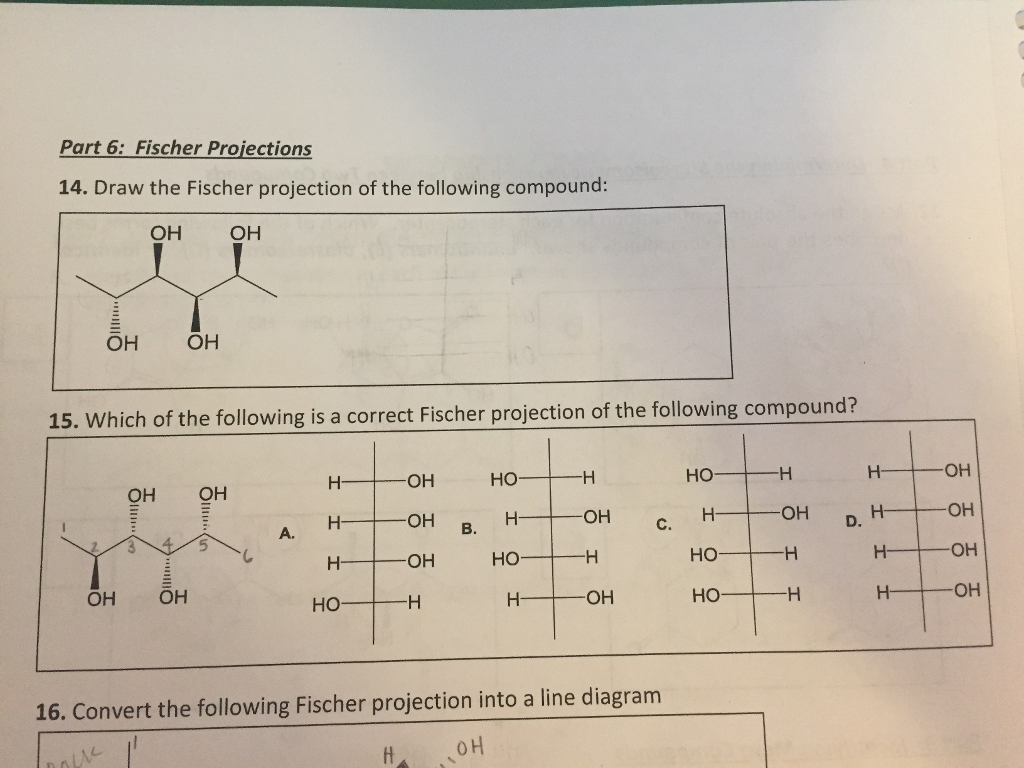
Choose The Correct Fischer Projection For The Following Compound:
Imagine you're a detective, but instead of solving mysteries with fingerprints and magnifying glasses, you're solving puzzles with molecules! Welcome to the fascinating, and sometimes delightfully quirky, world of organic chemistry. Today, we're going to put on our detective hats and tackle a very specific kind of molecular puzzle: choosing the correct Fischer projection. Now, don't let the fancy name scare you. Think of a Fischer projection as a special kind of drawing, like a map, that helps us understand how a molecule is arranged in 3D space. It’s like looking at a house from a bird’s-eye view versus seeing it straight on from the street – both show the house, but they highlight different things!
Our mission, should we choose to accept it (and we totally should, because it's fun!), is to identify the right Fischer projection for a particular compound. Think of it like picking out the perfect outfit for a special occasion. You've got a bunch of options, but only one truly fits the bill, looking just right and representing the true essence of the wearer. Our molecular "wearer" in this case is a molecule with a rather interesting structure. We're not going to get into the nitty-gritty of its chemical name just yet, because the real joy is in the visual detective work!
So, what is a Fischer projection, anyway? Picture this: we're taking a 3D molecule, which can be a bit tricky to draw flat on paper, and we're flattening it out in a very specific way. It’s like taking a tangled ball of yarn and carefully laying it out in a neat crisscross pattern. The horizontal lines represent atoms or groups sticking out towards you, like little hands reaching out from the page. The vertical lines represent things going away from you, disappearing into the background. It’s a clever convention, a secret handshake for chemists, that allows us to talk about the 3D arrangement of atoms without needing complex 3D models all the time.
Now, here's where the fun really begins. We'll be presented with a set of potential Fischer projections. Some will be spot on, like a perfectly brewed cup of tea. Others, however, will be a bit… off. Maybe a limb is pointing the wrong way, or a crucial detail is mixed up. It's our job to spot the imposter! It's like finding Waldo, but instead of a striped shirt, we're looking for the correct arrangement of atoms.
Let's say our target compound has a central atom, often a carbon atom, with four different things attached to it. This is a super common scenario in organic chemistry, and these are called chiral centers. These chiral centers are responsible for a molecule's "handedness," just like your left and right hands. They are mirror images of each other but cannot be perfectly superimposed. Think about gloves – a left glove won't fit your right hand, even though they're both gloves!
When we draw a Fischer projection, we often place the chiral center at the intersection of the cross. The groups attached to it then extend outwards along the horizontal and vertical lines. The crucial part is that rotating a molecule in 3D space can change how it looks in a Fischer projection, but only if we're not careful! We can rotate it around the bonds, kind of like spinning a mobile. However, some rotations are "allowed" and will still represent the same molecule, just viewed from a different angle. Other "rotations" are like trying to force a square peg into a round hole – they change the molecule's identity!
So, how do we become masters of this molecular matching game? One way is to understand the rules of rotation. If we have a Fischer projection, we can perform certain maneuvers to get to another valid Fischer projection of the same molecule. For instance, we can swap two groups on the horizontal line. This flips the molecule's "handedness" in the projection. Or, we can do a 180-degree rotation within the plane of the paper. Imagine the entire cross spinning around its center. These moves are like rearranging furniture in a room – the room is the same, but the arrangement might look different.
What we can't do is just swap groups on the vertical line without consequence, or do a 90-degree rotation. These actions are like changing the fundamental structure of the molecule itself, turning our correct projection into a completely different chemical entity. It’s like accidentally switching the flour and sugar in a recipe – the result will be disastrously different!
Our task, then, is to take the given compound and compare it to the provided Fischer projections. We're looking for the one that is either the direct representation of the molecule, or one that can be reached through a series of valid rotations and swaps from that direct representation. Sometimes, the molecule might be presented in a slightly jumbled 3D format, and we have to mentally (or even with our hands!) rotate it into the standard Fischer projection format before we start comparing. This is where the detective work really shines!
It’s a bit like a puzzle where the pieces are atoms and the goal is to fit them together in the correct 3D puzzle. While the underlying science might seem complex, the process of identifying the correct Fischer projection is a wonderful exercise in spatial reasoning and attention to detail. It’s a small but significant way that chemists unlock the secrets of how molecules behave and interact, leading to everything from life-saving medicines to delicious foods. So, next time you encounter a Fischer projection, don't just see lines and letters; see a tiny, flat map of a 3D world, waiting for you to discover its secrets!
